Abstract
Cushing’s syndrome causes increased morbidity and mortality due to cardiovascular and infectious diseases. Exogenous Cushing’s syndrome can render the adrenal glands unable to cope with severe infections and may result in Addisonian crisis, which can be fatal if not properly diagnosed and treated. During hospitalization for disease exacerbation, a man on chronic glucocorticoid therapy for Crohn’s disease and Cushingoid features developed polymicrobial septic shock together with hypotension that was unresponsive to fluids. On suspicion of relative adrenal insufficiency (cortisol levels were “inadequately” normal), intravenous hydrocortisone was started; norepinephrine was also required to normalize blood pressure. Following clinical improvement, oral cortisone acetate was started. On discharge, he was instructed on how to manage stressful events by increasing oral glucocorticoid treatment or starting a parenteral formulation, if required. Chronic glucocorticoid therapy can cause severe side-effects; in addition, hypoadrenalism can occur in critical illnesses (eg, severe infections). Prompt recognition and proper therapy of this condition can be life-saving.
Introduction
Cushing’s syndrome is a rare endocrine disease characterized by chronic hypercortisolism, and causes increased morbidity and mortality; death may be due to cardiovascular disease, infectious disease, or sepsis. 1 Infectious diseases can be polymicrobial, and fungal infections are very common. Exogenous Cushing’s syndrome is due to excessive or long-term exposure to glucocorticoids, mostly in oral or parenteral form. This may lead to partial/complete suppression of the hypothalamic-pituitary-adrenal (HPA) axis and, in turn, to adrenal insufficiency in conditions such as critical illnesses in which an increase in cortisol output is required; in such situations, prompt intravenous administration of hydrocortisone can be life-saving. In patients on chronic glucocorticoid therapy, baseline cortisol levels may help to foresee the risk of adrenal insufficiency. 2
Here, we describe the case of a middle-aged male patient with exogenous Cushing’s syndrome due to long-standing glucocorticoid therapy for Crohn’s disease who developed respiratory/renal failure and (relative) adrenal insufficiency during polymicrobial septic shock.
Case Report
A 51-year-old man was admitted to the emergency department for abdominal pain, diarrhea, and worsening back pain. History revealed long-standing (8 years) Crohn’s disease on therapy with prednisone 25 mg/day, hypertension treated with olmesartan 20 mg/amlodipine 5 mg, chronic obstructive pulmonary disease, obesity, and depression treated with paroxetine. The patient was not on therapy with bisphosphonates or other glucocorticoid-induced osteoporosis prevention treatments. One month earlier, he had suffered a dorsal trauma, and X-ray had revealed a T11 fracture, for which a back brace was prescribed. The patient showed typical Cushingoid features (obesity, moon face, dorsocervical hump, striae rubrae). Increased blood pressure (BP) (150/100 mm Hg), normal oxygen saturation (98%), increased heart rate (113 bpm), and fever (38°C) were recorded. Lung auscultation revealed mild bronchostenosis; the abdomen was diffusely painful, without peritonism. Blood tests revealed leucocytosis: 14,840/mm3 (n.v. 4200-9300) (neutrophil count: 90.6%), microcytic anemia (Hb: 10.8 g/dL, n.v. 14.2-17.2), thrombocytosis: 550,000/mm3 (n.v. 155,000-320,000), and mildly increased C-reactive protein (CRP): 2.2 mg/dL (n.v. 0-0.5). Nasopharyngeal swab for SARS-CoV2 proved negative. Contrast-enhanced CT showed parietal thickening of the descending colon, sigma, and rectum, due to hyperemia of the mucosa and submucosa (Crohn’s disease). T7, T10, and T11 fractures were also observed.
The patient was admitted to the gastroenterology ward, and therapy with intravenous (i.v.) saline, metronidazole 500 mg × 3, levofloxacin 500 mg, methylprednisolone 40 mg, pantoprazole 40 mg × 2, and mesalazine 4800 mg/day per os was started. The fever subsided in 1 day, but BP further increased (systolic: 160-190 mm Hg, diastolic: 110-130 mm Hg); for this reason methylprednisolone dosage was halved, and the dose of sartan/calcium channel blocker was doubled, and nebivolol and hydrochlorothiazide were added with decrease of BP. Magnetic resonance imaging confirmed the fractures of T7, T10, and T11, the former being recent, as evidenced by hyperintensity on signal short time sequence (Figure 1).
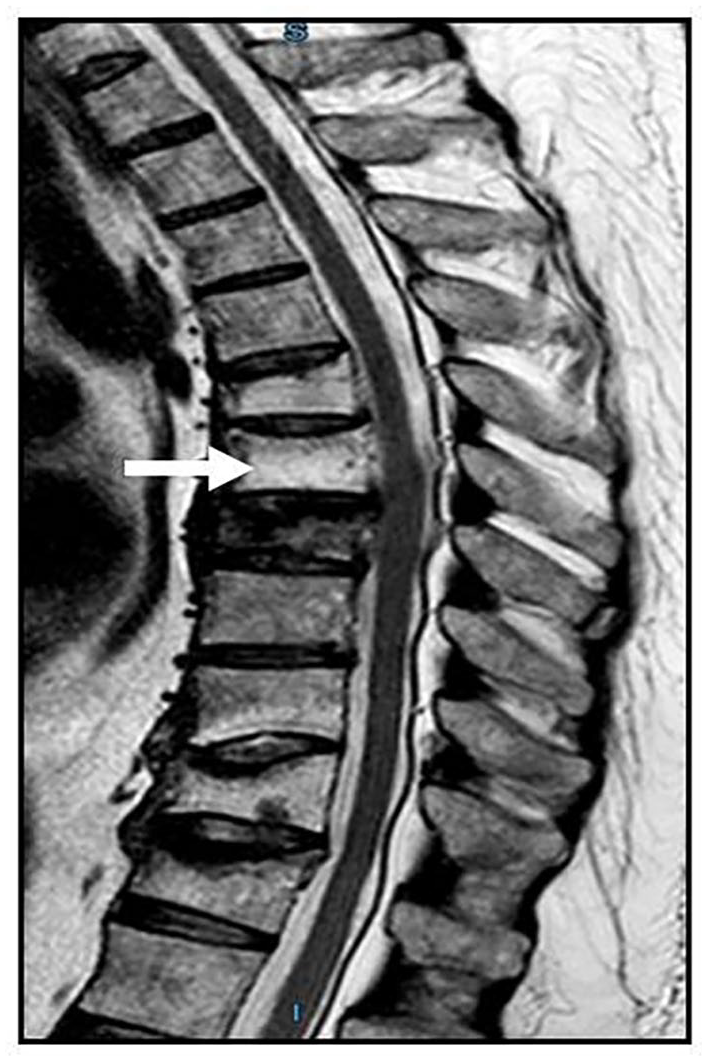
Dorsal magnetic resonance imaging showing vertebral fractures of T7, T10, and T11. The T7 body is reduced in height, with anterior wedge deformation and irregularities mostly of the upper somatic part, resulting from the fracture, which extends posteriorly up to the peduncle isthmus and to the postero-superior edge, which partially protrudes into the endocanalar space. Hyperintensity on signal short time sequence is due to edema from a recent trabecular microfracture (arrow). The T10 and, to a lesser extent, T11 bodies are reduced in height, with anterior wedge deformation.
Three days after reduction of glucocorticoid dose, patient’s health status worsened: hypotension (80/60 mm Hg) and respiratory failure occurred, and fever (38°C) relapsed and blood and urine cultures were collected (negative). Antihypertensive therapies were stopped. Hemogasanalysis showed hypoxemia (PO2: 54.4 mm Hg) with hypocapnia (PCO2: 31.5 mm Hg), normal pH: 7.43, and slightly reduced HCO3−: 21 (compensated respiratory alkalosis) with reduced P/F: 194, and oxygen therapy was started. Leucocyte count: 24,640/mm3 (neutrophil count: 77.8%) and CRP levels: 16.6 mg/dL further increased, and acute renal failure (creatinine: 2.2 mg/dL; n.v. 0.7-1.2), occurred. The patient was transferred to the semi-intensive unit, where he became soporous but arousable and septic shock was diagnosed. Colloid and crystalloid were infused, with scant benefit, and antibiotics were up-graded, with i.v. piperacillin/tazobactam and linezolid. Chest X-ray and echocardiogram proved unremarkable.
Medical history was reviewed and, given patient’s long-standing chronic therapy with glucocorticoids and the fact that i.v. methylprednisolone dosage had been reduced for grade 3 hypertension during hospitalization, a superimposed Addisonian crisis was suspected. Morning cortisol levels were evaluated and proved “normal”: 8.0 to 13.3 µg/dL (n.v. 6.2-19.4, 7-10 a.m., electrochemiluminescence immunoassay) but were deemed likely overestimated by cross-reactivity of the cortisol assay by methylprednisolone, and inadequate given the ongoing critical illness.
ACTH levels were in the lower normal range: 11.7 pg/mL (n.v. 9-52). Given the severe clinical condition, no Synacthen test to evaluate the cortisol response was done. A midline catheter was inserted and i.v. hydrocortisone (100 mg t.i.d.) was started; the patient became more alert, but BP increased only partially and norepinephrine infusion (0.1 µg/Kg/min) was started. As hemogasanalysis showed respiratory and metabolic acidosis (pH: 7.2), Continuous Positive Airway Pressure (C-PAP) Bilevel ventilation was started, and sodium bicarbonate was infused. As hemoglobin levels had dropped to 7.9 g/dL in absence of macroscopic bleeding, 2 units of packed red blood cells were transfused; hypokalemia: (2.7 mEq/L) occurred and potassium infusion was started with normalization of serum levels.
Since fever persisted, new blood (from midline catheter and peripheral blood), and urine cultures were performed, and β-
The clinical condition improved over a few days: fever subsided, blood pressure and creatinine normalized, and CRP levels dropped to 1.7 mg/dL. Noradrenaline, hydrocortisone, and oxygen were withdrawn; cortisone acetate (25 mg b.i.d. per os) was started. Subsequently, antihypertensive therapy was restarted. Antifungal therapy continued for 14 days after negativization of blood cultures. Early morning cortisol levels were re-checked (before cortisone acetate administration) and were clearly reduced: 3.1 µg/dL, as were ACTH levels: 3 pg/mL. The patient refused a Synacthen test and was eventually discharged in satisfactory clinical condition; gastroenterological follow-up was scheduled. He was instructed to increase oral corticosteroid therapy in stressful events or start parenteral formulations, whenever required.
Discussion
Iatrogenic Cushing’s syndrome is due to prolonged exposure to high doses of glucocorticoids, mostly in oral or parenteral form. Typical features are central obesity, dorsocervical hump, moon face, abdominal striae, proximal myopathy, bone loss, glucose intolerance, hypertension, depression, and psychosis.3,4
Since glucocorticoids stimulate osteoclast activity and inhibit osteoblast action, osteoporosis is frequent (22%-57%) in these patients, especially in males; the risk of low-energy fracture increases from the first 3 months of therapy onwards.1,3
In our patient, low-energy dorsal vertebral fractures occurred after several years of glucocorticoid therapy and were managed conservatively.
As patients with chronic (exogenous and endogenous) hypercortisolism are prone to infectious diseases and sepsis, they display increased morbidity and mortality. This finding is related to the duration and severity of hypercortisolism.1,5
Glucocorticoids affect both the cellular and humoral components of the immune system. With regard to the former component, they impair neutrophil function, macrophage maturation, and natural killer action. The humoral component is also affected, since glucocorticoids impair lymphocyte proliferation and action (reduced Th1-helper action and enhanced Th2 responses) and the maturation and action of macrophages, and reduce pro-inflammatory cytokines.1,5
Patients are prone to infection by virtually all pathogens (bacteria, viruses and fungi). Fungal sepsis due to Candida, Aspergillus, and Criptococcus is frequent and is related to the direct effect of glucocorticoids on fungal proliferation and translocation from the gastrointestinal tract. 5
Our patient with iatrogenic Cushing’s syndrome developed polymicrobial (gram-positive bacteria and fungi) severe septic shock during hospitalization, for which tailored antibiotic therapy was started.
Severe hypotension unresponsive to fluids occurred and once the patient was transferred to semi-intensive unit, superimposed adrenal insufficiency was suspected as he had been taking glucocorticoids for many years for Crohn’s disease. Blood samples for cortisol assay were collected, and i.v. hydrocortisone was administered with clinical improvement.
The clinical suspicion of adrenal failure during critical illness starts when hypotension refractory to fluids, unexplained coma, hyponatremia, or hyperkalemia occur. 6
A recent meta-analysis of patients taking corticosteroids chronically showed that the percentages of patients with adrenal insufficiency ranged from 4.2% for nasal administration to 49% for oral administration, and peaked at 52.2% for intra-articular administration. The risk also varied according to the dose, from 2.4% for low-dose administration to 21.5% for high-dose administration, and according to treatment duration: from 1.4% (1 month) to 27.4% (1 year or more). 7
Chronic glucocorticoid use is considered the most common cause of adrenal insufficiency; it inhibits both hypothalamic CRH and pituitary ACTH release, which leads to atrophy of the adrenal cortex. This is called tertiary adrenal insufficiency. 2
It is well known that sudden cessation or too rapid withdrawal of chronic glucocorticoid therapy can lead to a catastrophic adrenal crisis. In our patient, the oral long-term glucocorticoid therapy, which had been replaced by an i.v. formulation (methylprednisolone) on hospital admission, was erroneously and abruptly reduced owing to the occurrence of grade 3 hypertension.
This therapeutic mismanagement occurred in a non endocrinological setting and before the patient was transferred to the semi-intensive unit for acute adrenal insufficiency triggered by septic shock. This fact underlines the need for patients on chronic glucocorticoid therapy at a dosage able to interfere with HPA axis to be evaluated by an endocrinologist whenever sepsis or severe infections/diseases occur, in order to increase properly the glucocorticoid dosage.
Another remarkable item for the clinicians is the scant awareness of the fact that critical illnesses in these patients can elicit absolute or “relative” adrenal failure. The latest is called critical illness-related corticosteroid insufficiency (CIRCI) and is defined as the incapacity of the adrenal gland to release enough cortisol to cope with a severe illness. 8
Since any point in the HPA dysfunction can be affected, the current diagnostic tools available (basal cortisol levels or peak/delta of cortisol response to Synacthen test) are inadequate to identify patients with CIRCI. 8
Overall, clinical data reveal that up to 20% of critically ill patients show hypofunction of the HPA axis; in septic shock, this percentage can reach 60%. The pathogenesis can be multifactorial, ranging from drugs to renal failure, and disseminated intravascular coagulation. 8
Assaying morning cortisol may help predict the presence or absence of adrenal insufficiency. According to the Endocrine Society, cortisol levels <3 μg/dL at 8 to 9 a.m. are indicative of adrenal insufficiency, while levels >15 μg/dL exclude this diagnosis. Dynamic stimulatory testing (Synacthen test) is suggested for values between 3 and 15 μg/dL, and a normal cortisol response is set at >18 μg/dL.2,9
In critically ill patients, when serum binding proteins (albumin, transcortin) are near normal or normal, serum cortisol levels ⩾15 µg/dL are a reliable marker of normal adrenal function. In hypoproteinemic critically ill subjects, a random (adequate) serum total cortisol level is expected to be ⩾9.5 µg/dL. 10
Our patient, who had been on and off therapy with prednisone for 8 years for Crohn’s disease (continuously at a dosage 25 mg/day over the last year), had the features of Cushing’s syndrome. Worsening of his clinical condition (hypotension, fever, and desaturation) during hospitalization was deemed as the combination of septic shock and adrenal insufficiency. The latest was related to both incorrect reduction of i.v. glucocorticoid dosage made a few days earlier in another department for uncontrolled hypertension and CIRCI secondary to septic shock, which further increased glucocorticoid requirement. As regard the first item, initial methylprednisolone dosage (40 mg/day i.v.), which is equivalent to 200 mg hydrocortisone, approached the “stress” glucocorticoid replacement dose; however, once the dosage was erroneously halved, it became clearly insufficient.
In our patient, it is noteworthy to underline that cortisol levels, which proved within the normal range (8-13 µg/dL), were evaluated by standard immunoassay, which shows cross-reactivity up to 10% with methylprednisolone. This methodological bias may have falsely “elevated” the cortisol values with subsequent underestimation of the risk for adrenal insufficiency. This crucial methodological point had been stigmatized in a previous report, showing that several compounds which produce cross-reactivity in steroid hormone immunoassays (included that for cortisol) generally have a high degree of structural similarity to the target hormone. In particular, methylprednisolone has high cross-reactivity (⩾5%) for the cortisol assay, and is expected to produce substantial apparent cortisol levels at serum/plasma concentrations typical in patients administered this drug, with subsequent high likelihood of clinically important effects. 11
Besides being overestimated, in our patient cortisol levels were even inadequate in the setting of critical illness and CIRCI due to septic shock 12 superimposed. As regards this finding, previous report showed mean cortisol levels of 32 μg/dL during septic shock. 13
On suspicion of adrenal insufficiency, hydrocortisone was administered i.v., with a partial hemodynamic response; however, norepinephrine was required in order to support the cardiovascular system. After improvement in the patient’s clinical condition, cortisone acetate was started, and was maintained on discharge. The patient was carefully instructed on how to increase glucocorticoid therapy during stressful events or to switch to a parenteral formulation when required.
In summary, chronic glucocorticoid therapy can severely affects patients’ health as a result of hypercortisolism. In addition, by suppressing the HPA axis, it can put patients at risk of adrenal insufficiency mostly in the event of severe/critical diseases. In these situations, glucocorticoid replacement therapy must be increased.
Since adrenal failure is a potentially fatal condition, prompt diagnosis and therapy are mandatory.
Footnotes
Funding:
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contribution
Dr. L. Foppiani conceived the study, analyzed the data, and wrote the manuscript.
Ethical Approval
At the author’s institution, case reports or case series are deemed not to constitute research, and ethics approval is not required.
Informed Consent
Informed consent for publication of this manuscript was obtained orally from the patient at his bedside prior to discharge from hospital. It was clearly explained to the patient that his past medical history and the history of his hospital stay might be the subject of a case report written by his doctor. The patient fully agreed about the case report, and was instructed in broad terms on both the structure of the scientific report and the writing modality. However, at that time, informed consent was obtained orally because, although the author intended to write a case report, he did not know exactly when he would do so.
