Abstract
Objective:
Short Synacthen tests (SSTs) are expensive, dependent on Synacthen availability, and need supervision. To reduce SST testing, we examined the utility of pre-test cortisol (Cort0) and related parameters in predicting outcome.
Design and Measurements:
We retrospectively examined the following in all SSTs; (i) Cort0 (ii) indications (iii) and time and place of testing. Receiver operated characteristic (ROC) curves were devised for Cort0 to obtain the best cut-off for outcome prediction in those who had SSTs between 8 and 10 am (Group 1) and at other times (Group 2).
Results:
Of 506 SSTs, 13 were unsuitable for analysis. 111/493 SSTs (22.5%) were abnormal. (1) ROC curves predicted – (a) SST failure with 100% specificity when Cort0 was ⩽124 nmol/L (Group 1), or ⩽47 (Group 2); (b) a normal SST with 100% sensitivity when Cort0 ⩾314 nmol/L (Group 1) and ⩾323 nmol/L (Group 2). (2) There was significant correlation between Cort0 and 30-minute cortisol (rs = 0.65-0.78, P < .001). (3) Median Cort0 was lower in those who failed SSTs compared to those who passed (147 vs 298 nmol/L respectively, P < .001). (4) SST failure was commoner in Group 1 vs 2 (P = .001). (5) There was no difference in outcome between out-patient and inpatient SSTs. (6) SST failure was most common for ‘steroid related’ indications (39.6%, P < .001).
Conclusions:
This study indicates that (1) Cort0 ⩾ 323 (Group1) and ⩾314 nmol/L (Group 2) predicted a normal SST with 100% sensitivity; (2) Using these cut offs 141/493 (28.6%) tests may have been avoided; (3) supporting evidence should be considered in those with a lower pre-test predictability of failure.
Introduction
The short Synacthen test (SST) is the most commonly used test for assessing adrenal reserve in clinical practice. SSTs are easy to perform, have relatively few contraindications, and may be used to investigate suspected hypoadrenalism of different aetiologies – primary (Addison’s), secondary (pituitary adenomas) or tertiary (steroid or opioid induced). Also, 30-minute post Synacthen cortisol values correlate well with the insulin stress test (the ‘gold standard’).1,2 The validity of SSTs is thought to be unaffected by the time or circumstances of its performance. 3 Its clinical utility is therefore excellent. However, its usefulness is limited by its expense, its intermittent unavailability, 4 the need for clinical supervision, and the inconvenience to patients of having to attend hospital. The cost of Synacthen has risen sharply in the UK in recent years (£38-45.60) and the costs of reagents, consumables, and nursing and technician time should also be added to the total cost.
To eliminate the need for formal SSTs, the utility of random or pre-test cortisol (Cort0) concentrations, and test associated factors have been investigated by clinicians as predictors of SST outcome in previous studies both in the UK and elsewhere.5-15 These studies have defined different pre-test cortisol values for predicting outcome. This difference is likely explained by the patient cohorts studied, the different assay methodology used and the variability in numbers analysed. We retrospectively analyzed the results of SSTs done in our Health Board in 1 calendar year to establish local standards for patients in South Wales.
Aims of project – We aimed to assess the utility of the following factors in predicting SST outcomes – (1) pre-test cortisol (Cort0) concentrations, (2) pre-test SST related factors such as (a) time of day when SST was done; (b) inpatient or outpatient tests; and (c) indications for testing.
Methods
We retrospectively analysed data from 506 subjects who had SSTs during a single calendar year in the 3 acute hospitals of the Aneurin Bevan University Health Board (ABUHB) in South Wales, UK. Data were obtained from the biochemistry department’s database and were anonymized. This was a service improvement project approved by the Research and Development Department of ABUHB and did not require approval by an Ethics Committee.
SST protocol and testing: The decision to perform a SST was taken by the admitting physician at the admissions or general medical wards, or by an endocrinologist following inpatient referral or outpatient clinic review. SSTs were done according to a standard protocol – (a) those on steroids refrained from taking the morning dose on the day of the test if on a short acting preparation (hydrocortisone), and the previous afternoon/evening’s dose and the morning dose on the day of the test if on a longer acting steroid (eg, prednisolone); (b) an intravenous cannula was inserted half hour before the test for blood sample collection and injecting Synacthen 250 mcg (Abbott Diagnostics, North Illinois, Chicago, USA); (c) blood samples were collected immediately before (pre-test sample) and 30 minutes after the injection of Synacthen for serum cortisol assays; (d) tests were done irrespective of time of day; (e) both in the outpatient and inpatient settings. The local laboratory established cut off for SST failure was a serum cortisol concentration of ⩽450 nmol/L at 30 minutes after injection of Synacthen. We used the 30-minute cortisol cut off as it is validated against the insulin tolerance test,2,16 although the Endocrine Society accepts both 30 and 60-minute post Synacthen samples as valid. 17 We acknowledge that this strategy may result in between 7% and12% false positive results (which normalise in the 60 minute post Synacthen sample in the 60 minute SST) but have done so in line with local practice and pragmatic reasons. 18
Cortisol Assay: During the period under study, serum cortisol was assayed using a single assay, the Architect Assay (Abbott Diagnostics, North Illinois, Chicago, USA). The manufacturer’s 95% reference interval for specimens before 10 am was 101.2 to 535.7 nmol/L and for specimens after 5 pm was 79-477.8 nmol/L.
Statistical analysis: Data are shown as mean (± standard deviation, S.D) or median (interquartile range – IQR) as appropriate. We used non-parametric methods for comparison of parameters which were not normally distributed. Frequencies were compared with the chi squared test. Spearman rank analysis was used to test for univariate relationships between basal and 30 minutes cortisol concentrations for 2 cohorts, Groups 1 and 2 as described below, in both males and females.
To analyse pre-test cortisol as a predictor of SST outcome, we analysed the subjects in 2 groups – (a) those who had SSTs between 8 and 10 am (Group 1), and (b) those who had SSTs at other times of the day (Group 2).
We analysed receiver operated characteristic (ROC) curves for each possible test threshold or cut-off. An area under the ROC curve (AUC) (95% confidence intervals, CI) was used to express the overall accuracy of cortisol levels – an estimate of 0.5 indicates no discrimination, whereas 1.0 indicates perfect discrimination. A P < .05 was deemed to be significant. Statistical analyses were performed using AnalyseIt software (AnalyseIt, Yorkshire, UK).
Results
During the calendar year 1st February 2017 to 31st January 2018, 506 SSTs were done of which 493 were available for analysis (data unavailable for 11 and incomplete in 2 subjects). The median age (interquartile range, IQR) of subjects in Group 1 was 58 years (41, 73) (n = 165) and Group 2 was 60 years (45, 76) (n = 328), respectively. The indications for SSTs are detailed in Table 1.
Using the above local criteria, 111/493 (22.5%) failed the SST.
(a) (i)
(ii)
(b) There was a significant correlation between Cort0 and 30 minute cortisol (Cort30) in both groups (rs = 0.65, 0.78, respectively, P < .001).
(c) Median Cort0 (IQR) was significantly higher in those with a normal SST (298.5 [227-393] nmol/L) compared to those who failed the SST (147.0 [91-213] nmol/L) (P = .001) (Table 2).
(2)
(i) A higher SST failure rate was observed in Group 1 (46/165 [27.9%]), compared to Group 2 (65/328 (19.8%]) (P = .001). However, there was no significant difference in subjects with ‘steroid related’ indications in the 2 groups to account for this difference (P = .094).
(ii) There was no difference in the prevalence of SST failure between subjects who were in-patients in general medical wards (66/308, 21.4%) compared to those who were outpatients (38/185, 20.5%) (P = .063).
(3)
(4)
Indications for testing and SST failure.

Only ‘steroid related’ indications and weight loss were statistically different between the 2 different outcome groups (P = .001, .013, respectively). Other common indications showed no difference.

ROC analysis for Group 1 (8-10 am) (n = 165) and Group 2 (tests done at other times) (n = 328): (a) and (b) show the results of ROC curve analysis of Groups 1 and 2. Pre-test cortisol (Cort0) has good discriminatory value as evidenced by the AUC for each group.
Median pretest cortisol values in normal and failed SST.

Median pre-test cortisol concentrations were significantly different between the group who failed the test and passed it (P = .001).
Time and place of test and SST failures.
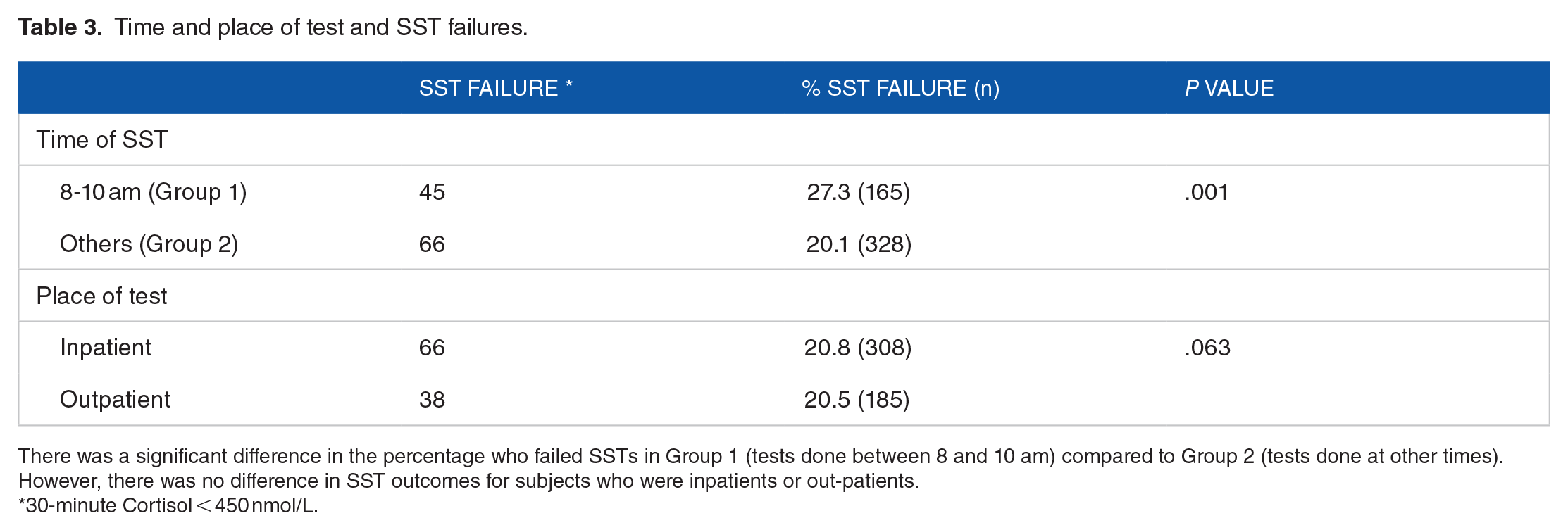
There was a significant difference in the percentage who failed SSTs in Group 1 (tests done between 8 and 10 am) compared to Group 2 (tests done at other times). However, there was no difference in SST outcomes for subjects who were inpatients or out-patients.
30-minute Cortisol < 450 nmol/L.
Discussion
We have shown in this retrospective analysis of 493 SSTs from 3 acute hospitals in South Wales that 22.5% of all SSTs were abnormal. Furthermore, pre-test cortisol (Cort0) of ⩾314 nmol/L in those who had early morning SSTs (Group 1) or ⩾323 nmol/L in those who had SSTs at other times (Group 2) would identify a normal SST with 100% sensitivity. Also a Cort0 of ⩽124 nmol/L (Group 1) or ⩽47 nmol/L (Group 2) would identify SST failure with 100% specificity (Figure 1a and b). These cut-off values if used, could have prevented 141/493 (28.6%) SSTs in this population of patients, with substantial cost savings and an improved patient experience.
In this study, SST failure occurred in a higher percentage of subjects – (a) who had tests done between 8 and 10 am (Group 1) (P = .001); and (b) who had SSTs done for ‘steroid related’ indications such as long-term steroid therapy, steroid withdrawal or a ‘low’ random cortisol (P = .001) (Table 1). While most studies report no influence of the timing of testing on SST outcome,3,19,20 1 study reported a higher failure rate in the afternoon. 21 We cannot explain why our study is different, although 2 factors may be pertinent – (a) the proportion of patients with ‘steroid related’ indications (who had a higher failure rate) is not a plausible explanation as this was similar in Group 1 and Group 2 (P = .094); (ii) higher cortisol levels in the morning reflecting the cortisol circadian rhythm may necessitate a higher cut-off for diagnosing SST failure in Group 1. These views are currently only speculative and need to be explored in further studies.
There was no difference between those who had SSTs done as outpatients compared to those who were inpatients. The subjects in the ‘in-patient’ group were non-critically ill and had illnesses generally seen in acute medical wards and not in ITU. Therefore these results cannot be extrapolated to critically ill patients in ITU settings.
The SST has been the mainstay of testing for ‘adrenal reserve’ in suspected primary adrenal or HPA axis dysfunction since its introduction many years ago. It is known that tetracosactide (the active component in Synacthen) is equipotent to natural ACTH for adrenal stimulation, and comparisons with the insulin stress test has validated its use in adults. 2 The clinical utility of the SST has hitherto depended on the ready availability of Synacthen, its low cost, the paucity of contraindications and its clinical equivalence to the insulin stress test. However, there has been a recent price hike in the UK for Synacthen, and a shortage of Synacthen due to manufacturing issues. 4 These coupled with the fact that patients are inconvenienced during multiple hospital visits have prompted clinicians to examine random or pre-test cortisol levels as guidance for the need for a formal SST. We felt that establishing standards to include other pertinent SST related parameters was desirable for our local population.
There have been many studies which examined the utility of a single serum cortisol value in predicting SST failure.5-15,18 No clear consensus has emerged from these studies because of differences between populations, protocols (cortisol cut off denoting a satisfactory response), and cortisol assays used. In the UK, 4 large studies analysed SSTs in their regions. A retrospective study from Sheffield in 3223 subjects indicated a pre-test cortisol of ⩽103 nmol/L predicted SST failure with 100% sensitivity, and a cortisol of ⩾574 nmol/L predicted a normal SST with 100% specificity. 22 A retrospective study from Cambridge which examined 450 subjects showed limits of ⩽56 nmol/L for test failure and ⩾374 nmol/L for a normal SST. 23 A retrospective study from Edinburgh 15 of 1624 SSTs, compared cortisol cutoffs between morning and afternoon, and outpatient and inpatient cohorts, and demonstrated a morning (8 am-12 pm) cortisol of ⩽275 nmol/L and an afternoon cortisol of ⩽250 nmol/L identified abnormal SSTs with a 96.2% and 96.1% sensitivity respectively. However, only the morning value was sufficiently sensitive for SSTs in inpatients. Another retrospective study from Bedford of 393 SSTs confirmed a morning serum cortisol of 354 nmol/L or more predicted normal adrenal function with a sensitivity of 100%. 24 The findings of our study therefore need to be interpreted with care and as applying only to the population tested in this analysis and the assay we have used. We would also like to highlight the strengths and limitations of our study – (1) strengths (a) the availability of a large number of SSTs for analysis; (b) the use of a single cortisol assay throughout the study period; and (c) the same laboratory being utilised for all analyses; (2) limitations – (a) the inherent disadvantages of a retrospective analysis, (b) the inability to compare with populations using different assays (and therefore different reference ranges and ‘cut-offs’), (c) the use of ‘total cortisol’ in the biochemical analysis (disregarding the variability of cortisol binding globulin concentrations in relevant situations), and (d) the lack of ‘critically ill’ subjects (in ITU for instance). Furthermore, the lack of guidance with regard to indications for SST, and the failure to assess pre-test probability of SST failure by individuals ordering SSTs, are shortcomings applicable to this study.
Conclusions
This retrospective analysis indicates that pre-test cortisol concentrations may be useful as an indicator of SST outcomes in this South Wales population. Using cut-off values which indicate a normal outcome with high specificity, it would have been possible to avoid 28.6% tests in this group, with cost savings and an enhanced patient experience. The higher failure rate in those who had SSTs in the morning is currently unexplained and needs to be explored in future studies. We recommend that multiple SST related factors should be considered in those who have indications with a low predictability of outcome, before the SST is done. However, we wish to emphasize that these results should not be extrapolated to populations using other assay methodology or to critically ill patients.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
