Abstract
Cystic fibrosis (CF) is an autosomal recessive chronic condition effecting approximately 70 000 to 100 000 people globally and is caused by a loss-of-function mutation in the CF transmembrane conductance regulator. Through improvements in clinical care, life expectancy in CF has increased considerably associated with rising incidence of secondary complications including CF-related diabetes (CFRD). CFRD is believed to result from β-cell loss as well as insufficient insulin secretion due to β-cell dysfunction, but the underlying pathophysiology is not yet fully understood. Here we review the morphological and cellular changes in addition to the architectural remodelling of the pancreatic exocrine and endocrine compartments in CF and CFRD pancreas. We consider also potential underlying proinflammatory signalling pathways impacting on endocrine and specifically β-cell function, concluding that further research focused on these mechanisms may uncover novel therapeutic targets enabling restoration of normal insulin secretion.
Keywords
Introduction
Cystic fibrosis (CF) is an inherited chronic condition caused by loss-of-function mutations in the gene coding for the CF transmembrane conductance regulator (CFTR) leading to impairment of anion transport across the cell membrane in epithelial cells mainly in lung, pancreas and gut.1,2 CF is considered the most common lethal autosomal recessive disease, with an estimated incidence of 1 in 2500 live births with a carrier probability of 1/36 in European populations.3,4 CF affects approximately 70 000 to 100 000 people globally, including around 10 600 people in the UK with 200 to 300 new cases diagnosed annually.3,5 Approximately 85% to 90% of people with CF have exocrine pancreatic insufficiency.6-8 Through incremental improvements in clinical care, life expectancy in CF has increased considerably associated with rising incidence of secondary complications including CF-related diabetes (CFRD). In childhood prevalence of CFRD is 2% increasing to 20% in adolescents and ultimately 33% to 50% in adults with CF.9,10 Diabetes in CF is associated with greater impairment in pulmonary function and increased mortality, although improvements in diabetes management are leading to decreases in mortality. 9 It has also been shown that individuals with CFRD have a significantly higher chance of chronic lung infections. 10 Patients are screened annually from as young as 6 to 9 years of age for impaired glucose tolerance with an Oral Glucose Tolerance Test, although screening can also include random and fasting blood glucose levels.9,11 Primarily, CFRD is believed to result from insufficient insulin secretion due to β-cell dysfunction, but the detailed pathophysiology is not yet fully understood.2,12-14
Architectural Remodelling in the Exocrine CF Pancreas
Iannucci et al 15 observed a total loss of exocrine pancreas with fat replacement in young adults with CFRD. A qualitative histological analysis of post mortem CF pancreata from patients aged over 5 years with relatively advanced pathology reported that exocrine acinar and duct cells were either absent or present in small amounts with a pattern of cystic dilatation. 16 This and other studies further demonstrated the presence of 2 distinct exocrine patterns observed during the end-stage CF: a fibrotic pattern, where the exocrine tissue was predominantly replaced by fibrotic tissue and a lipoatrophic pattern, where the exocrine tissue was predominantly replaced by fatty tissue.16,17
A study of pancreatic tissue including young children with CF reported the absence of exocrine lipofibrotic replacement and cystic dilatation of pancreatic ducts in very young children (<1 year old), but observed more severe exocrine changes in children between 1 and 4 years old. 18 A ferret model of CF has been established sharing many characteristics with clinical pancreatic pathology in CF.17,19-22 In neonatal CF ferrets, mild pathology within the pancreas with little indication of inflammation and fibrosis has been reported. 17
Furthermore, CF and non-CF ferret models of different clinical age groups were used to prospectively characterise the progression of early CF-related pancreatic pathologies, which have been difficult to elucidate in man due to highly limited tissue availability. 17 Pancreas histology in CF ferrets aged 3 to 4 weeks showed mild inflammatory cell infiltration with mild to moderate acinar ectasia. 17 In new born CF ferrets increased apoptosis of acinar and pancreatic duct cells was observed, demonstrating the presence of an early exocrine destruction, which may impact on endocrine pancreas morphology and has been proposed to lead to islet mass reduction in neonatal CF. 19
With increasing age, CF ferrets develop a significant loss of pancreatic exocrine tissue with an expanded ductal cross-sectional area and increased fibrotic and adipose tissue replacing the parenchyma, compared to wild-type (WT) ferrets.17,20 The increase in fat area suggests the initiation of an adipogenic program during the inflammatory phase of exocrine injury. 17
In CF ferrets aged 1 to 2 and 2 to 4 months activated stellate cells not seen in WT ferrets were detected surrounding the ductal epithelium. 17 In pathologic states, pancreatic stellate cells (PaSCs) transdifferentiate into activated proliferating myofibroblasts, losing fat droplets that are stored in their quiescent state. 23 Mouse lineage-tracing studies have shown that myofibroblasts can in turn transdifferentiate into adipocytes. 24 Myofibroblasts identified by α-smooth muscle actin (α-SMA) staining in fibrotic regions of ferret pancreas may be a possible source of adipocytes in CF. 17 Quiescent PaSCs which contribute to cell structure and parenchymal function via maintaining normal basement membrane, are postulated to be the source of myofibroblasts during CF pancreatic remodelling.17,25 It has previously been reported that myofibroblasts play a significant pathological role in pancreatic fibrosis associated with pancreas cancer, which may contribute to the progression of β-cell dysfunction. 26
Impact of CF on the Endocrine Pancreas
Morphological changes in the CF endocrine pancreas
Islet morphology in CF
CF not only leads to changes in exocrine morphology but also impacts on the endocrine pancreas compartment within the islets of Langerhans. Islets are micro-organs dispersed throughout the pancreas and composed of different endocrine cell types secreting hormones maintaining blood glucose homeostasis.
Animal studies have facilitated understanding the impact of reduced CFTR function on islet morphology and mass in different models including mice and ferrets. Systemic short-term chemical inhibition of CFTR in healthy mice led to a significant reduction in islet size, but comparable islet cell numbers to control animals. 27 This was also observed in a F508del mouse model with permanent CFTR impairment. 28
Changes in numbers and proportions of individual islet endocrine cell types have been observed in a CF ferret model which appears to mirror the different stages of clinical early disease development and progression including the dynamic changes in islet hormone expression and islet morphology.17,19-22 In new-born CF ferrets, β-cell mass as well as mRNA expression of islet hormones, analysed from bulk RNA, was comparable to ferrets without CF.17,22 However, significantly more small islets and fewer large islets were present in comparison to non-CF pancreata. 19 From the age of 9 days until 4 weeks, incrementally decreasing β-cell mass could be observed accompanied by a decrease in islet hormone mRNA transcripts.17,19,22 In 1 to 2 month old CF ferrets, which appear comparable to 1 to 2 year old children with CF, a significant increase in inflammatory cell infiltration as well as proinflammatory factors, and fibrosis was seen in the exocrine pancreas, in parallel with a marked disorganisation and restructuring of islets in the endocrine compartment.19,22 However, β-cell mass was comparable to 3 to 4-week-old ferrets remaining significantly decreased compared to non-CF counterparts.17,22 Gene expression of islet hormones was comparable to WT ferrets with the exception of pancreatic polypeptide (PP) showing significantly increased levels compared to non-CF ferrets. 22 In addition, 2 to 4 month old CF ferrets demonstrated a decline in inflammation but still significant fibrosis in the exocrine pancreas, whereas islet-like larger clusters of insulin-positive cells aggregated in the endocrine pancreas. 22 This was accompanied by increased (relative) β-cell mass and abundance of islet hormones as well as hormonal gene expression.17,22 Relative proportions of individual endocrine cell types change dynamically through the stages of disease progression, but average proportions remain largely comparable between WT and CF ferrets up to the age of 4 months with 60% to 80% insulin-positive, 10% to 20% glucagon-positive and 20% to 40% somatostatin-positive cells. 17 However, it has to be noted, that individual CF islets may display a more polarised endocrine cell type composition depending on islet size and disease stage.17,19 For example, small islets in 3 to 4 week old CF ferrets were comprised predominantly of β-cells, whereas bigger islets consisted primarily of α-cells. 17
Analysis of human post-mortem pancreata showed that CF can lead to substantial changes in pancreas morphology, as discussed previously. However, even in severely altered exocrine tissue, islets were still present and usually occurring in clusters.2,16,18 In tissue from younger patients showing low grade exocrine changes, the number of islet cells was comparable to age-matched controls, but increased exocrine damage and collagen deposits in the islet microenvironment impacted on islet morphology and number.18,29 The volume density of endocrine tissue as well as the number of endocrine cells/section was believed to be within a normal range in CF patients between the age of 5 and 48 years.16,30 In addition, endocrine cell mass was observed to be comparable in the first 6 months between CF and normal tissue, but the number of islets decreased through clustering and aggregation with length and severity of the disease.15,18
Despite little impact on endocrine volume, islet cell composition seemed to be affected in different clinical CF cohorts, with a decrease in β-cell mass compared to control pancreata.16,18,30-32 In addition, an increase in α-cells and δ-cells could be observed in young and adult CF pancreata, whereas a significant increase of PP-cells predominantly was observed in younger cohorts.15,16,18,33 Two studies reported a comparable β-cell area between CF and non-CF pancreata, with 1 study showing no changes in α-, δ- and PP-cell-mass whereas a further study reported an increase in these non-β-cell islet hormones.15,33 Overall, Löhr et al 16 identified no difference in islet cell composition between pancreata with the fibrotic or lipoatrophic exocrine disease pattern. Interestingly, patients with CF displayed a significantly increased number of chromogranin A positive, but endocrine hormone-negative cells in islets, clustered and single cells, compared to non-CF pancreata indicating possible regeneration. 30
In addition to changes in hormone composition, intra-islet fibrosis and perisinusoidal sclerosis could be observed and it was reported that islets appeared increasingly fragmented with accumulation of intra-insular fibrosis.16,29 Furthermore, islet vascularisation may be impacted through the marked changes in the islet microenvironment in later disease stages although no reduction in vascularisation was observed in a new-born CF pig model.2,34
Islet morphology in CFRD
With the progression of CF, secondary complications including CFRD can occur where islets are exposed to exocrine stressors but also to prolonged hyperglycaemia potentially further impairing islet morphology and function.
Two single cases of CFRD showed no further reduction of β-cell mass compared to the CF pancreata without diagnosed diabetes in 1 case series. 16 Other studies reported that CF patients with diabetes had significantly reduced islet number with altered morphology and loss of β-cells (significant in some studies) compared to pancreata from patients without diabetes with and without CF.15,16,18,29-33 This was accompanied by a significant increase in α-cell and δ-cell area compared to non-CF pancreata, but not different to non-diabetic CF tissue.18,33 Another cohort showed comparable glucagon- and significantly increased somatostatin-positive area in CFRD pancreata compared to controls. 31 Interestingly, a significant decrease of PP-cell area compared to non-diabetic CF cases could be observed in CFRD pancreata in 1 study, but not others.15,18,33
However, a significantly increased number of chromogranin A positive/islet hormone-negative cells, as well as polyhormonal cells were found in 3 cases of CFRD, possibly indicating attempted neogenesis or loss of end-differentiated endocrine cell identity. 30
In addition to changes in cell composition, islets in CFRD displayed amyloid deposition which was observed in 38% to 69% of CFRD pancreata with simultaneous apoptotic islet cells.18,29,33 In a study by Couce et al 29 amyloid deposition was also observed in 17% of pre-diabetic individuals with elevated HbA1c and some periods of insulin treatment. Electron microscopy studies of 2 single CFRD cases revealed the presence of amyloid fibrils in the cytoplasm as well as in the direct microenvironment of β-cells. 29 Apoptosis of β-cells was seen in 2 of 3 CF pancreata with amyloid formation indicating increased β-cell loss in the presence of islet amyloidosis. 18 Amyloid deposition is known to impact islets in type 2 diabetes with reduced β-cell area and increased β-cell apoptosis indicating the possible negative impact of amyloidosis on islets in CFRD. 35
Impact of CF on endocrine cell clusters and single endocrine cells
Clusters of a few endocrine cells and single hormone-positive cells can be observed throughout the pancreas with these being significantly more often chromogranin A positive and hormone negative or displaying poly-hormonal expression in CF and CFRD pancreata. 30 Furthermore, endocrine cells are present in ductal or acinar epithelium of CF pancreata displaying more variable hormone composition than normal islets with increased abundance of PP in young CF patients. 15 Löhr et al 16 observed endocrine cells budding off from the ductal epithelium in some human pancreata with the fibrotic pattern, which was also observed by Rotti et al 17 in 1 to 2 months old CF ferrets, with insulin-positive clusters appearing to be extension of the ductal epithelium with some cells displaying neogenesis. These findings indicate a possible pathway for neogenesis in adult and diseased pancreata which has been proposed in previous studies and which could be helpful in attempted preservation of β-cell function in patients with CFRD.36,37
Impact of CF on endocrine function
Inhibition studies in mice showed that short term impairment of CFTR function impacted on islet size, but not glucose tolerance or insulin secretion suggesting that longer term impairment of CFTR is necessary for an impact on β-cell function. 27
Studies in people with CF have shown that β-cell dysfunction can be present before the diagnosis of diabetes and that the abnormal glucose tolerance in CF can largely be attributed to impaired insulin secretion. 2 In patients with pancreas exocrine insufficient CF who had a normal glucose tolerance, metabolic studies have revealed decreased insulin and increased proinsulin secretion indicating impaired early-phase insulin secretion as well as changes in overall functional β-cell mass. 38
Knock-out animal models mirror the impact of CF on insulin secretion in response to glucose challenge in vivo and in vitro as exemplified in the CF ferret.19,21,22 Changing blood glucose levels during disease progression in ferrets correlated with changes in β-cell mass. 22 Decreased insulin secretion and significantly impaired first-phase insulin response during meal tolerance tests has been demonstrated.19,22 In addition, isolated islets displayed increased basal insulin secretion but diminished glucose-stimulated insulin secretion with elevated intracellular Ca2+ due to increased K-ATP inhibition. 21
Analysis of isolated islets from 5 human CF pancreata showed comparable insulin and glucagon secretion to control islets. 39 Interestingly, the gene expression of key expression markers from these islets was also comparable to control islets indicating maintained islet cell identity despite CF. 39 However, this is clearly a small study and the isolation process may itself impact islet function potentially masking the in vivo impact of CF.
Potential Signals Impacting on Endocrine Function
CFTR
CFTR is highly expressed on the apical membrane of epithelial cells in the pancreatic ductal epithelia, where it functions as a cAMP-regulated anion channel. 40 CFTR facilitates the transport of anions (chloride and bicarbonate) and water into the ductal lumen; resulting in an increased volume of alkaline fluids which maintains a soluble environment for the highly concentrated proteins that are secreted by the acinar cells of the exocrine pancreas.41-43 CF patients with a loss or a decline in CFTR function have been reported to have an obstructed and damaged lumen due to a low flow of secretions and protein precipitation.43,44 CFTR mutations are grouped into 5 classes with increasing severity and mutations of classes I to IV lead to exocrine pancreatic insufficiency due to a substantial loss of acinar cells within the pancreas resulting in reduced production of digestive enzymes.43,45-47
In the human pancreas CFTR expression is mainly observed in duct cells.21,39,48 The detection of CFTR protein expression in islets has proven difficult in human samples with some studies reporting no protein expression in human endocrine cells.21,39,48 However, other studies have reported expression of CFTR protein and mRNA in human tissue at low levels and in a small number of endocrine cells, as well as in single human β-cells and isolated islets.49,50
CFTR expression is observed in mouse islets, rodent primary β-cells and β-cell lines, where it has been shown to impact on electrophysiological responses and insulin secretion.28,49-53 A negative impact of CFTR loss of function on glucose-stimulated insulin secretion has been demonstrated in a rodent β-cell line alongside an increase in oxidative stress. 53 More detailed studies showed that the inhibition of CFTR impacted on priming and exocytosis of insulin granules in human and mouse isolated islets and additionally, a F508del mouse model showed that defective CFTR can impact on the processing and maturation of insulin.28,50 Further to the effect on insulin secretion, an impact of the impairment of CFTR function on glucagon and somatostatin secretion could be observed in a F508del mouse model. 28
In isolated ferret and human islets as well as in ferret pancreas in situ, single-molecule fluorescent in situ hybridisation demonstrated that CFTR mRNA was colocalised within Keratin-7 (KRT7)+ ductal cells not endocrine cells.21,48 It was thus suggested that paracrine mechanisms, involving pro-inflammatory signals secreted from islet-associated exocrine-derived cell types, mediate CFTR mutation effects on β-cell function in CFRD. 21 CFTR modulators including ivacaftor, lumacaftor, tezacaftor and elexacaftor which correct or potentiate CFTR channel function can be a treatment for some CFTR mutations.54-56 It was shown that ivacaftor alone or in combination with other modulators improved clinical outcomes including pulmonary function. 56 A study with the modulator combination lumacaftor and ivacaftor showed no improvement on glucose tolerance and insulin secretion after 6 to 8 weeks of treatment. 54 Whereas other studies and case reports observed an improvement in insulin secretion and resolving of CFRD in some people with CF through the treatment with modulators for 4 weeks till 12 months.55,57-60 This shows the potential benefit of modulator treatment on the pancreas. However, bigger cohorts and longer studies are needed to fully understand the impact of CFTR on the pancreas and islet tissue.54,55,57-60 Prolonged studies with younger cohorts might clarify if modulators and therefore improved CFTR function, can prevent or delay the onset of CFRD. 58 Taken together, these studies suggest that CFTR might impact on β-cell function via extrinsic paracrine signalling with or without an intrinsic role within islet endocrine cells.
Immune cell infiltration
Significant islet infiltration with immune cells has been observed in young human CF tissue with a number of different leukocytes including monocytes, macrophages and lymphocytes present. 18 In adult CF and CFRD tissue a decreased number of immune cells could be observed in islets compared to young pancreata.18,39 These immune cells comprised lymphocytes with monocytes and macrophages only found in the exocrine compartment.18,39 Islets in CF tissue with a fibrotic pattern showed significantly increased immune infiltration compared to controls. 18 The presence of inflammatory cells in the islet microenvironment in CF may negatively impact β-cell function through immune mechanisms comparable to those in type 1 and type 2 diabetes.61,62 CFRD is predominantly occurring in people with prolonged CF history and increased exocrine changes including fibrosis and inflammation.10,11,17 Immune cell infiltration in CF pancreata was increased in islets compared to exocrine tissue, but in general higher than in controls.18,39 The increased immune cell infiltration in younger patients might indicate an impact of inflammatory factors in the earlier development of islets. 18 Prolonged presence of immune cells in islets and in the exocrine pancreas could pose continuous stress towards islets and in combination with impaired CFTR function as well as islet remodelling, increasing islet dysfunction.18,39
Proinflammatory signals
In CF ferrets, during glycaemic crisis (metabolic imbalance associated with uncontrolled diabetes mellitus) inflammatory markers including interleukin (IL)-8, IL-1β, IL-6 and C-X-C motif chemokine ligand 10 (CXCL10) were increased in the pancreas and have been previously reported to be associated with acute pancreatitis.22,63-65 A study by Sun et al 21 used a CFTR knock out model of neonatal ferret islets and CFTR inhibitors to show that CFTR-expressing exocrine-derived cells appear to affect β-cell function by secreting pro-inflammatory factors such as IL-6. IL-6 production was increased in islets from CF ferrets and this was associated with a decline in islet insulin content and an elevated percentage of insulin content secreted at basal glucose levels, when compared with WT islets. 21
In addition, desmin-positive PaSCs expressing α-SMA demonstrated a proinflammatory state in CF relative to the WT pancreas. 21 Activated PaSCs synthesise and secrete extracellular matrix (ECM) proteins including collagen type I and III, fibronectin, pro-inflammatory cytokines and growth factors.66-70 Moreover, it has been reported that PaSC activation is induced by cytokines and growth factors produced by acinar cells, inflammatory cells, platelets and endothelial cells during pancreatic injury or inflammation. 71 In vitro studies have shown that transforming growth factor-β (TFG-β), tumour necrosis factor-α (TNF-α), IL-1, IL-6 and activin A, ethanol and its metabolites, oxidative stress and physical pressure can activate PaSCs.71-75
Gene expression analysis in CF ferret islets showed increased expression of PaSC activation, inflammation, fibrosis and remodelling markers including CXCL10, ACTA1, ACTA2, and TGF-β1. 21 It was suggested, due to evidence for PaSC activation of CF islets when isolated from the pancreas, the presence of the proinflammatory environment in culture and its influence on IL-6 secretion levels and other possible cytokines, that these pathways play a major role in CF islet dysfunction. 21
CFRD shares common features with both type 1 and type 2 diabetes that can include pancreatic exocrine insufficiency, multi-organ dysfunction, chronic infection, malabsorption, malnutrition and varying degrees of insulin resistance.29,76-80 Recently, increased β-cell microRNA-29 has been associated with the development of type 2 diabetes in the elderly whereby metabolic stress promotes islet inflammation by recruiting monocytes and macrophages via the miRNA-29/TRAF3/Cxcl10 pathway in β-cells, suggesting a possible pathway of islet dysfunction in CF pancreata. 81
Peri-islet and intra-islet immune infiltration is certainly seen in the CF pancreas as discussed previously. 39 Moreover, in vivo β-cell function is impaired by adaptive immune response inflammatory cytokines interferon gamma (IFN-γ) and TNF-α.39,82 Stimulated T-cells isolated from CF islets have been shown to secrete cytokines/chemokines including IL-6, IL-1β, CXCL10, TNF-α and IFN-γ. 39 Hence, it has been concluded that islet isolation from CF pancreas removes the islets from the inflammatory environment, thus reducing the effect of cytokines/chemokines, enabling a moderately normal islet secretion of insulin and glucagon in vitro. 39 Further, Hull et al 33 found cytokine IL-1β present in islets of paediatric CF (>10 years), adolescent CF and adult CFRD cases. These inflammatory cytokines can negatively impact on β-cell function and possibly lead to β-cell loss through apoptosis or dedifferentiation.39,82-87 In addition it is known that β-cells are able to secrete chemokines leading to recruitment of immune cells.21,84 Moreover, cytokine-mediated signalling in islets and the surrounding environment is increasingly believed to be important in the regulation of pancreatic β-cell function in various conditions including obesity, metabolic syndrome and type 2 diabetes.82,88-95
Summary
CF is associated with significant changes in the microenvironment of pancreatic islets which may play a major role in driving development of CFRD (Figure 1). These include replacement of acinar parenchyma with fibrotic or fatty tissue. These changes are associated with a proinflammatory environment with potential inflammatory cell infiltration of the islets and secretion of proinflammatory cytokines and chemokines. Together this hostile environment appears to mediate impaired β-cell function beyond any impact on absolute β-cell mass. Further research specifically targeting these pathways may lead to novel therapeutic options to maintain and restore normal insulin secretion preventing the considerable psychosocial and biomedical burden of CFRD.
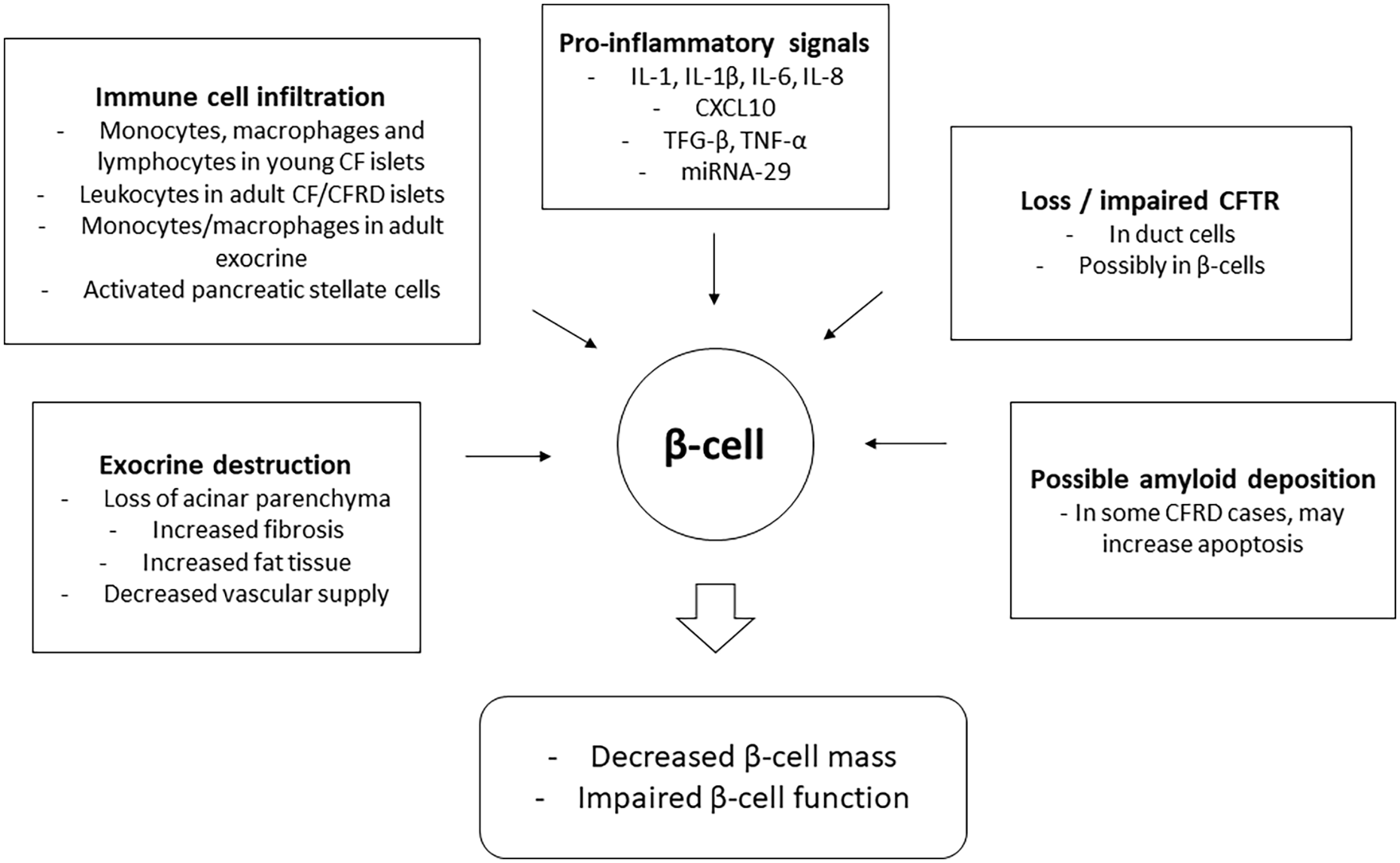
Possible stressors impacting on β-cell function in CF and CFRD pancreata.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are supported by the Cystic Fibrosis Trust (SRC 019).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors conceptualized, wrote and critically revised the manuscript. All authors approved the final version.
