Abstract
Objectives:
Diabetes in the 21st century presents one of the greatest burdens of disease on the global population. Digitally mediated interventions have become imperative in alleviating this disease epidemic. We aimed to systematically review randomized controlled trials (RCTs) on different health technologies for preventing Type 2 diabetes mellitus, and their efficacy in decreasing diabetes risk-related outcomes in at-risk patients in comparison to standard care.
Methods:
Five electronic databases were searched between October 2021 and December 2022. Studies including digital health technology interventions used for preventing diabetes development by reducing diabetes risk-related outcomes in at-risk adults (⩾18 years) were identified. Data on glycemic levels, incidence of T2DM, weight, and intervention descriptions were extracted, and the risk of bias (ROB) was assessed.
Results:
Nine studies met the inclusion criteria and 5 studies (56%) achieved clinically significant outcomes in at least one of the following: decreased weight (22%), glycemic levels (22%), or incidence of T2DM (11%). Two of the 3 (67%) computer-based interventions effectively reduced the HbA1c levels and mean weight of their study population, and 3 of 6 (50%) mobile based interventions (text messages, mobile app, and telehealth) decreased the incidence of T2DM and HbA1c levels. Four studies each had an overall low ROB and one had a high ROB due to attrition.
Conclusion:
Preliminary evidence identified in our review demonstrated that health technologies for diabetes prevention are effective for improving diabetes risk-related outcomes. Future research into digital technology protocol and studies of longer duration and more diverse populations are needed for clinical feasibility.
Plain Language Summary
In the 21st century, diabetes has become a major health challenge globally. To address this, we conducted a systematic review, looking at studies that used digital interventions to prevent Type 2 diabetes mellitus (T2DM) and how effective they are compared to standard care. In this study, we searched electronic databases from October 2021 to December 2022 and identified 9 studies that met our criteria. These studies focused on adults aged 18 and above who were at risk of developing diabetes. We looked at outcomes like glucose concentrations, T2DM incidence, and weight, and assessed the risk of bias in each study. The results showed that more than half of the studies showed significant outcomes. For instance, some interventions led to decreased weight, lower glucose concentrations, or reduced incidence of T2DM. Computer-based interventions and mobile-based interventions (including text messages, mobile apps, and telehealth) were particularly effective in improving these outcomes. In conclusion, our review suggests that digital health technologies can be effective in preventing diabetes and improving related outcomes. However, we note that more research is needed, especially looking at diverse populations and longer study durations, to confirm the clinical feasibility of these digital interventions for diabetes prevention. This is a promising step forward in using technology to tackle the growing diabetes epidemic, offering new ways to support individuals at risk and improve their health outcomes.
Introduction
Type 2 Diabetes Mellitus (T2DM) is defined as a metabolic disease involving defective insulin action. 1 The resulting hyperglycemia places individuals at high risk for all-cause mortality by health expenditure of around 966 billion USD. 2 An estimated 50% of diabetes cases globally remain undetected or undiagnosed 3 and over half of diabetes’ economic cost lies in its treatment. 2 Diabetes prevention programs (DPPs) are increasingly crucial in mediating this exponential disease epidemic. With healthcare systems’ gradual digitalization, the mediation of DPPs through technological platforms can aid in translating and disseminating these preventative interventions into clinical and public health settings.
Diabetes prevention is traditionally grounded in behavioral change theory, where lifestyle modifications are normalized through educational curriculum, health coaching and peer support in individual and group settings. 4 Landmark clinical trials prove that intensive lifestyle interventions targeting physical activity and healthy diets are effective in reducing the progression from prediabetes to diabetes by 58% in comparison to pharmacological interventions.5-8 They yield long-term results at follow-up with a T2DM reduction of up to 43% that persists for decades for particular populations,5,6 but with the growing global incidence of diabetes, this evidence requires translation into a multifaceted systems approach. 9 In a public health setting, literature indicates the need to develop local health economies that facilitate communication and accessibility between the different levels of health care. 9 At the patient level, barriers to accessibility and adherence need to be overcome.1,10 At the clinical practice level, the increasing prevalence of diabetes-related complications 9 requires a shift of patient management from episodic primary physician checks to a chronic care model. 11 At an administrative level, the economic burden on public health systems can be ameliorated by socio-structural change via health policies, health education 12 and the development of supportive community environments. 13
Digital health technology refers to the use of digital and information technologies, such as mobile devices, wearables, health information systems, software applications, and telehealth services, to improve the delivery and management of healthcare.14,15 It covers a wide range of tools and solutions crafted to improve the efficiency, accessibility, and quality of healthcare services while empowering individuals to actively engage in their health management. The origins of digital health technology can be traced back to the integration of computers into healthcare settings during the mid-20th century. Initially used for administrative tasks, these technologies gradually expanded to encompass clinical and patient care. 16 The rapid evolution of digital health technology was further fueled by the advent of the internet and the widespread use of personal computing devices. Over the past few decades, there has been a notable surge in the development and acceptance of digital health solutions. The widespread adoption of smartphones, wearable devices, and the availability of high-speed internet have been pivotal factors contributing to this growth. 17 Digital health technologies now encompass a diverse array of applications, including electronic health records (EHRs), telemedicine, mobile health apps, remote patient monitoring, health informatics, and the utilization of artificial intelligence for healthcare analytics.18,19
Herman 20 identified the key shared features accounting for the success of major DDP trials. These include individualization of patient interventions, flexibility between group and individual settings, a mixture of physical and behavioral activities and an “extensive network of training, feedback and clinical support.” 1 Evidence indicates that health technologies fulfill all these criteria by extending diabetes prevention from clinical settings to creating an individual’s daily health-protective environment. 10 Health technologies act as intermediary platforms that transfer information between patient and provider whilst normalizing behavioral interventions and lifestyle modifications in the patients’ surroundings and communities.
Regarding computer-based interventions, there is little evidence of its impact on diabetes prevention as software focuses on known diabetes and population management between various health providers, remote patient monitoring and diabetes self-management. 21 A Cochrane meta-analysis comparing 16 computer-mediated diabetes education programs demonstrated a small decrease in HbA1c in patients diagnosed with T2DM and has potential in translation to preventative interventions. 22
There is also a paucity of data on the efficacy of digital health technology in diabetes prevention. Past studies that showed evidence of positive correlations between digitally translated DPPs and improved diabetes risk-related outcomes have been limited by their short duration (5-24 months) and the lack of long-term follow-up. These affect the predictive validity of results by not accounting for fluctuating weight and glycemic levels. The heterogeneity of study location and participant demographic and high variability in program procedure affects the feasibility of public health implementation and scalability as study outcomes may not be generalizable to other national or global contexts. Due to these research gaps, this paper aims to systematically review and compare current evidence about digitalized DPPs’ efficacy in reducing the incidence of diabetes and identify any shared features that may assist in optimizing current interventions and improving health outcomes for at-risk individuals.
Methods
This systematic review was conducted using guidelines from the Cochrane Handbook for Systematic Reviews of Interventions 23 and the protocol was registered with the International Prospective Register of Systematic Reviews PROSPERO (#: CRD42022344100).
Search strategy
A systematic literature search was conducted using 5 electronic databases (PubMed, EBSCO, Ovid Medline, EMBASE and Cochrane Library) in October 2022 with a search strategy structured using the PICO tool 24 consisting of related keywords and medical subject headings (MeSH) as seen below:
Population: T2DM, prediabet*
Intervention: Technology health, digital technology, biomedical technology
Comparison: standard treatment, control
Outcomes: decrease* risk, weight loss, decreas* BMI, decreas* blood glucose, decreas* hyperglycemia, improv* glycemic control, decreas* waist circumference
Boolean search terms “OR” and “AND” enforced a distinction between specified and generalized terms. Filters limiting peer-reviewed journal articles to randomized controlled trials (RCTs) and study recency to the last 10 years were applied. The final search strategy was as follows: (((MM T2DM OR prediabet*[MeSH Major Topic]) AND (standard treatment OR control)) AND (MM Technology, health OR digital technology OR biomedical technology)) AND (decreas* risk OR weight loss OR decreas* BMI OR decreas* blood glucose OR decreas* hyperglycemia OR improv* glycemic control OR decreas* waist circumference). The same search strategy was replicated across the various databases.
Eligibility criteria
Titles and abstracts of studies were screened by 2 independent reviewers (VN, PA) for eligibility. Results were compared to finalize selection and any disagreements were resolved through discussion. Studies were eligible for inclusion if they: (i) assessed adult (>18 years) populations who were at risk of developing diabetes; (ii) involved technology-based interventions which could be fully digitally-mediated or delivered in a mixed digital and face-to-face format; (iii) involved interventions explicitly aimed at reducing the risk of diabetes development; (iv) had outcome features related to body weight, glycemic levels or T2DM incidence (decreased BMI, decreased waist circumference, weight loss percentage, decreased fasting blood glucose or HbA1c); (v) in the RCT study format; and (vi) are available in English with full text.
Exclusion criteria included: (i) studies involving populations with diabetes diagnoses; (ii) interventions with no digital technology component; and (iii) interventions aimed at managing existing diabetes rather than preventing diabetes incidence.
Data extraction
Due to the small scale of the systematic review (2 reviewers and less than 20 studies), data was stored using standard data extraction templates on Google Forms based on Cochrane guidelines. 23 Data was extracted from all studies by 1 reviewer (VN) then a second reviewer (PG) independently repeated extraction on a random 20% of selected studies to confirm accuracy. Disagreements were resolved with discussion. Data about primary outcomes related to reducing diabetes risk were sought for extraction to compare intervention efficacy. Studies were grouped by these primary outcomes (decreased BMI, weight, glycemic levels, and T2DM incidence) and the mean and standard deviation of baseline and end-of-study results were tabulated with P-values and confidence intervals, where available, to compare statistical significance. Study variables extracted include the following intervention characteristics: author, year of publication, population sample size, study design, country of study, the study’s definition of “at-risk” and intervention characteristics, primary outcome unit of measurement and study duration. Narrative synthesis was chosen to summarize these intervention characteristics and outcomes due to clinical heterogeneity (population baseline characteristics and intervention methods) and methodological heterogeneity.
Assessing bias
The quality of selected studies was assessed for risk of bias (RoB) using the Cochrane Collaboration Tool 25 by 1 reviewer (VN). A random 20% of studies were screened by a second reviewer (PG) to confirm reliability. Disagreements were resolved with discussion. Studies were rated either “low risk,” “some concerns” or “high risk” over 5 different domains. They were deemed “low risk” if they scored “low risk” on all domains or if the domains of “some concern” were highly unlikely to affect study results and deemed “high risk” if they scored “high risk” in at least 1 domain. Results were presented in Robvis 26 traffic light and weighted bar plots.
Quality of evidence was assessed using the GRADE scale 27 where reviewers’ confidence in the similarity of the true effect and the estimated effect were graded by 4 levels of certainty—“high,” “moderate,” “low,” and “very low.”
Results
Reporting of the study findings followed the Prisma protocol as shown in the checklist (see Supplemental Table 1). The PRISMA flow diagram 28 shown in Figure 1 details the process of study selection and reasons for exclusion. The initial literature search identified 226 articles and after duplicates were removed 151 publications were screened by title and abstract. Forty studies were sought for retrieval and 1 study was not available on public databases. Thirty-nine studies from the following databases, PubMed (8), Ebsco (3) Ovid Medline (14), Cochrane (6), and other sources (8) underwent double screening and were assessed for eligibility after which 9 studies were included in this systematic review.

Flowchart of study selection.
The basic characteristics of the reviewed studies are summarized in Table 1. Four of the studies that were included in the full-text review were from PubMed, 3 from Cochrane, and 2 from Medline libraries. Many of the studies (n = 44.4%) were conducted in America29-33 with a single study site each in New Zealand, 34 India, 35 Hong Kong, 36 Bangladesh, 37 and 1 jointly conducted in the UK and India. 31 Eight out of 9 studies were parallel 2-arm RCTs29-37 while 1 study was a 3-arm cluster RCT. 37 Six studies recruited participants from primary healthcare settings29-34, 3 studies recruited from the workplace,32,35,36 and 1 from the community. 37
Characteristics of the selected articles.

Abbreviations: BMI, body mass index; DPP, diabetes prevention program; FBG, fasting blood glucose; HbA1c, hemoglobin A1c; RCT, randomized controlled trial; T2DM, type 2 diabetes mellitus.
Eight (89%) out of 9 studies’ populations were of mixed gender29-34,36,37 with 1 study population being exclusively male. 35 The 4 American studies defined an individual at risk of diabetes or in the prediabetes stage with HbA1c scores of 5.7% to 6.4% or 39 to 46 mmol/mol.29-31,33 Other studies recruited participants in the upper prediabetes range (HbA1c > 5.9%) 32 or defined at-risk populations by BMIs greater than 23 kg/m2, positive family histories of type 2 diabetes mellitus (T2DM), oral glucose tolerance test scores (14-200 mg/dL) or fasting plasma glucose scores (100-125 mg/dL). 35
Intervention methods varied across studies where differing combinations of health coaching, educational resources, peer support groups and goal tracking were mediated through various digital platforms.29-37 A single intervention 31 used individually tailored websites and interactive emails while 7 interventions29,30,32,33,35-37 used mobile devices via text or voice messages,29,32,33,35-37 telephone-mediated coaching 29 or mobile applications.30,34 Digital technology was supplementary to in-person or online coaching by a health coach in 5 studies29,30,33,34,37, four studies were automated and algorithm-driven.31,32,35,36
Table 2 summarizes the primary outcomes and results of the included studies. Three studies31,33,34 determined intervention efficacy by changes in HbA1c levels from baseline to the end of 12-month interventions with Block et al 31 demonstrating significant reductions in HbA1c (−2.8 mmol/mol [95% CI −0.27, −0.24], P = .002) from a DPP mediated through individually tailored websites and interactive emails. Katula et al’s 33 study demonstrated a shift in HbA1c levels from the prediabetes to the normal range (−0.23% [95% CI −0.26, −0.20]) due to an internet-accessible device (mobile, computer, smartwatch) mediated DPP, while McLeod et al’s 34 mobile and web-based individual and peer support coaching intervention showed no difference between study arms.
The primary outcomes of the reviewed randomized controlled trials.

Abbreviations: BMI, body mass index; DPP, diabetes prevention program; FBG, fasting blood glucose; HbA1c, hemoglobin A1c; RCT, randomized controlled trial; T2DM, type 2 diabetes mellitus; RR, risk ratio.
The primary outcome of the 2 studies29,30 was a percentage change in weight or BMI. Fukuoka et al’s 30 mixed intervention with in-person DPP sessions, supplementary interactive mobile app and pedometer use proved effective in weight loss (6.8% [95% CI,4.6,9.1]) while Fischer et al 29 proved the efficacy of text-message enhancement of DPPs in Spanish populations with a comparable portion of the study population losing 3% baseline weight (47.1% [95% CI 33.4-60.8]) compared to English speakers (20.6% [95% CI 7/0-34.2]).
Four studies32,35-37 examined an outcome of diabetes incidence in relation to DPP interventions mediated over mobile phone messaging with 3 studies32,35,36 yielding clinically significant results. Ramachandran et al 38 and Wong et al’s 36 studies examining behavioral change theory-oriented text messages both affected the lower cumulative incidence of T2DM in comparison to standard care (−18% [hazard ratio 0.64, 95% CI 0.45, 0.92; P = 015]; −11.11% [95% CI, 0.24, 1.61, P = .021]). Fortrell et al’s 37 voice-messaging intervention and Nanditha et al’s 32 contrastingly, showed no evidence of effect between intervention and control cohorts.
Risk of bias
Using Cochrane Collaboration Tool’s RoB 2, 25 Figure 2 details the result of assessment by domains for the selected studies. Four studies were identified with an overall low ROB,29,33,35,37 another with a high ROB due to attrition (incomplete follow-up and patient retention) 36 and for 4 other studies, there were some concerns of attrition bias (patient non-compliance or retention) to a lesser degree.30-32,34 There was no selection bias in the reviewed studies due to the adequate randomization and allocation concealment in the studies. Regarding performance bias, by nature of the interventions, the majority of patients and caregivers could not be blinded but this did not affect study outcomes. Detection bias was present in 2 studies whose outcomes were not statistically significant29,36 and no reporting bias was identified as all measured outcomes were reported in the final analysis.
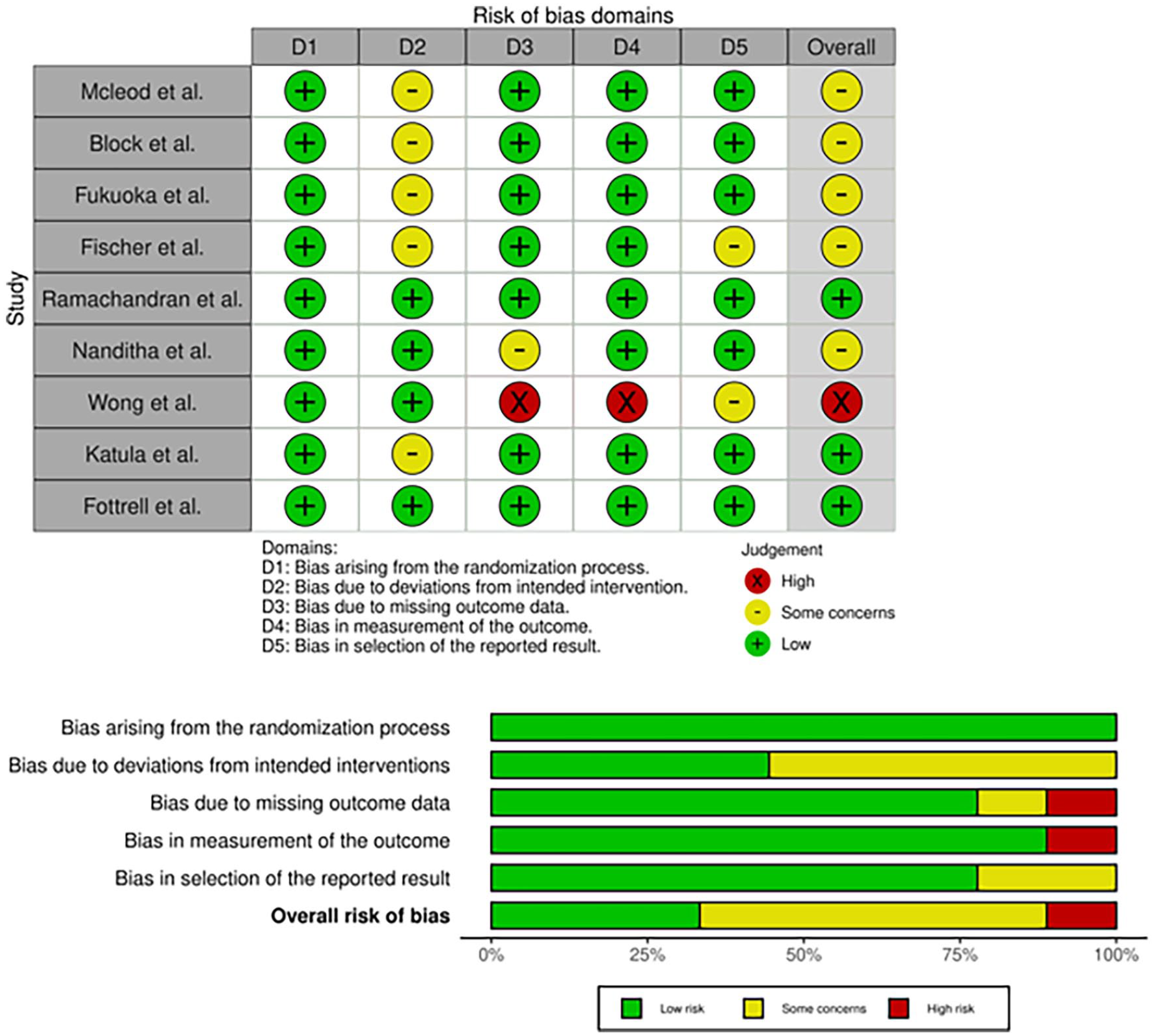
Risk of bias assessment for the various domains.
Discussion
This review study identified 5 studies30,31,35-37 that showed clinically significant reductions of various diabetes risk related outcomes especially glycemic levels, weight, BMI and reduced incidence of T2DM, possibly strengthening available literature on the efficacy of 2 main digital modalities (computer and mobile devices) in diabetes prevention. Despite the statistical significance of these RCTs, the studies were significantly heterogeneous in their outcomes due to the large variation in the intervention methods. The study findings suggest that the efficacy of individual DPPs may be due to the synergistic effect of various modes of delivery (technological and in-person) rather than the superiority of one modality over the others. However, due to the small number of studies about each digital modality, the impact of these variations in the intervention could not be properly assessed.
In this study, there were 2 RCTs31,33 involving computer-based DPPs that yielded statistically significant results, proving the capacity of computers to fully automate DPPs or to act in a supplementary role – providing resources alongside in-person curriculums. For one study, 31 after 6 months, the use of individualized websites and interactive emails improved the glycemic level, body weight and 8-year diabetes risk of people with prediabetes. The use of computer-mediated peer support groups, tracking and behavioral change curriculums yielded significant weight loss at 12 months, 33 whereas mobile-mediated interventions led to positive outcomes such as greater weight loss over time and reduction in the risk of developing diabetes at 12 months.30,35,36 A text-messaging intervention where delivered content was individualised 39 and one where messages were standardized to collective participant preferences, 36 both decreased the cumulative incidence of T2DM. In agreement with previous evidence,39,40 supporting in-person DPP sessions with a mobile app for tracking progress and mediating educational resources achieved clinically significant weight loss, 30 thus affirming the strength of mobile-mediated interventions in increasing patient adherence through positive reinforcement.39,40 This has also been linked to high retention rates in clinical trials.29,30,32,33,35-37
On the other hand, mobile-mediated DPPs are effective in consistently reinforcing patient education,39,41 self-monitoring and social support 39 which prevents non-adherence. The high global levels of smartphone ownership 40 further decrease economic expenditure and accommodate a widespread dissemination of intervention programs. Four text message-based programs focused on stage-based behavioral change were shown to significantly reduce the progression of prediabetes to diabetes with preventative outcomes of weight loss and diet logging.29,35,38,42 In examining a broad digitalization of DPPs, a 2017 meta-analysis correlates smartphone apps mediating health professional personal contact with increased weight loss, in comparison to complete automated contact. 43 Evidently, by replacing in-person group exercise sessions with phone-app delivered home regimens, Fukuoka et al’s 30 intervention yielded no change in glucose levels, supporting how retaining partial human factors in delivery is still imperative in patient adherence and response.
Regarding the effect of computer-based therapies in decreasing HbA1c in at-risk patients, the evidence is judged to be moderate in quality. The studies have a generally low risk of bias besides some concerns of attrition for participant retention.31,33 There is no great imprecision, indirectness, or publication bias but the heterogeneity of the intervention method and the data of statistical significance only being sourced from 3 studies, lowers confidence in the review’s results. For mobile-based therapies and their impact on reducing the incidence of T2DM, the evidence is judged to be of low quality. There is moderate to high risk in 2 out of 3 studies of statistical significance and the sample size in those studies was small (61 and 104 participants).30,36 The RCTs have no obvious imprecision, indirectness, or publication bias but the heterogeneity of intervention and study protocol were variable.
Digital health technology interventions play a pivotal role in augmenting existing strategies for the prevention of type 2 diabetes mellitus (T2DM). These interventions leverage innovative technological solutions to enhance outreach, education, monitoring, and management of individuals at risk for T2DM. By incorporating mobile applications, wearable devices, and online platforms, digital health technologies provide personalized and real-time interventions, promoting lifestyle modifications and behavior change. These tools facilitate continuous glucose monitoring, physical activity tracking, and dietary management, empowering individuals to take an active role in their health. Moreover, digital interventions enable healthcare providers to remotely monitor patients, offer timely if not real-time feedback, and tailor interventions based on individual progress. The integration of data analytics and machine learning further refines predictive models, identifying high-risk populations and customizing intervention strategies. In essence, digital health technology interventions not only complement but also transform conventional approaches to T2DM prevention, fostering a more dynamic, individualized, and proactive healthcare paradigm.
Implications for practice
This study provided evidence for the generalized efficacy of health technologies in preventing diabetes by reducing risk-related outcomes as 5 studies within this review demonstrated statistically significant reductions in HbA1c and diabetes incidence. Further research is needed to translate this data into clinical settings. The feasibility of digitally mediated DPPs is supported by the gradual modernization of the healthcare system and public accessibility to technology but this can be better understood with more pragmatic trials involving diverse populations or interventions that are culturally adapted (which showed a positive outcome in one study. 29 We also provided evidence of appropriateness, as diabetes prevention involves self-management and literature which identifies mobile-devices efficacy in increasing patient engagement39,40 and the high retention rates of studies within this review29,30,32,33,35-37 support this. Evidence of meaningfulness was unavailable as patient-rated outcomes were not examined in this review and effectiveness is difficult to determine with the moderate to low quality evidence of the chosen studies.
Establishing a sustainable infrastructure for digital diabetes prevention programs is imperative for its long-term success. This infrastructure should prioritize interoperability with existing health information systems, ensuring seamless data exchange and continuity of care. Robust cybersecurity measures, including secure data storage and transmission, are essential to protect patient information and comply with healthcare data protection regulations. The use of scalable, cloud-based architecture enables flexibility and accommodates a growing user base, while user-friendly interfaces cater to a diverse range of users. Integration with wearable and IoT devices allows for real-time data collection, and analytics tools provide insights for continuous program improvement. Training modules and support mechanisms ensure both healthcare providers and end-users can effectively utilize the program. Regulatory compliance frameworks, reliable telecommunication infrastructure, and continuous monitoring and maintenance further contribute to the infrastructure’s resilience. By addressing these components comprehensively, digital diabetes prevention programs can seamlessly integrate into healthcare systems, promoting sustained engagement and positive health outcomes.
Limitations and future research
The following limitations should be considered when interpreting this data. First, there was a small number of studies that met the eligibility criteria in this systematic review and the heterogeneity of study design (population, intervention mode of delivery, method, duration) collectively led to difficulty in comparison and inability to perform a meta-analysis to assess the strength of data. Although we have restricted the studies to increase the robustness of our study, future studies might include other non-RCTs for a better understanding of the impact of DPPs. Second, the study populations in the included studies lacked diversity which could affect the scalability and accessibility of these interventions. The fact that some studies were single-gendered, others recruited from specific workplaces or ethnic groups without cultural adaptation of the DPP program, may have affected the participant engagement. Third, the study durations were short ranging from 5 to 36 months with the longest follow-up at 24 months and both these factors limit the external validity of DPP interventions which target a chronic and global health issue. Also, the reviewed studies were all written in English including the Hong Kong Study. This could potentially exclude crucial findings from studies not written in English. Articles not written in English from non-English speaking jurisdictions should also be reviewed in future studies.
In this study, we identified a research gap with studies that examined specific aspects of health technologies for increased patient engagement. Two studies imply the importance of partial human factors in interventions, attributing high retention rates to healthcare staff delivering the intervention 27 and the participatory in-person activities. 37 Therefore, studying the interactive nature of digital and in-person modalities may optimize intervention protocol. Understanding the difference between personalized or standardized interventions and full automation or partial digital mediation is important and can be further explored in future studies.
An examination of diverse digital health approaches across different populations, healthcare systems, and cultural contexts is needed to develop targeted and culturally sensitive digital intervention solutions. Also, research should focus on identifying determinants of successful implementation, such as user engagement, usability, and adherence, to optimize intervention designs for maximum impact. Thirdly, continuous evaluation of digital interventions over the long term is crucial to assess their effectiveness and sustainability, informing potential updates or modifications. Fourthly, exploring the integration of digital health technologies into existing healthcare systems, considering interoperability, data security, and impact on workflows, is essential for successful implementation on a broader scale. Lastly, investigating the cost-effectiveness and scalability of these interventions will be vital for their widespread adoption and integration into routine healthcare practices.
Conclusion
In conclusion, this review supports the current literature’s generalized perspective on the efficacy of health technology in diabetes prevention and presents some evidence of positive correlations between digitally translated DPPs and improved diabetes risk-related outcomes. Yet, the heterogeneity in intervention protocol and study procedure and the lack of diverse populations or long-term data and follow-up undermine the possibilities of clinical effectiveness and the feasibility of public health implementation and scalability due to the risk of economic burden and the chance of patient non-adherence. Future research should include studies of larger diverse populations with longer duration and follow-up data and specific aspects of health technologies that increase patient engagement and the possibility of clinically significant outcomes.
Supplemental Material
sj-docx-1-end-10.1177_11795514241246419 – Supplemental material for The Role of Digital Health Technology Interventions in the Prevention of Type 2 Diabetes Mellitus: A Systematic Review
Supplemental material, sj-docx-1-end-10.1177_11795514241246419 for The Role of Digital Health Technology Interventions in the Prevention of Type 2 Diabetes Mellitus: A Systematic Review by Vivien Nguyen, Paige Ara, David Simmons and Uchechukwu Levi Osuagwu in Clinical Medicine Insights: Endocrinology and Diabetes
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
