Abstract
Hyperinsulinemia promotes fat accumulation, causing obesity. Being an inflammatory state, obesity can induce further inflammation and is a risk factor for HPA (hypothalamic pituitary axis) dysregulation through hypercortisolism-related hyperglycemia. In another hypothesis, the sympathetic nervous system (SNS) plays a significant role in the regulation of hormone secretion from the pancreas such as an increase in catecholamines and glucagon as well as a decrease in plasma insulin levels, a disruption on SNS activity increases insulin levels, and induces glycogenolysis in the liver and lipolysis in adipose tissue during hypoglycemia. Hyperglycemia-hyperinsulinemia exacerbates inflammation and increases the oxidative stress along with regulating the levels of norepinephrine in the brain sympathetic system. Increased inflammatory cytokines have also been shown to disrupt neurotransmitter metabolism and synaptic plasticity which play a role in the development of depression via inhibiting serotonin, dopamine, melatonin, and glutamate signaling. An increased level of plasma insulin over time in the absence of exercising causes accumulation of lipid droplets in hepatocytes and striated muscles thus preventing the movement of glucose transporters shown to result in an increase in insulin resistance due to obesity and further culminates into depression. Further hyperinsulinemia-hyperglycemia condition arising due to exogenous insulin supplementation for diabetes management may also lead to physiological hyperinsulinemia associated depression. Triple therapy with SSRI, bupropion, and cognitive behavioral therapy aids in improving glycemic control, lowering fasting blood glucose, decreasing the chances of relapse, as well as decreasing cortisol levels to improve cognition and the underlying depression. Restoring the gut microbiota has also been shown to restore insulin sensitivity and reduce anxiety and depression symptoms in patients.
Introduction
Disorders pertaining to metabolic abnormalities have frequently been described as cofactors in the progression of disease in patients with mental health problems. Rising rates of obesity, diabetes mellitus (DM), and depression have energized the focus on understanding the links between hyperinsulinemia and depression. A commercial diet with high fructose also exhibits a hyper insulinemic state. 1 Endogenous insulin is a peptide hormone produced by beta cells of islets of Langerhans of the pancreas, whereas exogenous insulin is any insulin that the body does not make on its own: that one receives via injection, or insulin pumps. Insulin regulates the metabolism of carbohydrates, fats, and protein. Insulin directly or indirectly affects every organ including adipose tissue, liver, muscle, brain, bone, kidneys, and vasculature. Seventy-five percent of total insulin secretion occurs in pulsatile fashion,2,3 first transported to the liver through the portal system and thus pulsatile insulin secretion preserves the sensitivity to insulin. 4 Impaired pulsatile insulin secretion is seen in type 2 DM. 5 Excessive insulin secretion in insulinoma and non-insulinoma pancreatic hypoglycemic syndrome leads to hypoglycemia. Dysregulated hyperinsulinemia, a condition in which there is an excess insulin level circulating in blood in relationship to its usual level relative to the blood glucose level, which does not lead to hypoglycemia. 2 Dysregulated hyperinsulinemia is seen in patients with insulin resistance. Herein, we use term hyperinsulinemia for dysregulated hyperinsulinemia.
Hyperinsulinemia is a state in which too much free insulin is residing in the body at a given time due to various physiologic processes taking place. This is not to be confused with insulin resistance, the process in which there is an inability of the hepatocytes, straited muscles, and neurons to internalize glucose. Insulin resistance is defined as a subnormal biological response to normal insulin concentrations. 6 The triglycerides synthesized after high fructose, sucrose, fat, or alcohol consumption lead to formation of lipid droplets. The lipid droplet size and location are the major predeterminants of insulin resistance in skeletal muscles. Large sized droplets, mainly found in obese sedentary individuals and type 2 diabetes patients, obstruct the translocation of glucose and hence impair glucose uptake. 1 Hyperinsulinemia is the result of the pancreas producing more insulin to circumvent the insulin resistance and help the cells take in glucose. Hyperinsulinemia can result from a variety of metabolic diseases and conditions. Some endurant athletes have insulin sensitivity that is roughly 3 times higher than healthy nonathletes. It means these athletes rapidly consume the sugar from their bloodstream and into their muscles without having to produce excessive amounts of insulin. In obesity, increased release of tumor necrosis factor-1 (TNF-1) and decreased release of protective adipocytokine, adiponectin results in pathogenesis of insulin resistance. 7 Insulin secretion and clearance are regulated processes that influence the development and progression of hyperinsulinemia. Environmental, genetic, and dietary factors are associated with hyperinsulinemia. 2 Through the years, researchers have explored the relationship between depression and hyperinsulinemia. A study based on multivariate analysis of 79 plasma metabolites in recurrent major depressive disorder (MDD) patients revealed hyperinsulinemia had the highest significance level for depression after adjustment for glucose levels, body mass index (BMI), and fasting time. 1
Hyperinsulinemia is associated with hypertension, obesity, dyslipidemia, and glucose intolerance, which is collectively known as Metabolic Syndrome. There are controversial reports in the literature, showing hyperactivity of HPA axis in obesity. It is uncertain whether dysregulation of HPA axis leads to obesity or whether obesity hyperactivates HPA axis. 8 HPA axis hyperactivity results in increase in corticotropin releasing hormone (CRH) and cortisol secretion, and impaired efficacy of glucocorticoid-mediated feedback, which contribute to the pathogenesis of mood disorders. 9 Several studies point to hyperactivity of HPA axis as the state marker of major depression. 10 Hyperinsulinemia stimulates the HPA axis resulting in high cortisol levels.11,12 Cortisol also drives insulin resistance 13 via proliferation of adipokines and the secretion of proinflammatory cytokines. 14 A 2018 study by Wurtman and Wurtman 15 found the evidence of a bidirectional association suggesting obesity increased the risk of depression by 55% and depression increased the risk of becoming obese by 58%. The prevalence of depression in patients with pre-existing diabetes mellitus is about twice as high as compared to a group of healthy individuals. Prevalence of depression in people with Type 1 diabetes is about 12% than in their healthy counterparts (3.2%) 16 as well as being about 24% higher in those with preexisting type 2 diabetes. 17 The prevalence rate of depression in comorbid patients has been 10% to 15%.18-20 In another study, in patients already suffering from major depressive disorder, the likelihood of developing type 2 diabetes was 32% higher compared to healthy individuals and 41% higher chance of developing diabetes mellitus compared to the healthy group. 21 There was also a worse prognosis in patients who were diagnosed with both major depressive disorder and diabetes, as well as a higher rate of mortality in comparison to the healthier patient population. 21
The dysregulation within the HPA axis that causes an increase in cortisol along with the stress that creates a proinflammatory state can interfere with pancreatic beta cells which leads to an increase in resistance to insulin and an increased likelihood of developing type 2 diabetes in patients with depression. Psychological stress in patients with depression results in increase in counter-regulatory hormones like catecholamines, glucocorticoids, growth hormone, and glucagon which results in increased blood glucose level and leading to insulin resistance. 22 . Older adults with depression experiencing HPA dysregulation are more likely to develop comorbidity (ie, depression and a form of metabolic disorder) than younger adults. Management of these comorbid patients seems to yield more positive results when the treatment regimen is based around a multidrug approach via SSRIs, bupropion along with cognitive behavioral therapy, all of which when given together show an improvement in glycemic control, blood glucose levels, and cognition. In clinical setting of diabetes management, exogenous insulin plays an important role in controlling diabetes which may lead to peripheral hyper insulinemic-hyperglycemic state and may have a similar effect as of physiological hyperinsulinemia associated depression?
Role of Sympathetic Nervous System (SNS) in Hyperinsulinemia and Insulin Resistance
Pancreatic islets are innervated by both branches of the autonomic nervous system. Parasympathetic nervous system activity stimulates secretion of insulin in hyperglycemic conditions (though the release of acetylcholine and its binding to muscarinic receptors (m3AchR) present on beta cells). Parasympathetic nervous system activity stimulates glucagon secretion during hypoglycemia. Sympathetic nervous system stimulates glucagon secretion and inhibits insulin secretion in response to hypoglycemia. 23
SNS also has effects on glycogenolysis in the liver and lipolysis in adipose tissue during hypoglycemia. Insulin appears to exacerbate inflammation and increase the number of markers of oxidative stress along with regulating the levels of norepinephrine in the brain.24,25 Increased SNS activity induces insulin resistance by promoting vasoconstriction, vascular structural changes, impaired endothelial function, and capillary rarefaction.26,27
Straznicky et al 28 were able to determine an inverse relationship between peripheral arterial stiffness and insulin clearance as well as a positive correlation between insulin clearance and insulin sensitivity. The same results were echoed in a study by O’Callaghan et al 29 which showed phenylephrine infusion during the euglycemic clamp thrust increased the mean arterial pressure and led to a reduction in insulin clearance as compared to placebo. Both studies potentially show cased an unrealized link between the actions of the SNS and its effects on insulin resistance at systemic levels. The release of insulin from the β-cells in the pancreas generally depends on the circulating levels of glucose but the opposing actions of the SNS and the parasympathetic nervous system may lead to the release of insulin as well30,31
An increased level of plasma insulin over time, however, has been shown to result in an increase in insulin resistance, further providing evidence of a connection between norepinephrine and hyperinsulinemia. 4 With norepinephrine causing an increase in the amount of glucose present in the plasma through gluconeogenesis, further insulin resistance has been seen in part due to a decrease in the conversion of glucose into glycogen, thus impairing the uptake across the cell membranes. 32
Baron et al 33 conducted a study to explore the relationship between insulin and norepinephrine on blood pressure. His data showed that during states of hyperinsulinemia, obese patients were more sensitive to the effects of norepinephrine than lean patients. 33 It was also suggested that the mean arterial pressure in obese individuals was generally 9% higher than non-obese. The main mechanism of high blood pressure in these obese patients was increased uptake of norepinephrine mediated by insulin, thus revealing an increased risk of hypertension with obesity and hyperinsulinemia. 33 Additionally, Liang and Cincotta 34 performed an experiment with ob/ob mice that sought to explore the effects of circulating norepinephrine on hyperglycemia, hyperinsulinemia, and hyperglucagonemia. Although full human model studies need to be conducted to further understand the link between hyperinsulinemia, norepinephrine, and depression, this study demonstrated various possible physiological links between the actions of the 2 hormones. 34
Exogenous Hyperinsulinemia and Insulin antibody
A study by Shen et al 35 found some evidence of high levels of insulin antibodies circulating in the blood of patients who had been diagnosed with diabetes mellitus and were being treated previously with exogenous insulin, potentially leading to a hyperinsulinemia–hypoglycemia state. Furthermore, a high insulin level in states of insulin resistance may result in hypertension due to insulin’s effect on the SNS with the release of catecholamines. 36 In another situation, our lab reported that exogenous concentrated insulin (U500) therapy cause insulin antibodies in T2D patient. 37 Hyperglycemia–hyperinsulinemia situation consequently increases the body weight. 37 Recently we have observed in our clinical setting that diabetic patients receiving exogenous insulin (U100-U500) exhibit a state of hyper insulinemia-hyper glycemia as this exogenous insulin does not pass through the hepatoportal system and hence this state of hyper insulinemia-hyper glycemia may lead to a similar effect as of physiological hyperinsulinemia associated depression (data not shown).
Proinflammatory State and Cytokine Activation
Furthermore, it is also thought emotional, psychological and physiological stressors all play a role in promoting the activation of cytokines and a proinflammatory state through the functions of IL-6, IL-1, and TNF-a. Because these factors are considered relevant messengers in the immune regulation of the HPA axis, erratic activation may lead to HPA dysregulation and a state of increased insulin resistance. Thus, the unhealthy lifestyle like noncompliance to medications, inability to follow a healthy diet or exercise regimen that often comes with depression may promote states of hyperinsulinemia through perpetuating a prolonged state of inflammatory response. A meta-analysis incorporating 24 studies where they investigated the cytokines in association with depression, showed consistent findings of elevated pro-inflammatory markers such as Tumor Necrosis Factor-Alpha (TNF-α), interleukin-6 (IL-6), and interleukin-1 beta (IL-1β) in those suffering from depression. This phenomenon could shed further light on depression and its connection to a sustained proinflammatory state. 38 Furthermore, another meta-analysis spread over a span of 40 years incorporating 361 studies was also able to show a relationship between depression and dysregulation of the HPA axis with varying degrees of significance within the groups. 39
HPA Axis Dysregulation Due to Stress
It has been suggested that dysregulation within the HPA axis during times of stress can lead to flattening of the diurnal cortisol curve, which in turn halts the cortisol awakening response. Both of these phenomena, in conjunction with being in a proinflammatory state, have been shown to increase the likelihood of increased insulin resistance and T2DM.40,41 Multiple studies conducted in the early 2000s were able to establish a connection between insulin resistance, diabetes, and the diagnosis of major depression disorder (MDD). They suggested prevalence for patients found to have been suffering with both MDD and diabetes was as high as 10% to 15% irrespective of sex.18-20 HPA axis dysfunction is seen as a prominent contributor along with consistent acute and generalized chronic stressors in the development of major depressive disorder and diabetes and one possible explanation could be that an increase in visceral adipose tissue may disrupt the regulatory effects of cortisol, leading to further hyperglycemia and insulin release, resulting in a state of hyperinsulinemia and subclinical hypercortisolism.40,42,43 The prevalence and incidence of MDD in diabetic patients has increased; depression is about 3 times higher in those with type 1 diabetes (T1D) and about twice as high in those with T2DM, in comparison to patients without diabetes in the general population around the world based on a systemic review on epidemiology of patients with diabetes and depression, along with a worse prognosis and an increase in mortality for those with both diabetes and depression rather than the depression alone.44-46 On the other hand, studies have shown that a diagnosis of major depressive disorder increases the risk of developing diabetes mellitus type 2 significantly when compared to a healthy population. 47 One such connection could be the possible socioeconomic status of an individual, with those in lower classes showing a higher likelihood of developing both MDD and diabetes.48,49 Stress resulting from hyperglycemic condition and low socio-economic status (SES) can cause HPA axis abnormalities, including activation within the hypothalamus, which leads to increased levels of inflammatory cytokines, which can also cause an increase in the resistance to insulin by interfering with β-cells in the pancreas and thus potentiating a risk for type 2 diabetes. Inflammatory cytokines have also been shown to disrupt neurotransmitter metabolism and synaptic plasticity which could potentially play a role in the development of depression in relation to chronic inflammation.50-53
Diabetes and Depression in the Context Lifestyle
It is hypothesized that acute hyperinsulinemia improves mood by increasing serotonin levels. Insulin enhances peripheral uptake of large neutral amino acids by skeletal muscles thus sparing tryptophan and in turn enhancing tryptophan and serotonin levels in brain. However chronic hyperinsulinemia leads to impairment of brain remodeling and thus the effects of hyperinsulinemia depend on the duration of hyperinsulinemia leading to synaptic long-term depression, neural apoptosis, and reduced dopamine secretion. 1 A slightly different perspective on the correlation between depression and diabetes may shine a light on their deeper relationship, as patients who are suffering from major depressive disorder may be less inclined to follow a steady, healthy diet or exercise regimen as well as not being compliant with taking their medications regularly as directed. These individuals may also be less inclined to show up to or make doctor’s appointments due to a lack of trust in others and an overall fear of being stigmatized and ultimately being judged by others due to their diagnosis. This experience could further lead to obesity, a sedentary lifestyle, and bad habits such as smoking and drinking. Low self-esteem, unhealthy diet, and staying physically inactive are examples of self-neglect that may be associated with an individual who is suffering from depression. 54 Studies have confirmed a higher rate of negligence toward self-care and self-help in those who suffer from both diseases, and this includes medical non-adherence when treating either depression or diabetes.55,56
Management in Comorbid Patients
Management of comorbid patients has often been approached with the goal of tackling both depression and diabetes at the same time. Petrak et al 57 suggested that treatment priority to depression should be given first, as once the patient is less depressed, they are more likely to adhere to their medication regimen. Treating comorbid patients with selective serotonin reuptake inhibitors (SSRIs), which increase serotonin activity by inhibiting the reuptake of serotonin, and cognitive behavioral therapy, a mode of therapy in which individuals are taught to identify and change negative thinking patterns, emotional responses and behaviors in response to triggers, has been shown to decrease cortisol levels, which in turn improves symptoms of depression and cognition. 40 ,58-60 Another strategy that has shown positive results is combining SSRIs with bupropion, an antidepressant that works by inhibiting the uptake of both dopamine and norepinephrine therefore increasing the availability of both. There is evidence that when patients receive bupropion, they have improved glycemic control, lower fasting blood glucose levels, and decreased rates of relapse61-63 These findings further advance the theory that when approaching a treatment plan with a comorbid patient, it is important to look at the individual and their symptoms, and base a strategy around their specific needs. Utilizing both SSRIs and bupropion, with CBT (cognitive behavioral therapy) as an adjunct therapy, have shown to produce the best results overall for the patient.
While it is important to continue studying the relationship between diabetes and depression by conducting further experiments, it is just as important to be aware of the individual situations of patients with both conditions. Often depression is overlooked in patients with diabetes, but efforts should be made to keep an open mind with such patients and to evaluate each patient individually. The evidence suggests it is better to catch and treat comorbid patients early in the course of the disease process to limit adverse outcomes in longevity and provide them with a healthier life. It is important to try to understand the bidirectional relationship between depression and diabetes, as it is an essential component of optimal patient care.
The Gut Microbiota
Clinical studies have demonstrated a decrease in both anxiety and depressive symptoms in patients who have had counseling aimed at restoring the gut microbiota, as well as there being evidence of restored insulin sensitivity in these same individuals. Studies have also shown that by reducing cytokine production by the gut microbiota by increasing homocysteine levels, patients were able to benefit from the antidepressant effects of caffeinated products such as coffee and chocolate. 1 ,64-67
Conclusion
Patients suffering from diabetes (both type 1 and type 2) receiving exogenous insulin to control the hyperglycemic condition may experience peripheral hyperglycemia–hyperinsulinemia, which increases the risk of acute/chronic stress leading to a proinflammatory state and causing dysregulation of the HPA axis. This malfunctioning of HPA axis can cause disarray in both the sympathetic and parasympathetic nervous systems. This could be seen as the reason for a higher rate of morbidity in diabetes associated major depressive disorder.
Comorbid patients have been shown to benefit from therapy concentrated around combining SSRI’s, buproprion, and cognitive behavioral therapy together. There is evidence of improved glycemic control, lower fasting blood glucose levels, and a decreased chance of relapse in these patients. There is also some evidence which suggests it is advantageous to catch and treat comorbid patients earlier in the course of the disease to limit adverse long-term outcomes.
Future Work
Further studies should focus on looking at the relationship between diabetes and major depressive disorder through hormonal changes brought forth by the dysregulation within the HPA axis as well as what changes take place due to the prolonged proinflammatory state and its relation to the insulin resistance. Effects of circulating norepinephrine on hyperglycemia, hyperinsulinemia, and hyperglucagonemia mediated lipogenesis and obesity need to be investigated in detail.
Diabetic hyperglycemic and hyper insulinemic state induces a pro-inflammatory state, this physiological situation causes HPA axis dysregulation promote glycogenolysis in liver and potentiate further hyperglycemia–hyperinsulinemia consecutively induce lipogenesis and obesity. In totality, this physiological situation culminates into a depressive disorder (Figure 1).
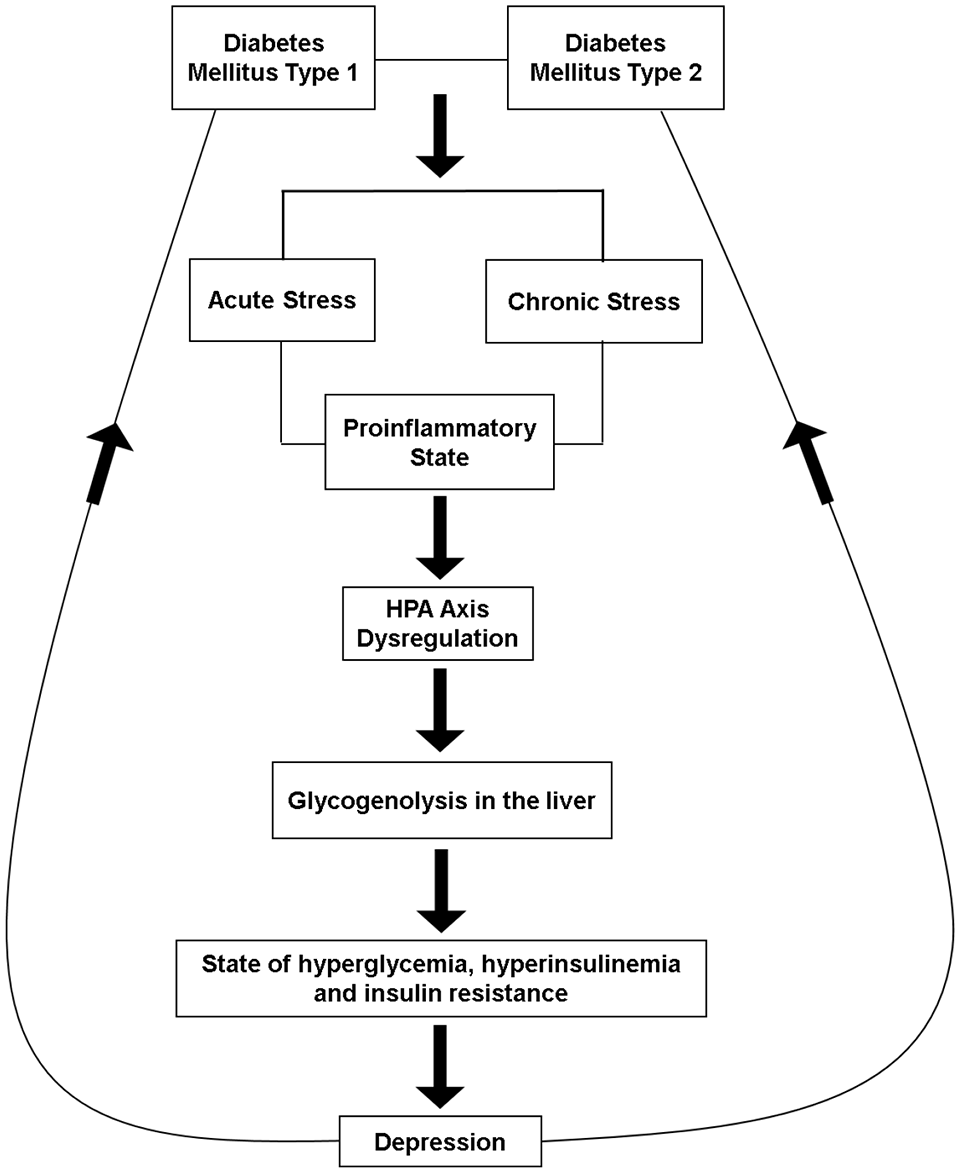
Sketch hypothesis.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by University of Toledo, College of Medicine and Life Sciences grants.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent for Publication
All Authors have approved the final article and given consent for publication.
