Abstract
Background:
The reproductive system is heavily dependent on ovarian follicles, which are made up of germ cells (oocytes) and granulosa cells (GCs), including cumulus granulosa cells (CGCs) and mural granulosa cells (MGCs). Understanding their normal and steroid-induced functions is the key to understanding the pathophysiology of endocrinal diseases in women.
Objective:
This study investigated the differentially expressed proteins by CGCs and MGCs of patients with polycystic ovarian syndrome (PCOS) and without subsequent exposure to dehydroepiandrosterone sulfate (DHEAS) and functional differentiation.
Design:
The present study was observational and experimental study carried out in hospital involving 80 female patients undergoing IVF for infertility.
Methods:
In this study, we isolated CGCs and MGCs from the follicular fluid of both PCOS and non-PCOS patients undergoing in vitro fertilization (IVF). The cells were cultured and treated with DHEAS for 48 hours, and these cells were extracted, digested, and analyzed by tandem mass spectrometry followed by processing of the results using open-source bioinformatics tools.
Results:
The present investigation discovered 276 and 341 proteins in CGCs and MGCs, respectively. DHEAS reduced the number of proteins expressed by CGCs and MGCs to 34 and 57 from 91 and 94, respectively. Venn results of CGCs revealed 49, 53, 36, and 21 proteins in normal CGCs, PCOS-CGCs, post-DHEAS, and PCOS-CGCs, respectively. Venn analysis of MGCs showed 51 proteins specific to PCOS and 29 shared by normal and PCOS samples after DHEAS therapy. MGCs express the most binding and catalytic proteins, whereas CGCs express transporter-related proteins. A protein pathway study demonstrated considerable differences between normal and PCOS samples, while DHEAS-treated samples of both cell lines showed distinct pathways. String findings identified important network route components such as albumin, actin, apolipoprotein, complement component C3, and heat shock protein.
Conclusion:
This is the first study to show how DHEAS-induced stress affects the expression of proteins by MGCs and CGCs isolated from normal and PCOS patients. Further studies are recommended to identify PCOS biomarkers from CGCs and MGCs expressed under the influence of DHEAS.


Introduction
Several difficult problems in obstetrics and gynecology have been explored using the promising new field of scientific breakthrough “OMICS” technology. 1 These methods rely on only a small quantity of biological material by performing a single experiment with cutting-edge molecular biology tools. 2 Although proteome research has traditionally been used in biomedical engineering for 2 distinct but overlapping purposes—identifying novel treatment targets and studying biomarkers—these 2 lines of investigation now often intersect. When proteins are examined closely, causal links are established between the normal state and pathological condition of the cells/tissues. The presence or absence of such roots is a dysfunction prominent to a disease, and these proteins become effective indicators that are further termed markers.3,4 These markers are also helpful in creating genetic maps, planning a treatment, assessing risk, and determining the optimal dosage of a drug to obtain the desired effect. For this reason, typical biomarker-based diagnostics do not rely as much on human judgment; they minimize the bias in diagnosis, allowing for greater specificity and sensitivity. Biomarkers are indicators that have the ability to predict the extent of the illness and responses to therapy along with the diagnosis. 5
Polycystic ovarian syndrome (PCOS) affects approximately 5 to 20% of women worldwide and is the leading cause of infertility in young adults, and its pathogenesis is still poorly understood. 6 In addition to the increased risk of diabetes mellitus, endometrial cancer, cardiovascular disease, and coronary artery disease, the presence of hyperandrogenism and hyperandrogenemia, hyperinsulinemia, insulin resistance, oligo- or anovulation, polycystic ovarian morphology, hirsutism, and so on, are the hallmarks of this ailment. 7 One of the classical and most recognizable signs of PCOS is hyperandrogenism, a condition that affects nearly 60 to 80% of all PCOS patients. 8 Because of insulin resistance and anovulation, it develops when ovarian cells secrete abnormal amounts of steroid hormones. 9 Anovulation, hypothalamic-pituitary-gonadal tract dysfunction, polycystic ovarian morphology and reproductive competency are all negatively impacted by steroid levels in the body; thus, it is important to keep them in check to ensure proper ovarian functioning and health. 10
Proteins, lipids, growth factors, mucopolysaccharides, steroidal hormones such as follicle-stimulating hormone (FSH), luteinizing hormone (LH), human chorionic gonadotropin (hCG), estrogen, progesterone, and dehydroepiandrosterone sulfate (DHEAS) and other endocrine secretions of the body all work together for the development of ovarian follicles in mammals. 11 These factors affect cellular and metabolic factors involved in the various developmental phases, including oocyte meiosis, ovarian cell differentiation, oocyte development within follicles, follicle maturation, and ovulation. 12 Therefore, the health of follicle cells is vital for reproduction in females and is used as a source of physiologically active components that promote follicular development, ovulation, and fertilization of the egg. Therefore, these follicular cells (which are composed of a variety of somatic cells and germ cells) can be used as disease biomarkers, and their metabolic and physiological states may be determined by analyzing alterations in certain components.13,14
A follicle is made up of gametes cells (ovule) and specialized complementary somatic cells, granulosa cells (GCs), stromal cells, and thecal cells. GCs comprise 2 types of cells, mural granulosa cells (MGCs) and cumulus granulosa cells (CGCs), which secrete various nutritive growth factors for the maturation of oocytes and communicate with neighboring cells via gap junctions; these cells also have an essential role in follicle development and preventing follicular atresia. CGCs and MGCs arise from the same progenitor but have differential locations inside the follicle and physiology. 15 After ovulation, MGCs start releasing progesterone, and during the follicular phase, they release estrogens. CGCs, which form the outermost layer, are called the cumulus-oocyte complex (COC) and penetrate the zona pellucida matrix by transzonal cytosolic projections. 16 These cells also have a great role in oocyte quality extrapolation, maturation of oocytes and initiation of the steroidogenesis process. 17 All sex steroids serve essential roles in the maturation of germ cells and sustaining whole body healthiness in addition to specialized operative functions in reproductive tissues and maintenance of pregnancy. 18 It has been revealed that androgens play a measurable role in the course of folliculogenesis by stimulating follicular growth, differentiation of GCs, production of estradiol and progesterone, and facilitating oocyte maturation. 19
The composition and abundance of the protein in ovarian follicles, including GCs, are not well understood, despite the growing importance of proteomic studies in deciphering biological processes. 20 Regardless of being the major target of androgens, GCs express the highest levels of androgen receptors in the prenatal and antral follicles of monkey ovarian granulosa. 21 These receptor expression levels could be affected by the elements communicated to the follicles through the local microenvirons. However, the mammalian steroidal hormone DHEAS possesses an integral segment for sexual characteristic development and reproduction in females. The unconjugated form of the DHEA hormone is thought to be involved in biological functions by interacting with its receptors, while the conjugated form of the sulfate group has been intended to be used solely for excretion. 22 Conjugated steroids are inert reservoirs meant for steroid hormones; however, as humans produce a significant amount of sulfonated steroids, particularly during pregnancy, it is still unclear what their physiological purpose is. The brain, adrenal medulla, and gonads are the organs where DHEA is typically synthesized in the human body. Cholesterol serves as the key precursor for this production in all parts. 23
Although the specific physiological role of DHEA and DHEAS is unknown, these steroid hormones are capable of functioning as androgens, estrogens, and neuro-steroids and play a variety of roles in the human body. DHEA is the foremost source of natural estrogens and the most prevalent circulating steroid hormone in people. 24 It is transformed into testosterone in the ovary connective tissue (theca/stroma), followed by the granulosa cells metabolizing it to produce estradiol. As a result, it takes the position of prohormone, which is a metabolic intermediate in the production of ovarian follicular steroids and possesses a major endogenous precursor. 25 Few in vitro methods have been developed to examine the impact of sulfonated DHEA at the cellular level. 26 However, the exact effect of DHEAS on MGCS and CGCs and their differential protein expression have not yet been discovered.
The vast bulk literature on proteomic studies of women’s follicular fluid has focused solely on the analysis of fluid obtained from females who underwent intracytoplasmic sperm injection (ICSI)/in vitro fertilization (IVF) through controlled ovarian stimulation. Thousands of proteins were reported as being identified in these studies, the huge majority of which are linked to plasma composition and the acute phase response/complement cascade. 27 These studies are limited to follicular fluid and have not yet explored any steroidal effects on fluid compositions or effects at the cellular level. Because ovarian follicles are essential components of the reproductive system and contain the eggs (female gamete) as well as a number of specialized supporting somatic cells, it is crucial to understand their functioning and the influence of the steroid hormone on them. To the best of our knowledge, no research has been done to date using proteome analysis to examine the effects of DHEAS on MGCs and CGCs in PCOS patients in comparison to healthy women.
Therefore, to acquire more information about the proteins and genes associated with PCOS and determine the molecular basis, this study was designed. The primary rationale behind incorporating DHEAS into this study on PCOS is its versatile character, facilitating the conversion into androgens (such as testosterone and androstenedione) and estrogen. 28 The conjugated variant of DHEA possesses superior solubility and an extended half-life compared to DHEA. 29 Its hypothesized functions encompass the regulation of follicle maturation, contribution to the development of female reproductive organs, and participation in governing the ovarian cycle. The primary objective of this study was to investigate the differential protein expression of GCs with and without exposure to DHEAS to learn more about the proteins and genes associated with PCOS. Another objective for carrying out this research was to find distinctions between CGCs and MGCs, elucidate their proteomes for functional differentiation and their individual specific relations in the development of PCOS. Upon protein analysis, we can acquire more knowledge about the hereditary makeup and biochemical mechanisms that contribute to PCOS. The investigation's findings will provide perceptiveness into the molecular importance of DHEAS in GCs and may serve as an argument for the defining and creation of novel ideas pertaining to the treatment of the hyperandrogenic local ovarian environment.
Materials and Methods
Study participants and center
This study was conducted between March 2021 and October 2022 at Indira IVF Fertility Centre and Morpheus Prasad International IVF Centre, Dehradun (Uttarakhand)-India. Eighty women with PCOS and 80 non-PCOS women matched controls of the same ethnicity, age range, and BMI were included in this study. Protein estimation was performed at the Department of Biosciences and Bioengineering, IIT Roorkee (Uttrakhand), India. The Mass Spectrometry Facility was provided by Advanced Technology Platform Centre (ATPC), managed by the Regional Centre for Biotechnology (RCB), Faridabad (Haryana)-India for this research.
Inclusion criteria
The women with PCOS were defined and selected as per the Rotterdam Criteria. 30 Women without gynecological issues who undergo IVF/ICSI due to male-factor infertility otherwise normal constitute a control group.
Criterion of exclusion
Based on the patient’s history and present clinical record, androgen-producing tumors, hyperprolactinemia, active thyroid illness, and Cushing syndrome were excluded from the study. Women taking oral contraceptives, anti-hypertensive, anti-inflammatory, and lipid-lowering medicines for at least 3 months before the study were additional exclusion criteria for both PCOS and control women. Patients who disagreed to sign an informed consent form were also excluded from the research.
Bioethics
All the participating patients provided written informed consent for the complete study with their preferred language, and the University Research and Ethics Committee (UREC) of DIT University, Dehradun approved the protocol for the use of leftover specimens from patients for the overall study (DITU/UREC/2019/07/2 on July 9, 2019). All data were anonymized and kept confidential.
Stimulation and collection of follicular fluid from the patients
PCOS patients and healthy controls were identified using the abovementioned criteria. Controlled ovarian stimulation performed as per the clinical short- or long-term gonadotropin-releasing hormone agonist protocol was part of IVF/ICSI. Follicular fluid was obtained (average 4.1 ± 0.3 punctures), and egg retrieval was conducted after 36 hours under mild anesthesia. Each subject’s follicular fluid was collected in 15 mL round-bottomed polypropylene centrifuge tubes, and the temperature was maintained at 37°C for further processing.
GCC isolation
GCs were extracted from follicular aspirates following the standard procedure reported previously.31,32 In brief, after oocyte extraction from COCs, the cumulus aggregate was meticulously removed using a hypodermic syringe. The residual fluid and cellular mixture were added into the test tubes at this stage. The separated CGCs were then washed multiple times with phosphate-buffered saline (PBS) (HiMedia) and neutralized with DMEM/F12 medium (a 1:1 mixture of DMEM with Ham’s F-12 is DMEM/F-12. This mix includes the high proportions of glucose, amino acids, and vitamins and within DMEM, the vast variety of biological components found in F-12).
The cell viability and count were evaluated using trypan blue (Sigma‒Aldrich, USA) staining. Subsequently, the cells were placed into a T25 tissue culture flask containing DMEM/F12 medium supplemented with 20% fetal bovine serum (HiMedia), 1% antibiotic-antimycotic solution (Sigma‒Aldrich), dexamethasone (Sigma‒Aldrich), and ascorbic acid (HiMedia) and maintained in a humidified atmosphere at 37°C with 5% CO2. Conditioned medium was removed from the flasks when the cells reached 70 to 80% confluence after 3 to 5 days of culture. Dexamethasone was used in this culture as an anti-inflammatory and osteogenic differentiation medium agent, and ascorbic acid was used as an essential vitamin for the growth of the cells and its ability to suppress the generation of reactive oxygen species.33,34
MGC isolation
For isolation of MGCs, the follicular fluid taken from the initial stage was held in reserve at 37°C after oocyte separation and GCC segregation and contained a granulosa cell mixture. The test tubes containing follicular fluid were centrifuged at 800g for 10 minutes at 37°C. The supernatant of the fluid was discarded, and the residual cells at the bottom of the tube were resuspended in a small volume of PBS. This cellular suspension was at that moment layered very carefully (to elude mixing at this juncture) over the same quantity of 50% (v/v) PercollTM (Sigma‒Aldrich) (a density gradient separating solution). Then, this cell suspension with PercollTM solution was centrifuged at 500g for 20 minutes at 37°C. After this centrifugation, a couple of layers are formed. The interphase layer, which contained exclusively MGCs due to their density, was rigorously separated with a micropipette and washed numerous times with PBS. After suspending MGC clusters, 0.25% trypsin (Sigma‒Aldrich) was used for separation of cells from the clusters and held at 37°C for 10 minutes to remove them. After that, it was neutralized with DMEM/F-12 medium during the second centrifugation. Cell viability, counting and culture were performed in the same way as for the CGCs.
Cell culture and DHEAS exposure
After 5 days of cell culture, sufficient numbers of cells were obtained. The cells were then divided for DHEAS exposure. The groups were further named, with 4 groups for each category of PCOS and normal or non-PCOS GCs. In this study, the individual group was comprised by pooling 10 patient GCs for the complete study. Hence, the total number of patients involved in the study was 80. Prior to administering DHEAS, cells were plated in 6- and 24-well microplates at a density of 1 × 104 cells/mL (2 × 03 cells/cm2). Each group of cells was divided into 4 groups, in which the 2 cell types were introduced with 10−5 M DHEAS (Sigma‒Aldrich).35,36 Following DHEAS treatment, the same culturing conditions were maintained as in the preliminary culture, and these cells underwent exposure for 2 days. After that, these developed GCs were isolated 37 and maintained in storage at −80°C until use.
Protein extraction
The cells were denatured by 5 volumes of cool acetone before being precipitated at −20°C overnight and centrifuged at 4°C for 15 minutes. The extra fluid was discarded. The pellets were resuspended in a rehydration solution (7 M urea, 2 M thiourea, 4% [w/v]) after air-drying. Trimethylammonium compound 3-[(3-cholamidopropyl)] DL-dithiothreitol (DTT) 100 mM, 0.5% ampholytes (pH 4-7), 0.01% bromophenol blue (BPB), and 1% (v/v) protease inhibitor make up the CHAPS formula. The Bradford test was used for the detection of the protein. 38
In solution trypsin digestion
All reagents were prepared immediately prior to use. The water used in all components of the procedure was 18 Ω or Milli-Q water. Acetonitrile and methanol were HPLC grade. The trypsin used was of sequencing grade. Twenty micrograms of protein sample were placed in 50 µl of 100 mM TEAB in a sterile microcentrifuge tube. Protein was rehydrated by adding 0.25 μL of 1 M DTT to 50 μL of protein solution and incubated for half an hour at room temperature. Alkylation of protein was performed by adding 1 μL of 1 M iodoacetamide to 50 μL of protein solution and incubating in the dark for half an hour at room temperature. After rehydration and alkylation, the sample was incubated with trypsin (Promega, USA) at a final protease:protein ratio of 1:100 (w/w) for in-solution digestion at 37°C for 16 hours. The peptides were desalted with Pierce™ C18 Tips in a 100 µL bed according to the manufacturer’s directions, and peptides were eluted in 100 µL of 70% ACN in 0.1% FA. Eluates were dried using a speed-vac centrifuge.
Tandem mass spectrometry
Vacuum-dried peptides were suspended in 10 μL of 2% ACN in 0.1% FA and analyzed by a TripleTOF® 5600+ (ABsciex) mass spectrometer. The trap and analytical column with specifications Chrome-XP, 120 Å, 0.3 × 150 mm, and 3C18-CL-120, 3 μm, was attached to the mass analyzer. The spray nozzle and electro ionization source injected the peptide sample into a mass spectrometer. Analytical chromatography separated peptides in a continuous gradient of elution with 2 to 90% ACN at 5 µL/minutes for 35 minutes. The system mixes 2 reservoirs with solvent composition. Reservoirs A and B, with 2% ACN in 0.1% FA and 98% water, were used. The instrument was set to automatic MS and MS/MS mode for the acquisition. The original spectra were scanned from 350 to 1250 m/z. Product scanned at 150 to 1600 m/z ISVF = 5500; GS1 = 25; GS2 = 22; CUR = 30 were the ion source parameters.
MS data processing and analysis
The MASCOT program (version 2.7.0, Matrix Science) (https://www.maxquant.org) searched raw mass spectrometry files and assigned peptide sequences to the Swiss-Prot protein database’s Homo Sapiens taxonomy. The search parameters allowed up to 2 missed trypsin cleavages and 0.3 Da precursor and fragment ion mass tolerances. Carboxymethyl (C) was fixed, while deamidated (NQ) and oxidation (M) were variable. Peptide identification was allowed if the criterion exceeded 50% likelihood (P, .5).
Bioinformatics
The GeneAnalytics (http://geneanalytics.genecards.org) and MalaCards (https://www.malacards.org/)39,40 databases were utilized as bases for the network-based prediction technique, which took into account gene expression, gene ontology, distribution, and illness or disorder networks. We performed subcellular localization, protein‒protein interaction analysis, and functional enrichment analysis utilizing the free web tool String (https://string-db.org) 41 and other important tools, such as UniProt (https://www.uniprot.org/), 42 for the protein search. The PANTHER (Protein analysis through evolutionary relationships) Classification System (https://www.pantherdb.org/), a freely available data extractor, was devised to categorize proteins (and their genes) to make high-throughput research more accessible. The NCBI database (https://www.ncbi.nlm.nih.gov/), 43 Swiss-Prot (protein sequence database), RefSeq (https://www.ncbi.nlm.nih.gov/refseq/), GenBank (https://www.ncbi.nlm.nih.gov/genbank/), 44 and other open data arrays were employed for protein functional elucidations, ontology studies, and protein disease relations.45 -47
Results
Study group sociodemographic characteristics
A total of 160 patients were screened, of which 115 patients met the inclusion and exclusion criteria. For the examinations and comparison after DHEAS exposure, 80 patients were finally selected. Now equal numbers (n = 40) for PCOS and non-PCOS patients GCs reserved, the mean age of the population was 27.8 years with a minimum age of 23 years and the maximum age was 42 years.
Expression of proteins in GCs
In our protein samples of the control population and women with PCOS analyzed by LC‒MS/MS, a total of 617 proteins were identified in CGCs and MGCs. The CGCs proteins accounted for 276, whereas 341 proteins were found in MGCs (Figure 1). This shows that MGCs are more expressive than CGCs.
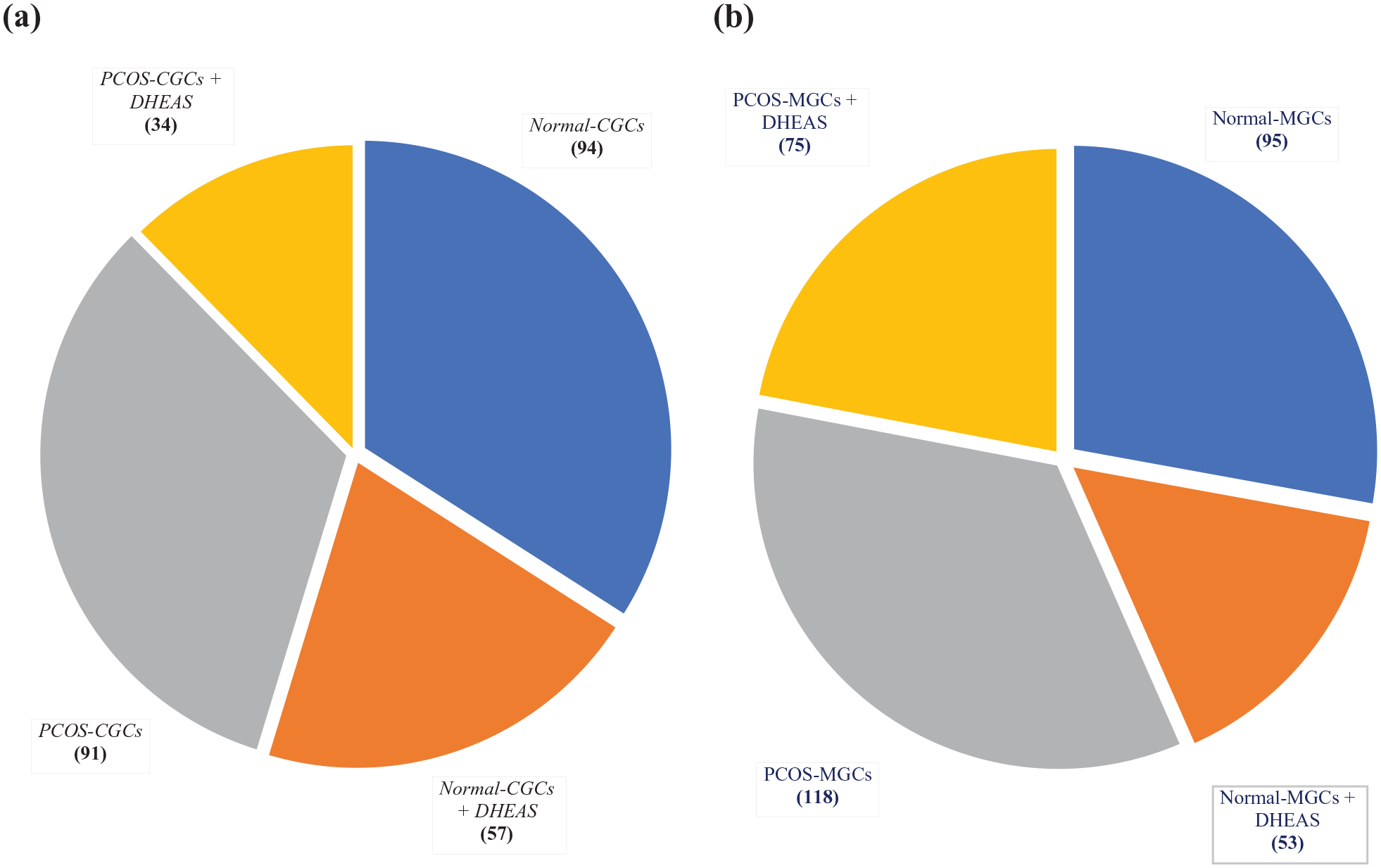
Differential protein expression in PCOS and non-PCOS patient GCs with and without exposure to DHEAS: (a) proteins expressed by CGCs and (b) proteins expressed by MGCs.
Altered expression of proteins in CGCs of normal individuals and women with PCOS
As demonstrated in Figure 1, a total of 276 proteins were considered for the bioinformatics studies from CGCs, of which 91 were expressed in PCOS patient’s CGCs (PCOS-CGCs) and only 34 proteins were obtained after exposure to DHEAS in PCOS patient’s CGCs (PCOS-CGCs + DHEAS). This shows suppression of protein expression by DHEAS in PCOS CGCs. Similar effects were observed in non-PCOS CGCs, where a total of 94 proteins were reduced to 57 proteins after DHEAS exposure, in which 40 proteins were differentially expressed in “Normal-CGCs + DHEAS,” including ACTBL, ADCY1, AL1A1, AL8A1, ANKE1, APOA1, APOB, ASML, CCD81, CDC37, CLIP3, COMP, DHE3, EIF2D, HBA, HS71A, HS90A, IL7RA, ISM2, KLK15, MTEF4, MYH7B, MYO5B, NALP5, NB5R2, NCF1B, NOTO, NWD1, PCDG5, PEDF, PLCB1, PLVAP, RAP2A, RD3L, SELO, SH321, TET5B, THUM2, TRAP1, and TRFE.
In comparison to untreated PCOS patient CGCs, the 35 proteins were included exclusively in DHEAS-treated PCOS patient cells “PCOS-CGCs + DHEAS” (DPC1, A2MG, AHNK, ALBU, ANKY2, CASP2, CC192, CP4FN, DYH8, G3P, G6PD, GXLT1, INP5E, KCNKI, LNP, M3K5, MAGI1, MAGI2, MAGT1, MEOX1, MST1L, NAMPT, NC301, NEO1, NSRP1, O52L2, PFD6, PLVAP, ROCK2, SC6A9, SHRM4, SP110, TIE1, TSSK4, and ZBT32). Showing the differential expression of CGCs in PCOS and non-PCOS.
Altered expression of proteins in MGCs of normal individuals and women with PCOS
We identified approximately 500 different proteins (Figure 1b) in all MGCs, whereas a total of 341 proteins were further considered for bioinformatics studies, of which 118 were expressed in PCOS patient MGCs (PCOS-MGCs) and 75 were obtained after exposure to DHEAS in PCOS patient MGCs (PCOS-MGCs + DHEAS). A total of 95 proteins were found in control samples, and upon DHEAS treatment, 53 proteins were expressed, of which 36 proteins were differentially expressed in “Normal-MGCs + DHEAS” (ACON, AL1A1, AL8A1, AP4E1, ASPP1, BRD8, CFA57, CIA2A, CO3, COMP, DHE3, ERIC3, ESPNL, FGD4, FIBA, HS71A, IF122, ISM2, KAD2, MAGT1, MGAL, MTEF4, NDKA, NWD1, PLVAP, RAP2A, SIA8D, SORL, TAOK3, TDR15, TET5B, THUM2, TRFL, TSH1, TTHY, WDR43, and WLS).
In comparison to untreated PCOS patient MGCs, the 61 proteins included exclusively in DHEAS-treated PCOS patient cells from 118 to 75 “PCOS-MGCs + DHEAS” (ALDH2, C19L2, CA2D1, CAC1A, CAPSL, CASP2, CE120, CJ090, CO6A5, CP4F2, CPLN1, CSMD1, CTSRD, CUL3, DEP1A, DRD3, DTX3, DYH17, DYH5, ELOB, EMIL2, FABD, FUZZY, G6PD, GCP2, GNAL, GPC5, GVIN1, INP5E, KCNKI, KS6C1, M3K7, MAGT1, MTREX, MYOM2, NAA16, NAMPT, NEO1, NSRP1, OPALI, PCX2, PMA6E, PO4F1, PSB6, PTCD1, R3HCL, S22AI, SHLD2, SHRM4, SMAG2, SNP25, SNX29, SYYC, TIE1, TRAK2, TRIPC, TYK2). This demonstrates that DHEAS exposure suppresses the expression of MGCs similar to CGCs.
Venn analysis of CGCs
Comparison of all these 4 cells types; (Figure 2) Normal-CGCs, Normal-CGCs + DHEAS, PCOS-CGCs, and PCOS-CGCs + DHEAS, the Venn results shows 49 proteins included exclusively in “Normal-CGCs”: (ACOT2, ALAT2, ARVC, ASIC2, ASPD, CAND1, CLU, CNTRB, COQA1, CS081, CSMD1, CTSRD, CTU2, DHRS2, DHX35, DIRA1, DRC10, ELOB, ERIC3, FZD3, GRP75, H3C, HAND1, IQGA3, KAT6A, LDHA, LIPB1, LOXL4, MADD, MPCP, MTREX, NOP53, OPALI, OR1B1, PLCD3, PLXD1, PRAX, PTEN, SIA8D, SMC3, TELO2, TITIN, TMM62, TPRA1, TVAZ2, UEVLD, VILI, WDR48, and WRN) and 53 proteins included exclusively in “PCOS-CGCs” (A1BG, ABCA3, ACOXL, ARAP1, ARL6, AT10A, ATS7, BIP, CAN1, CC191, CCD22, CELR1, CNTN6, COL12, CTBP2, DCOR, DNJB1, DRD3, DSCL1, EXOSX, GSTA1, H2B1A, H31T, HSP76, IGJ, ITPR3, IZUM3, K1109, KAD9, KCNJ5, MDEAS, MVP, NEST, PARP1, PC4L1, PHF2, PKCB1, PKD1, PSB6, RP3A, RPA2, SCN1A, SCRB2, SIG10, SLIK4, SPAT7, SYUB, TBA1A, THBG, VW5B2, ZEP2, ZFP3, and ZFR2). This shows an exclusive expression of cells under 2 different pathological conditions.

Venn analysis of PCOS and non-PCOS patient GC expression with and without exposure to DHEAS (a) CGCs and (b) MGCs.
After treatment with DHEAS in the CGCs, we found 36 proteins that were included exclusively in “Normal-CGCs + DHEAS” (ACTBL, ADCY1, AL1A1, AL8A1, ANKE1, APOA1, APOB, ASML, CCD81, CLIP3, COMP, DHE3, EIF2D, HS71A, HS90A, IL7RA, ISM2, KLK15, MTEF4, MYH7B, MYO5B, NALP5, NB5R2, NCF1B, NWD1, PCDG5, PEDF, PLCB1, RAP2A, RD3L, SELO, SH321, TET5B, THUM2, TRAP1, and TRFE) and 21 proteins that were found exclusively in “PCOS-CGCs + DHEAS” (CASP2, CC192, CP4FN, DYH8, G3P, G6PD, INP5E, KCNKI, M3K5, MAGT1, MEOX1, MST1L, NC301, NEO1, NSRP1, O52L2, PFD6, ROCK2, SC6A9). Only 1 protein (PLVAP) was found in both the “normal CGCs + DHEAS” and “PCOS CGCs + DHEAS” groups.
Five common proteins in “Normal-CGCs” and “Normal-CGCs + DHEAS” (FES, SORL, TRFL, WDR43, and WLS) and 2 common proteins in “Normal-CGCs,” “Normal-CGCs + DHEAS,” “PCOS-CGCs,” and “PCOS-CGCs + DHEAS” (A2MG and ALBU) and 2 common proteins in “Normal-CGCs,” “Normal-CGCs + DHEAS,” and “PCOS-CGCs + DHEAS” (TIE1 and TSSK4). These results depict the overlap in the process of cellular expression under stressed conditions.
Venn analysis of MGCs
When we compared all 4 cell types, Normal-MGCs, Normal-MGCs + DHEAS, PCOS-MGCs, and PCOS-MGCs + DHEAS, the Venn analysis shows 51 proteins included exclusively in “PCOS-MGCs + DHEAS” (ALDH2, C19L2, CA2D1, CAC1A, CAPSL, CASP2, CE120, CJ090, CO6A5, CP4F2, CPLN1, CSMD1, CUL3, DEP1A, DRD3, DTX3, DYH17, DYH5, EMIL2, FABD, FUZZY, G6PD, GCP2, GNAL, GPC5, GVIN1, INP5E, KCNKI, KS6C1, M3K7, MTREX, MYOM2, NAA16, NEO1, NSRP1, PCX2, PMA6E, PO4F1, PSB6, PTCD1, R3HCL, S22AI, SHLD2, SHRM4, SMAG2, SNP25, SNX29, SYYC, TRAK2, TYK2, and VGFR3).
The 29 common proteins in “Normal-MGCs” and “PCOS-MGCs + DHEAS” were A2AP, ABCA3, ACTB, ARHGG, ASB6, BIP, CCD22, CELR1, COL12, ENPL, F126B, FES, GALT1, GXLT1, H2B1C, H3Y1, H4, HBA, HPRT, MDEAS, MELT, NCKPL, NCOA5, PDIA3, RDX3, RIF1, RP25L, TPRA1, and TSYL2. When we compared the PCOS cells treated with DHEAS, 5 common proteins were found in Normal-MGCs + DHEAS “PCOS-MGCs C” and “PCOS-MGCs + DHEAS” (A1BG, DHI2, O5AP2, TBA1A, and VP13D). In the control sample comparison, 3 common proteins were observed in “Normal-MGCs” and “Normal-MGCs + DHEAS” (AHNK, HS90A, and TRAP1).
Only 6 common proteins were found in “Normal-MGCs,” “normal-MGCs + DHEAS,” “PCOS-MGCs,” and “PCOS-MGCs + DHEAS” (A2MG, ALBU, FETUA, ITIH2, SGO2, and SPT6H), a single protein, MAGT1_HUMAN, was found in “normal-MGCs + DHEAS” and “PCOS-MGCs + DHEAS,” and only 1 protein, CO3, was common in “normal-MGCs + DHEAS,” “PCOS-MGCs,” and “normal-MGCs + DHEAS.” A total of 32 proteins were exclusively included in “Normal-MGCs + DHEAS” (ACON, AL1A1, AL8A1, AP4E1, ASPP1, BRD8, CFA57, CIA2A, COMP, DHE3, ERIC3, ESPNL, FGD4, HS71A, IF122, ISM2, KAD2, MGAL, MTEF4, NDKA, NWD1, PLVAP, RAP2A, SIA8D, SORL, TAOK3, TDR15, TET5B, THUM2, TSH1, TTHY, and WLS). These results are indicative of common signaling processes in MGCs under 2 different conditions.
Gene ontology study
Molecular functions of proteins expressed by control samples in the presence and absence of DHEAS treatment
The maximum number of proteins belongs to the binding, 42 proteins of CGCs the Normal-CGCs, 24 proteins of Normal-CGCs + DHEAS, 4 proteins of PCOS-CGCs, 7 proteins of PCOS-CGCs + DHEAS and 44 proteins of MGCs the PCOS-MGCs, 18 proteins of PCOS-MGCs + DHEAS, 29 proteins of Normal-MGCs, 17 proteins of Normal-MGCs + DHEAS. The second largest number of proteins of both categories of GCs was involved in catalytic activity, with 37 proteins of MGCs of PCOS-MGCs, 18 proteins of PCOS-MGCs + DHEAS, 30 proteins of Normal-MGCs, and 18 proteins of Normal-MGCs + DHEAS.
In CGCs, 37 proteins of normal CGCs, 19 proteins of normal CGCs + DHEAS, 38 proteins of PCOS-CGCs, and 10 proteins of PCOS-CGCs + DHEAS had the same activity. The other functions include transporter activity, translation regulator activity, transcription regulator activity, molecular function regulator, cytoskeletal motor activity, ATP-dependent activity, structural molecule activity, adaptor activity, and molecular transducer activity. Decreased activity was observed in most of the proteins after treatment with DHEAS, and a significant decrease in binding and catalytic activity was observed after treatment (Figure 3a and b).

The Panther GO-Slim showing the Molecular Function classifies the frequency of genes expressed by (a) CGCs and (b) MGCs.
Biological process of proteins expressed by control samples in the presence and absence of DHEAS treatment
The gene ontology results of the proteins show a maximum number (60-90 proteins of the individual) of proteins belonging to biological regulation and cellular and metabolic processes. The normal GCs significantly fell in the number of cellular biological processes, as the results showed 90 proteins of normal CGCs, 60 proteins of normal MGCs, 33 proteins of normal CGCs + DHEAS and 30 proteins of normal MGCs + DHEAS. In PCOS sample cases, 51 proteins of PCOS-CGCs, 24 proteins of PCOS-CGCs + DHEAS, 72 proteins of PCOS-MGCs, and 45 proteins of PCOS-MGCs + DHEAS marked declines were observed. Second, most of the proteins were involved in localization and stimulus response in the biological process domain. The remaining proteins were found to be involved in biological adhesion, interspecies interaction between organisms, response to stimulus, locomotion, signaling, and developmental, immune system, multicellular organismal, and reproductive processes. As shown in Figure 4a and b, there was a significant decrease in biological processes such as stimulus response, biological adhesion and signaling observed in DHEAS-treated GC proteins.

Panther GO-Slim showing the Biological process classifies the frequency of genes expressed by (a) CGCs and (b) MGCs.
Cellular components of proteins expressed by control samples in the presence and absence of DHEAS treatment
The majority of the proteins belonged to the cellular anatomical entity (Figure 5a and b). 58 proteins of PCOS-CGCs, 18 proteins of PCOS-CGCs + DHEAS and 70 proteins of PCOS-MGCs, 40 proteins of PCOS-MGCs + DHEAS. The protein-containing complex class is attained by the remaining proteins. There was a significant reduction observed after the DHEAS treatment of GCs.

The panther GO-Slim showing the cellular components of GCs: (a) CGCs and (b) MGCs.
Classes of proteins expressed by control samples in the presence and absence of DHEAS treatment
The GO analysis showed the involvement of GC proteins in various categories (Figure 6), and the majority of proteins belonged to the metabolite interconversion enzyme and protein-modifying enzyme classes. The metabolite interconversion enzyme class included 17 proteins of PCOS-MGCs, 9 of PCOS-MGCs + DHEAS, 7 proteins of PCOS-CGCs, and 6 of PCOS-CGCs + DHEAS. Other important classes include cytoskeletal protein, protein-binding activity modulator, RNA metabolism and transmembrane signal receptor and transporter.

Panther GO-Slim results showing the Protein Class categorizes the genes with and without DHEAS exposure in GCs.
This analysis shows that the higher number of proteins from the normal control samples contained significantly more proteins in all categories, while there were fewer proteins differentially categorized in all classes. Calcium-binding protein and defense/immunity protein were found only in PCOS samples of MGCs and were lower in CGCs. Protein-modifying enzymes were highly present in both DHEAS-treated PCOS CGCs and MGCs, while the Chaperone, Chromatin-binding protein, immunity protein, DNA metabolism protein, and Extracellular matrix protein classes were significantly lesser or absent in both PCOS type samples. The calcium-binding protein, immunity protein, DNA metabolism protein, RNA metabolism protein, scaffold protein, structural protein and translational protein classes were absent in CGCs after DHEAS treatment. Therefore, these results show the generous differences between the protein classes of normal and PCOS patient samples and significant alterations observed in DHEAS-treated GCs.
Panther pathway involved in proteins expressed by control samples in the presence and absence of DHEAS treatment
Only DHEAS-treated PCOS-MGCs and some PCOS MGC proteins exhibited the following signaling pathways: alpha adrenergic receptor signaling, beta adrenergic receptor signaling, endogenous cannabinoid, endothelin, GABA-B receptor II, histamine H2 receptor-mediated signaling, interleukin, TGF-beta, thyrotropin-releasing hormone receptor, toll receptor, VEGF, p38 MAPK, PGDF, enkephalin release, and angiogenesis. These pathways were completely absent in the control sample proteins of MGCs (Figure 7).

The GO results representing the diverse biological pathways expressed by the CGCs and MGCs proteins of both control and PCOS categorizes. The abovementioned data their serial number represents below mentioned categories of the proteins, respectively.
In CGC protein samples, the normal and DHEAS-treated groups belonged to 2-arachidonoylglycerol biosynthesis, 5HT receptor-mediated signaling, 5-hydroxytryptamine degradation, Alzheimer’s disease-presenilin, adrenaline and noradrenaline biosynthesis, alpha adrenergic receptor signaling, adenine and hypoxanthine salvage pathway, angiogenesis, oxytocin receptor-mediated signaling, metabotropic and ionotropic glutamate receptor, opioid proenkephalin, and proopiomelanocortin, TGF-beta signaling, thyrotropin-releasing hormone receptor signaling, toll receptor signaling, VEGF signaling, PI3 kinase, and angiotensin II-stimulated signaling, while these pathways were not present in PCOS samples of the CGCs. Pathways such as inflammation mediated by cytokine and chemokine signaling, the beta adrenergic receptor signaling pathway, the heterotrimeric G-protein signaling pathway, the metabotropic glutamate receptor pathway, the muscarinic and nicotinic pharmacodynamics pathway, and thyrotropin-releasing hormone receptor signaling were highly enriched in normal CGCs compared to PCOS CGCs.
In MGC samples, alpha adrenergic receptor, angiogenesis, beta adrenergic receptor, dopamine receptor mediated, endogenous cannabinoid, endothelin, heterotrimeric G-protein signaling G (i,s,q,o)α mediated, GABA-B receptor II, gonadotropin-releasing hormone receptor, histamine H2 receptor mediated, enkephalin release, insulin/IGF protein & MAP kinase cascade, interleukin, muscarinic acetylcholine receptor 2 and 4, nicotine pharmacodynamics, oxytocin receptor mediated, p38 MAPK, PDGF, proline biosynthesis, TGF-beta signaling, thyrotropin-releasing hormone receptor, toll receptor, VEGF, and so on, signaling pathways present in PCOS MGCs and DHEAS-treated MGCs. The protein pathway analysis of the proteins shows significant differences between the normal and PCOS samples, and the DHEAS-treated samples of both represent the differential pathways (as described in Figure 7).
String results
String results of CGCs
The normal CCC string shows that 4 proteins (ALB, ACTB, HIST1H4F, and POLR2A) were core nodes linked with subnetworks, including AFP, ITIH1, ASHG, ITIH2, GC, LDHA, PTEN, and SKIV2L2, and the others included MAGI2, HBB, WDR43, AFP, A2M, H3F3C, SERPINF2, SUPT6H, and HIST1H2AA. The DHEAS-treated normal CGCs involve the major networks of APOA1, APOB, TF, ALB, AFP, GC, ASHG, and C3; secondarily, these networks are connected to networks including A2M, LTF, HBB, HBA2, and HSP90AA1. PCOS-CGCs proteins show the core networks of ALB, AHSG, ACTB, HIST1H2BA, HIST1H4F, A2M, and ITH2, and these networks are further connected with subunits of HBA2, HSPA5, A1BG, AFP, PILR2A, SERPINF2, HBB, GC, PARP1, DNAJB1, SERPINA7, C3, etc. DHEAS-treated PCOS-CGCs consist of only 1 core protein, GAPDH, which is linked to ALB, G6PD, and A2M as small secondary networks (Figure 8a–d).

STRING diagram representing the protein interaction pathway derived from: (a) normal-CGCs, (b) normal-CGCs + DHEAS, (c) PCOS-CGCs, and (d) PCOS-CGCs + DHEAS.
String results of MGCs
The normal-MGC proteins involved the major networks of HIST1H4F, HSP90B1, HSP90AA1, and HSPA5. These core networks further connected with the networks including ACTB, P4HB, PDIA6, ALB, PRDX3, ATP5A1, PDIA3, SERPINF2, LDHA, GC, AFP, and so on. DHEAS-treated normal MGCs contain TTR, C3, AFP, AHSG, FGA, and GC as core networks linked with HBB, ACO3, ITIH2, LTF, A2M, and HSP90AA1.
The PCOS-MGCs proteins involved the major networks of ACTB, SERPINF2, HIST1H4F, AFP, GC, RBP4, and LTF, which are further linked to secondary networks of HIST1H2BC, ASHG, ITIH2, A1BG, C3, HBA2, HSPA5, PDIA3, PRDX3, HBB proteins, and so on. The DHEAS-treated PCOS-MPC proteins on the string show 2 small core networks, AHSG and ALB, in addition to 1 smaller network, SUPT6H. AHSG and ALB networks were linked to 4 to 5 subunit single protein networks, including ITIH2, A2M, C3, A1BG, and CACNA1A (Figure 9a–d).

STRING diagram representing the protein interaction pathway derived from: (a) normal-MGCs, (b) normal-MGCs + DHEAS, (c) PCOS-MGCs, and (d) PCOS-MGCs + DHEAS.
Disease gene relationships
PCOS is a highly heterogeneous and complicated condition. The genetic basis for PCOS varies between and within families; however, it is linked to a common mechanism. Due to the complexity and variability of the human genome, single genes or even genes that are associated with the GCs of both categories of CGCs and MGCs are summarized (Table 1 and Figure 10) using the GeneCards Human Gene Database.
Disease gene relationships.


Cycle matrix representing the protein interaction pathway derived from: (a) normal-MGCs, (b) normal-MGCs + DHEAS, (c) PCOS-MGCs, and (d) PCOS-MGCs + DHEAS.
Discussion
The results of our study show that the mural and cumulus GCs of normal and PCOS patients express proteins differently in the presence and absence of DHEAS. These proteins are noted to be central to different molecular and biological functions of the body. While hyperandrogenism is a fundamental distinguishing characteristic of PCOS, the particular mechanism(s) through which androgens mediate the formation of the PCOS phenotype by normal CGCs, as well as the effect in PCOS CGCs, remains unexplained. The current study reveals data to support the assumption that androgens have impacts, most likely through the cellular and molecular processes engaged following initial exposure, as well as the mechanisms and pathways that evolve. Our findings provide empirical support that the variation in the protein expression and processes of PCOS cells may be attributable to differences in the underlying steroid signal transduction pathways implicated. The objective of the study is achieved successfully by the use of various open source databases and advanced research equipment. In this study, exposure of CGCs obtained from PCOS patients to DHEAS reduced the expression of a number of proteins, which could be due to downregulation of the WNT signaling pathway, as mentioned in recently published literature, irrespective of the source of cells and pathological conditions. 48
Adiponectin protein expression in MGCs was found to be involved in periovulatory remodeling of vascular endothelial growth factor genes and ovarian follicles. 49 The MAPK signaling pathway, which is 1 of the 2 best-characterized insulin signaling pathways, was also observed in our investigation. It mediates mitogenic effects and is consequently responsible for regulating various cellular processes, including differentiation, proliferation, response to stress, cellular survivability, and death. In fact, this differentially produced protein concentrated in this pathway may also target the ovary directly, resulting in infertility in PCOS patients. 50 Upon exposure of CGCs and MGCs, 2 important proteins, DENND1A and THADA, were expressed to play a central role in androgen biosynthesis, and metabolic regulation has been observed to be activated in DHEAS-exposed cells. 51 It was also found that the differentially expressed proteins found in our study are related to molecular functions of lipid binding, complement binding, cytokine and chemokine activity. 52 The results of our study support earlier studies and propose that these mechanisms are involved in obesity and that the inflammatory mechanism inherent to PCOS may be mediated by GCs under the influence of DHEAS.
Other pathways show that there are multiple pathways of PCOS that are activated when MGCs are exposed to DHEAS, such as alpha adrenergic receptor signaling, 53 beta adrenergic receptor signaling, 54 and endogenous cannabinoids, 55 along with the involvement of endothelin, GABA-B receptor II, interleukin, TGF-beta, thyrotropin-releasing hormone receptor, Toll receptor, VEGF, p38 MAPK, PGDF, and enkephalin release. Treatment of CGCs with DHEAS shows altered 2-arachidonoylglycerol biosynthesis, 56 adrenaline and noradrenaline biosynthesis, alpha adrenergic receptor signaling, 34 TGF-beta signaling, 57 Toll receptor signaling, 58 VEGF signaling, 59 and PI3 kinase. 60 Based on these results and previous studies, we propose that MGCs and CGCs behave differently in the presence of DHEAS, possibly activating the pathogenesis of PCOS.
String results show that there are multiple generic conditions central to the pathogenesis of PCOS. DHEAS-treated PCOS-CGCs consist of only 1 core protein, GAPDH, which is linked to ALB, G6PD and A2M as small secondary networks. The PCOS-MGCs proteins involved the major networks of ACTB, SERPINF2, HIST1H4F, AFP, GC, RBP4, and LTF, which are further linked to secondary networks of HIST1H2BC, ASHG, ITIH2, A1BG, C3, HBA2, HSPA5, PDIA3, PRDX3, HBB proteins, and so on. The DHEAS-treated PCOS-MPC proteins on the string show 2 small core networks, AHSG and ALB, in addition to 1 smaller network, SUPT6H. AHSG and ALB networks were linked to 4 to 5 subunit single protein networks, including ITIH2, A2M, C3, A1BG, and CACNA1A. The ACTB protein is important for cell signaling, 61 including the regulation of tight junction networks and control of the actin cytoskeleton.62,63 The expression of the chaperone protein HSP90B1 negatively impacts cell growth and survival, apoptotic processes and cell cycle development. HSP90B1 functions with many intracellular binding sites and transcription factors and is an important protein that also plays a role in protecting other proteins from protease degradation, including progesterone, glucocorticoid, and AKT receptors, and is in charge of the stability and development of such proteins after activation. 64 These results indicate that ALB, G6PD, ACTB, AHSG, and SUPT6H expressed by MGCs and CGCs are primarily involved in the occurrence of PCOS.
The majority of phase II enzymes implicated in cellular detoxification are glutathione S-transferases (GSTs), in which the GSTA1 gene is significant for pregnancy-related pathological conditions and its association with maternal hypertensive disorders. 65 It has been suggested that GSTA's function may be related to oxygen stress caused by ROS (reactive oxygen species) produced during steroidogenesis since the highest level of GSTA overexpression was found in steroidogenically active cells. 66 Pkd1 is an additional important gene of interest that encodes polycystin1 (PC1), a large membrane-spanning glycoprotein that is crucial for tissue and organ structural integrity. 67 These proteins regulate a wide range of intricate biological processes, including cell growth and division, membrane trafficking, cellular secretion, defense, signal transmission, and angiogenesis. The hypoxic glycolytic metabolism regulation mechanism of PKD1 may be related to polycystic ovarian morphology. HIF-1 is also implicated as a significant part of several cancerous cell biobehaviors through HIF-1 and glycolytic enzymes, and it controls hypoxia metabolism in cancer cells. 68 The expression of these GSTs, PC1, PKD1, and HIF-1 by GCs under DHEAS exposure shows that there is a lack of control of the ROS production inhibitory effect, which may augment PCOS pathogenesis.
Studies have revealed that nicotinamide phosphoribosyltransferase (NAMPT), an adipokine, can influence the growth and steroidogenesis of ovarian follicles and act to increase insulin sensitivity. 69 Insulin and IGF-1 work together with FSH and LH in the ovary to increase estrogen synthesis by GCs and testosterone production by thecal stromal cells. According to some research, PCOS is linked with insulin resistance in the ovary. Hyperinsulinemia and peripheral insulin intolerance are frequently linked to PCOS. It is controversial whether the ovary is resistant to insulin. An earlier study showed that, in contrast to granulosa-lutein cells from women without PCOS, the effect of insulin on glucose metabolism is considerably decreased in granulosa-lutein cells isolated from the ovaries of women with typical PCOS. As an outcome, it appears that the ovary exhibits specific insulin resistance in PCOS. The quality of oocytes is directly impacted negatively by insulin resistance as well. In addition to human GCs, human egg cells also exhibit NAMPT. 70 These results indicate the role of GCs in insulin resistance, hyperinsulinemia, and hyperandrogenism, which are key manifestations of PCOS, through the NAMPT signaling process. The proper functioning of metabolic balance and immunological responses in PCOS largely depends on the critical participation of immune cells and chemicals involved in immune control, and here the immunity proteins also found to higher in PCOS MGCs then CGCs. 71 The reason is to elevation of immunity protein shows the pathological state and is the indication of their activation. The immune system typically identifies pathogenic conditions by recognizing unique structural traits that set them apart from normal states. Earlier reports also support our results, irregular signaling pathways in PCOS conditions are being investigated using differential expression profiling of GCs proteins. 72 For that reason, this reveals intricate disruptions in various biological functions, including cell communication, immune responses, insulin signaling, oxidative processes, fatty acid metabolism, and inflammation.
An important strength of the present study is that to the best of our knowledge, we successfully distinguish the proteins and their relation to various molecular and biological pathways with DHEAS exposure in GCs for the first time. These pathways are supported by the existing data and generous new aspects of proteomics for the pathogenic and pathophysiological paths. However, the current study has some limitations. First, the ethnicities were limited to those from Uttarakhand, a state of India, and our sample size was somewhat modest due to financing limitations. The availability of patients meeting inclusion and exclusion criteria at the study center, and the length of the study, that is, 2 years, we chose 80 (8 groups × 10 patients) as the sample size. For the implementation of statistical analysis, we required samples at least in triplicate, that is, 240 patients who met the inclusion and exclusion criteria, which was not possible due to a lack of number patients at the study center during the study period. Therefore, there is a need to extend further study to understand the effects of DHEAS in patients who have not undergone the ovarian hyperstimulation protocol and are carried at different ethnicity levels. It is also recommended to verify the proteins expressed by using higher levels of molecular biology-based technologies.
Conclusions
The present study was conducted using CGCs and MGCS of women who underwent the regulated hyperovarian stimulation process. The results of this study indicate that there is a strong relationship between proteins expressed by CGCs and MGCs under the influence of DHEAS. Multiple pathways were identified through the Panther pathway, linking it to the PCOS pathway. These results confirm that there is an alteration in protein expression by these cells under the influence of DHEAS. A better understanding of the different characters, pathways, and dispositions of genes and proteins found in control and PCOS populations, as well as the appearance and disappearance of those genes and proteins after DHEAS exposure to GCs, will assist in developing new propositions and paths towards the establishment of new targets and markers. However, further studies may be recommended in women who are not exposed to ovarian stimulation.
Footnotes
Acknowledgements
We acknowledge Mr. Achal Tiwari, Ms. Laxmi Arya, Mr. Nitin Kataria, and all other technical and nontechnical staff of Indira IVF Fertility Centre, Dehradun for their help and support throughout the study. We also thank Dr. Neha Sharma, Application Scientist, ATPC, RCB, Faridabad (Haryna)-India.
List of abbreviations
BMI: Body mass index
CGCs: Cumulus Granulosa cells
DHEAS: Dehydroepiandrosterone
DHEAS: Dehydroepiandrosterone sulfate
GO: Gene Ontology
ICSI: Intracytoplasmic sperm injection
IVF: In vitro fertilization
MGCs: Mural Granulosa cells
Normal-MGCs + DHEAS: Non-PCOS patients Mural Granulosa cells exposed to DHEAS
Normal-MGCs: Untreated Non-PCOS patients Mural Granulosa cells
Normal-CGCs + DHEAS: Non-PCOS cumulus granulosa cells exposed to DHEAS
Normal-CGCs: Untreated non-PCOS cumulus granulosa cells
OMIM: Online Mendelian Inheritance in Man (OMIM®)
PBS: Phosphate buffer saline
PCOS: Polycystic ovary syndrome
PCOS-MGCs + DHEAS: PCOS patients’ Mural Granulosa cells exposed to DHEAS
PCOS-MGCs: Untreated PCOS patients Mural Granulosa cells
PCOS-CGCs + DHEAS: cumulus granulosa cells exposed to DHEAS in PCOS patients
PCOS-CGCs: cumulus granulosa cells from untreated PCOS patients
