Abstract
Background:
Diabetes mellitus-induced hyperglycemia increases oxidative stress and inflammatory cytokine production, which play a significant role in the damage and apoptosis of pancreatic β cells. Therefore, the administration of medications that can reduce oxidative stress and inflammation plays an important role in diabetes treatment.
Objective:
To probe the Clinacanthus nutans leaf extract effect on oxidative stress and inflammatory markers and the Langerhans islet area in diabetic rat models.
Design:
An experimental laboratory in the animal model.
Methods:
Twenty-five diabetic rat models were randomly assigned into 5 clusters. Clusters 1, 2, and 3 were administered with C. nutans leaf extract in aqueous suspension with vehicle 1% Na-CMC at 75 mg/kg body weight (BW), 150 mg/kg BW, and 300 mg/kg BW, respectively. Cluster 4 was diabetic control rats administered with metformin at a 21 mg/rat dose. Cluster 5 was a control diabetic rat only administered with 1% Na-CMC suspension. Treatment was administered orally for 14 days. On the 15th day, the rats were sacrificed to obtain blood samples and pancreatic tissues. Serum interleukin (IL)-6, malondialdehyde (MDA), and tumor necrosis factor (TNF-α) were measured using the enzyme-linked immunosorbent assay (ELISA) method. Histopathological examination was performed by counting the Langerhans islet areas.
Results:
The average IL-6, MDA, and TNF-α levels declined in the cluster receiving C. nutans extract and were significantly different from the untreated cluster (P < .05). Histopathological examination revealed a significant upsurge in the Langerhans islets area in diabetic rats receiving C. nutans extract at doses of 75 and 150 mg/kg (P < .05).
Conclusion:
C. nutans leaf extract reduced the serum MDA, TNF-α, and IL-6 levels, and increased the Langerhans islets area in a diabetic rat model.
Introduction
Diabetes mellitus (DM) has emerged as a global health issue in recent decades due to its increasing prevalence. The number of patients with DM is increasing from 463 million (8.3%) in 2019 to 537 (10.5%) in 2021, and if no intervention is implemented, 783 million (12.2%) people are estimated to have DM by 2045. Indonesia ranks fifth (9.5 million) among countries with the highest number of people with DM, and this number has increased compared to 2019 at seventh (10.7 million).1,2
The primary problems associated with DM are high blood glucose levels due to a shortcoming in insulin production from pancreatic β cells and insulin resistance activity in peripheral tissues.3,4 Persistent hyperglycemia increases the formation of free radicals that cause oxidative stress.5,6 Several studies reported that the oxidant-antioxidant status of the diabetic rat model can be observed by the increased malondialdehyde (MDA) levels.7 -9 Oxidative stress induces low-grade chronic inflammation characterized by an increase in various cytokines, such as interleukin-6 (IL-6), interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β).10,11 It causes β-cell dysfunction and insulin production and secretion defects and exacerbates hyperglycemia. 12 The Langerhans islets are a cluster of pancreatic cells consisting of a few to several thousands of endocrine cells, and concurrently they assemble up to 2% of the total mass of the pancreas. The cell types of the islet are as follows: α (30%), β (60%), δ (10%), ε, and pancreatic polypeptide cells. Reducing the number of β cells will decrease the area of the Langerhans islets. 13 Oxidative stress is closely related to proinflammatory cytokines on impaired function and β-cell death. Therefore, intervention in the inflammatory pathway is expected to be a therapeutic approach to treat DM. Herbal plants with antioxidant and anti-inflammatory properties are currently developed for this purpose. 14
Clinacanthus nutans Lindau, from the Acanthaceae family, is commonly cultivated in tropical Asian countries, such as Indonesia, China, Malaysia, and Thailand. 15 This plant is traditionally mixed with fresh juices and vegetables or with concoctions and is used to treat skin problems, insect bites, cancer, inflammatory disorders, and DM. 16 The examination of toxicity revealed that C. nutans extracts did not imply any toxicity; thus, they can be used to produce potent medicinal medicine for various diseases.17,18 The plant contains many phytochemicals, including flavonoids, glycosides, pheophytin, flavones, sulfur-containing glycosides, triterpenoids, phytosterol, and alkaloids. 19 Previous studies found that C. nutans polar extract has anti-inflammatory, antiviral, anticancer, immunomodulatory, and antidiabetic activity; its semipolar extract has wound-healing, anticancer, and antiviral properties; and its nonpolar extract has anticancer properties. 20 Previous studies reported that C. nutans can be used as an antidiabetic drug. The C. nutans extract has been found to reduce fasting blood glucose levels in diabetic rats21,22; regardless, the proof of its effectiveness in reducing diabetic complications is restricted. Some studies also demonstrated that the extract can increase the serum insulin grade and improve lipid profiles in diabetic rats.23 -25 However, no studies reported its effect in modulating inflammation and repairing pancreatic beta-cell dysfunction. Hence, this study was designed to explore the effects of C. nutans extracts on lipid peroxidation, proinflammatory cytokines TNF-α and IL-6, and Langerhans islets areas.
Material and Methods
Plant material
C. nutans leaves were obtained and determined from Balai Materia Medica Batu, Indonesia. The leaves were ground to a fine powder. For 3 days, C. nutans leaf powder was soaked in 70% ethanol using the maceration method and incubated at room temperature with gentle shaking. The extract was concentrated using a rotary evaporator (Buchi, Flawil, Switzerland), forming a thick extract.
Chemical
Streptozotocin (STZ) was purchased from Sigma-Aldrich Chemicals (Singapore), stored at −20°C, and protected from sunlight. The enzyme-linked immunosorbent assay (ELISA) kits for IL-6 (Multi science, catalog number EK306/2) and TNF-α (Multi science, catalog product number EK382/2) were purchased from Multi science distributor. All other chemicals were obtained from Brataco. Corp.
Animals
Male Wistar rats weighing 150 to 200 g were used as experimental animals in this study. All rats were housed in collective cages at the Laboratory of Pharmacology, Faculty of Medicine, Universitas Airlangga, under controlled temperature with a 12/12-hours light/dark cycle. Standard rodent food and water were provided to all rats ad labium before and during the experiment. All processes were guided by the principles in the Guide for Care and Use of Laboratory Animals by the National Academics of Science. Ethical clearance was obtained from the ethics committee in the Faculty of Medicine, Universitas Airlangga (Ethics number: 146/EC/KEPK/FKUA/2019).
Diabetic induction
Rats fasted for 4 hours before induction to empty their stomach and reduce the risk of aspiration. Rats were induced with STZ intraperitoneally at a dose of 50 mg/kg body weight (BW) dissolved in 0.1 M sodium citrate buffer and at a pH of 4.5. After the STZ injection, the rats were administered a 10% dextrose solution overnight to avoid sudden hypoglycemic post-injection. Blood glucose levels were measured from the tail vein 2 days post-injection using a glucometer. Rats with fasting blood glucose grades of ⩾200 mg/dL were considered in diabetic rat models and utilized for this experiment.6,20,26 Fasting blood glucose levels were measured in all diabetic rat models before starting the experiment.
Experimental treatment
The Federer formula {(t−1)(r−1) > 15}has been used to determine the sample size, which considers the number of treatment groups (t) and the number of replications or sample size (r). The formula resulted in a minimum sample size of 4.75 for our study with 5 treatment groups.
Twenty-five diabetic rat models were randomly assigned into 5 clusters comprising 5 animals. Clusters 1, 2, and 3 were administered with C. nutans leaf extract in aqueous suspension with vehicle 1% Na-CMC at 75 mg/kg BW, 150 mg/kg BW, and 300 mg/kg BW. Cluster 4 comprising diabetic control rats was administered with metformin at a 21 mg/rat dose. Cluster 5 comprising a control diabetic rat was administered with 1% Na-CMC suspension. Treatment was provided intragastrically for 14 days. BW was measured at pre- and post-treatment.
Determination of proinflammatory cytokine and lipid peroxidation
At the end of the experiment, after a 12-hour fast, the rats were decapitated. Blood specimens were extracted through cardiac puncture and centrifuged at 4000g, at 4°C for 15 minutes to collect the serum. The serum IL-6 and TNF concentrations were determined using a rat ELISA kit. MDA levels were calorimetrically evaluated using the Thiobarbituric acid reactive substance technique.
Histopathologic examination
The pancreas was rapidly cleared, cleaned with normal saline, and fixed in 10% buffered formalin for histological examination. The tissue was embedded in paraffin using a tissue-embedding technique, sectioned at 3 to 5 μm thickness, and dyed with hematoxylin and eosin (HE). All tissue slices were visualized using an optical microscope Olympus CX31. Histological analysis was conducted by counting the Langerhans islets area using cell SENS and ImageJ software. The Langerhans islet was observed on each slide at 400× magnification. The total of all Langerhans islet areas per slide was calculated to determine the general area.
Statistical analysis
All data were analyzed by one-way analysis of variance, followed by a post hoc test using Statistical Package for the Social Sciences Statistics for Windows, Version 27.0.1.0 (IBM Corp., NY, USA) statistic software. Significant differences between clusters are set at P < .05.
Results
Effects of C. nutans extract on BW in diabetic rats
A standard indicator of DM is decreased BW. In this study, diabetic rats displayed a declined BW, similar to rats administered with metformin and C. nutans extract, except at a dose of 75 mg/kg, a slight gain in BW is observed (Table 1).
Body weight (BW) at pre- and post-treatment.

Data are presented as means ± standard deviations. Cluster 1, diabetic rats administered. with C. nutans leaf extract at doses of 75 mg/kg; Cluster 2, diabetic rats administered. with C. nutans leaf extract at doses of 150 mg/kg; Cluster 3, diabetic rats administered. with C. nutans leaf extract at doses of 300 mg/kg; Cluster 4, diabetic rat model administered. with metformin at doses of 21 mg/rat; Cluster 5, control diabetic rat.
Effects of C. nutans extracts on MDA, TNF-α, and IL-6 serum levels in diabetic rats
In diabetic rats, the IL-6, MDA, and TNF-α serum levels were upregulated. Treatment with C. nutans extracts significantly reduced lipid peroxidase levels in all clusters as compared to untreated diabetic rats (p cluster 1 = .004, p cluster 2 = .007, p cluster 3 = .001), and in the cluster receiving metformin (p cluster 4 = .002). No significant differences were observed between clusters 1, 2, and 3 (Figure 1a). The grade of proinflammatory cytokines TNF-α in clusters treated with C. nutans extract and metformin was significantly lower than that of control diabetic rats (p cluster 1 = .005, p cluster 2 = .001, p cluster 3 = .000, p cluster 4 = .08) (Figure 1b). Similarly, the proinflammatory cytokine IL-6 level in clusters treated with C. nutans extract and metformin was significantly lower than that of the control diabetic rats (p cluster 1 = .73 p cluster 2 = .73, p cluster 3 = .01, p cluster 4 = .08). These results suggest that C. nutans extract may reduce oxidative damage and inflammation (Figure 1c).
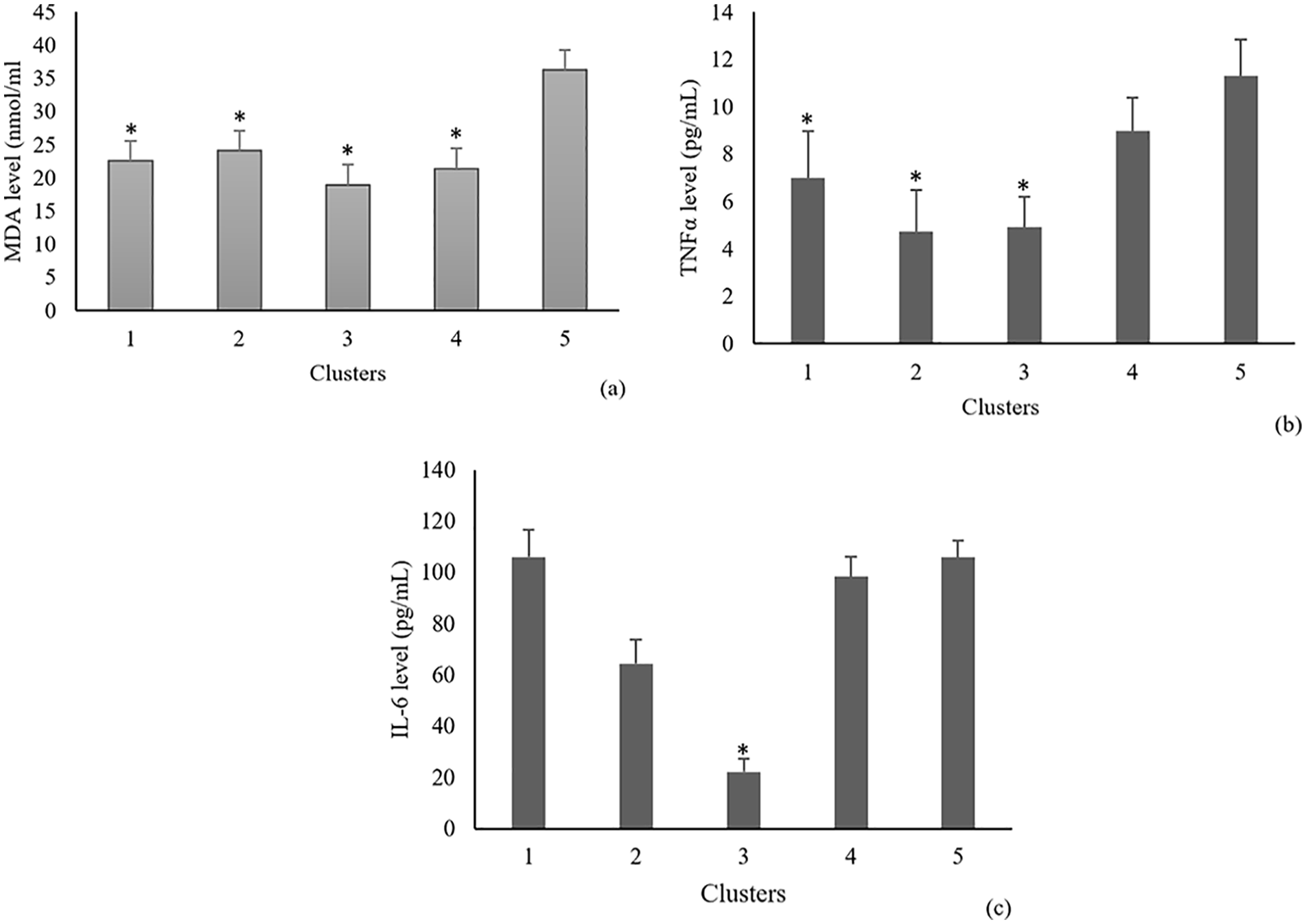
MDA (a), TNF-α (b), and IL-6 (c) serum levels post-treatment with C. nutans extract in diabetic rats model. Data are presented as means ± standard deviations. Cluster 1, diabetic rats administered. with C. nutans leaf extract at doses of 75 mg/kg; Cluster 2, diabetic rats administered. with C. nutans leaf extract at doses of 150 mg/kg; Cluster 3, diabetic rats administered. with C. nutans leaf extract at doses of 300 mg/kg; Cluster 4, diabetic rat model administered. with metformin at doses of 21 mg/rat; Cluster 5, control diabetic rat.
Effects of C. nutans extracts on the Langerhans islet area in diabetic rats
Diabetic rats had damage and pancreatic atrophy, which reduced the size of the Langerhans islets. Nevertheless, the cluster treated with C. nutans extract had a significantly higher area of Langerhans islets than diabetic rats not receiving treatment (p cluster 1 = .005, p cluster 2 = .002) (Figure 2). The findings demonstrated that C. nutans effectively repaired the area of damaged islets in diabetic rats.

Effects of C. nutans Extracts on the Langerhans Islet Area in Diabetic Rats based on histopathological examination of the pancreas [HE stain, 400x]. Histopathological image of the Langerhans islet area (yellow arrow) (a), Langerhans islet area calculation (b). Cluster 1, diabetic rats administered. with C. nutans leaf extract at doses of 75 mg/kg; Cluster 2, diabetic rats administered. with C. nutans leaf extract at doses of 150 mg/kg; Cluster 3, diabetic rats administered. with C. nutans leaf extract at doses of 300 mg/kg; Cluster 4, diabetic rat model administered. with metformin at doses of 21 mg/rat; Cluster 5, control diabetic rat.
Discussion
As previously described, hyperglycemia causes oxidative stress and inflammation27 -30 and induces mitochondria and endoplasmic reticulum to produce free radicals, reactive oxygen species (ROS), and reactive nitrogen species. Oxidative stress occurs due to an imbalance between a large number of free radicals and the inability of endogenous antioxidants, such as glutathione peroxidase, catalase, and superoxide dismutase. 11 MDA is a byproduct of lipid peroxidation, which may be defined as the oxidative degradation of polyunsaturated lipids. 31 This study indicated a notable accumulation of the MDA level in the diabetic rat model, indicating the presence of oxidative damage, and providing an appropriate model to assess the antidiabetic activity of C. nutans extract. This study confirmed that C. nutans extract significantly decreased the serum IL-6, MDA, and TNF-α levels. Although MDA levels slightly increased in cluster 2, the increase was not significant; thus, this might be due to physiological processes. The result was corroborated by an earlier in vivo study that showed comparable results. MDA and TNF-α levels were markedly decreased in the aortic tissue of diabetic rats treated with C. nutans methanolic extract. 32 These effects of C. nutans extract in diabetic rats were comparable to those of metformin, indicating that C. nutans extract has antioxidant activity in the rat DM model. Several plants, such as Nigella sativa, Mulberry juice, chicory, chamomile tea, bell pepper, and golden sea cucumber, can reduce blood glucose levels through their antioxidant properties.33,34
Oxidative stress initiates inflammation via Nf-κβ and c-Jun N-kinases (JNK). 35 The production of proinflammatory cytokines IL-6 and TNF-α was regulated by a transcription factor, NF-kB. 36 DM increases JNKs, which are significant regulators of inflammation. TNF-α overexpression is a potent JNK regulator. 37 Diabetic animals display significantly increased proinflammatory cytokine marker levels, such as IL-1β, IL-6, and TNF-α. These recent results showed that the administration of C. nutans extracts in diabetic rats significantly reduced serum levels of proinflammatory cytokines TNF-α and IL-6, as did metformin. The current study results are similar to that of the previous in vitro study demonstrating lowered TNF-α in lipopolysaccharide-induced inflammation of RAW264.7 cells via C. nutans leaf extract. 38 Abdulwahid et al observed decreased IL-6 production in high-fat diet mice that could be attributed to the phenolic compound in C. nutans extract. 39 In obesity, an increased body mass index increased proinflammatory mediators, including IL-6, and corresponds with insulin resistance and an increased risk of type 2 DM. 29 Therefore, the findings of the current study indicated that C. nutans leaf extract could be a potential antidiabetic medicine as it demonstrated a comparable therapeutic outcome to metformin.
Oxidative stress has a crucial role in the cell cycle of pancreatic β cells. ROS accumulation suppresses the cyclins D1 and D2 expressions, which are proteins responsible for inducing cell proliferation, and increases the p21, and p27 expressions, which inhibit cyclin D1. Meanwhile, ROS caused FoxO1 translocation, a nuclear activator, from the cytoplasm into the nucleus and inhibited Pdx1. Likewise, ROS also instantly downregulates transcription factors MafA, Pdx-1, Ngn3, and Nkx6.1 and reduces β-cell differentiation and proliferation.27,40 This study shows that DM reduces the Langerhans islet area. Pancreatic β cells are susceptible to oxidative stress and inflammation, causing cell damage, and apoptosis. Therefore, antioxidant supplementation is a promising therapeutic approach for β-cell regeneration through neogenesis and proliferation.11,27
The potential of C. nutans extract in reducing oxidative stress and inflammation in diabetic rats is related to its various phytochemical contents. Phytochemical tests found high phenolic and flavonoid contents in this extract with polar solvents.41,42 Furthermore, in vitro antioxidant activity tests showed that C. nutans extract has strong antioxidant potential through mechanisms including scavenging free radicals 2,2-diphenyl-1-picrylhydrazyl,42,43 inhibition of production of free radicals in macrophage cells, and elimination of nitric oxide and hydrogen peroxide.44,45
The computational simulation confirmed the mechanism of action of C. nutans extract as an antioxidant and anti-inflammatory medication. The compounds apigenin, 3,3-di-O-Methylellagic acid, Clinacoside, and Vitexin show high binding affinity to Aldose reductase, an enzyme crucial in the polyol pathway and trigger ROS accumulation46,47. Conversely, Clinacoside, Shaftoside, Vitexin, Orientin, Isovitexin, and Isoorientin exhibit an affinity for nitric oxide synthase, an enzyme that catalyzes the nitric oxide production by the vasculature, which plays an important role in triggering oxidative stress in inflammatory conditions. 48
By proving the effectiveness of herbal plants, their safety should be ensured before administering them to humans. Evaluation of the toxicity of C. nutans extract has been widely reported in several studies. The acute toxicity test did not show any toxicity to vital organs, such as the liver, kidneys, heart, lungs, spleen, or other essential organs, and did not result in mortality.48,49 Similarly, the sub-chronic toxicity test showed no significant changes in all hematological, biochemical, and histopathological parameters.49,50 Meanwhile, a sub-chronic toxicity test discovered liver and kidney damage at 1000 mg/kg BW for 28 days. 51 Therefore, its use must still be cautiously administered.
It is important to mention that this study only used animal models and further clinical studies are necessary to confirm the effects of C. nutans leaf extract on humans with DM. Additionally, the study did not comprehensively compare the effects of C. nutans leaf extract with other standard DM treatments, so more research is needed to determine if it can be used on its own or in conjunction with other treatments. Furthermore, the study did not include power analysis for sample size calculation, which can result in issues such as insufficient statistical power, a higher likelihood of type II errors, uncertainties in effect size estimation, and concerns about the generalizability of the findings.
Conclusion
This study revealed that the Clinacanthus nutans L extracts reduce serum levels of tumor necrosis factor-α (TNF-α), malondialdehyde (MDA), and interleukin-6 (IL-6), which are known to be elevated in diabetes and contribute to inflammation and oxidative stress in the diabetic rat model induced with STZ. This result affirms that the extracts demonstrate potential anti-inflammatory and antioxidant properties. Furthermore, the study discovered that Clinacanthus nutans L extracts improve the Langerhans islet area, which is significant in diabetes as it is responsible for insulin production. The enhancement of the Langerhans islet area suggests that the extracts positively impact pancreatic function and insulin secretion. The results suggest that extracts of Clinacanthus nutans L have therapeutic potential in managing diabetes, as they may help regulate blood sugar levels, reduce inflammation and oxidative stress, and improve pancreatic function.
