Abstract
Background:
Upper and lower respiratory tract infections are common conditions for which medical advice is sought, and their management relies on the use of prescription and over-the-counter (OTC) medicines. Ambroxol, bromhexine, carbocysteine, erdosteine, N-acetyl cysteine (NAC), and sobrerol are mucoactive agents for which clinical trials have been conducted, have been awarded well-established status by regulatory authorities, and are available as OTC or prescription products.
Objective:
To briefly review the evidence-based efficacy and safety of these substances in the therapy of upper respiratory airways infections.
Methods:
We conducted searches in MEDLINE and other databases for clinical trials and reviews done on the efficacy and safety of ambroxol, bromhexine, carbocysteine, erdosteine, NAC, and sobrerol.
Results:
Clinical trials have shown that these mucolytics have an important place in the relief of cough symptoms by easing the elimination of mucus. All drugs have shown comparable efficacy in the symptomatic treatment of productive cough, with some shared characteristics and some specific features.
Conclusions and relevance:
All mucolytics reviewed have a good safety profile, although some precautions should be taken when using ambroxol and bromhexine, and the use of NAC and carbocysteine should be monitored in special patient groups. Overall, however, the available evidence from randomised, controlled, and observational trials, as well as pragmatic, real-life experience, suggests that these products are useful in the therapy of upper respiratory airways infections, including bronchitis, sinusitis, and rhinosinusitis.
Background
Respiratory tract diseases affect a large number of people worldwide: in the European Union (EU), for instance, 7% of hospital admissions are linked to respiratory illnesses, which are responsible for approximately 12% of all-cause deaths. 1
Upper and lower respiratory tract infections (RTIs) are common conditions for which medical advice is regularly sought, and their management relies on the use of prescription and over-the-counter (OTC) medicines. 2 Economically, RTI treatment is linked to significant expenses: in 2011, chronic obstructive pulmonary disease (COPD) and asthma, in the EU, caused €42.8 billion in direct (primary care, hospital outpatient and inpatient care, drugs, and oxygen) costs; indirect costs (lost production including work absence and early retirement) amounted to €39.5 billion. 3
Mucoactive drugs are regularly used as a therapeutic option for mucus alterations, including hypersecretion. Mucus-thinning (mucolytics), cough-inducing (expectorants), and cough transport facilitating (mucokinetics) drugs have been available for clinical use to ease airway clearance in diverse indications such as bronchiectasis, COPD, acute, and chronic bronchitis or simply to relieve symptoms of acute cough caused by RTI. 4 The effects of these drugs in the treatment of various acute and chronic inflammatory diseases of the upper and lower respiratory tract have been investigated in a large number of clinical studies, which, however, were designed and conducted well before the principles of Good Clinical Practice were established. Many trials were open and non-controlled, and only relatively few randomised controlled trials (RCTs) are available for each individual drug in each indication. For erdosteine and N-acetyl cysteine (NAC), however, well-designed, blinded, placebo-controlled trials have recently shown a reduction in the frequency and duration of exacerbations in patients with COPD.5–10
In the past years, a number of systematic reviews have been published on the use of mucoactive agents in chronic bronchitis or COPD, 11 bronchiectasis, 12 acute cough, 13 or as an adjunct to antibiotics in acute pneumonia. 14 Almost all of them have concluded that mucolytics and mucokinetics have only a weak evidence-based support. The approach used in these reviews is scientifically sound and rigorous, but it reflects only the evidence provided by RCTs that were selected based on very strict criteria. No consideration has been given to results of other RCTs, and open, prospective, or retrospective studies, or to patient’s self-perceived and physician-assessed efficacy. In addition, all reviews are based on a very limited amount of studies and aggregate outcomes of trials conducted with different drugs.11–14
Ambroxol, bromhexine, carbocysteine, erdosteine, NAC, and sobrerol have all been awarded well-established status by regulatory authorities and are available as OTC or prescription products for selected indications. This work aims to evaluate the evidence-based efficacy and safety of these substances in the therapy of upper respiratory airways infections was provided not only by RCTs but also by observational studies.
Methods
We conducted searches in MEDLINE and other databases (search terms: antitussiv*, cough, productive cough, expectorant, mucolytic, ambroxol, bromhexine, carbocysteine, erdosteine, N-acetyl cysteine, sobrerol) for clinical trials and reviews on the efficacy and safety of these substances. No time limit has been set for the search.
We included in our evaluation RCTs and open, controlled, and uncontrolled trials with the aim to evaluate the reported efficacy and safety of these mucolytics with no restrictions on trial design. For products for which recent analyses have been published, we have considered all studies discussed in the reviews, but for the sake of brevity, we do not list or provide specific comments on individual trials unless needed. We did not limit the analysis to the proportion of participants who were cured but we considered also subjective and objective end points.
Ambroxol
Ambroxol exerts stimulating effects on mucociliary clearance and it increases cough effectiveness through its mucokinetic properties and stimulating surfactant secretion. 4 It has been available on the market for almost 50 years in several galenic forms, including ampules for parenteral use to treat the infant respiratory distress syndrome.
Physiologically, ambroxol has been shown to exert secreto-lytic, antioxidant, and anaesthetic activities. 15 This explains its usefulness in the prevention 16 and treatment of upper RTIs (URTI) associated with abnormal mucus secretion or impaired mucus transport. Its efficacy has been shown in more than 100 clinical observational, uncontrolled, or randomised, controlled, double-blind trials on more than 15 000 adult and paediatric patients with various forms of acute and chronic diseases of the upper and lower respiratory tract. 15 Animal and human studies have shown that ambroxol, administered concomitantly with amoxicillin, or ampicillin and erythromycin, increases the antibiotic levels in the lung.17–20
The product is safe, 15 although the Pharmacovigilance Risk Assessment Committee (PRAC) of the European Medicines Agency (EMA) has recommended to update the ambroxol product information by including the risk of allergic reactions and serious cutaneous adverse reactions. 21 Recently, a case of ambroxol-induced focal epileptic seizure in a patient with epilepsy has also been reported. 22
To our knowledge, no interactions of ambroxol with other drugs have so far been demonstrated. The use of ambroxol in children younger than 2 years is not recommended.
Bromhexine
Bromhexine, like ambroxol, is available in several galenic formulations. Animal and human studies suggest that bromhexine influences mucus production, sputum quality and quantity, ciliary activity, and cough severity and frequency. 23 Zanasi et al 23 evaluated approximately 40 clinical studies conducted in patients with COPD, chronic or acute bronchitis, URTI and lower RTI, and bronchiectasis. The study designs ranged from observational, uncontrolled, to randomised, controlled, double-blind trials and included adults as well as children. Overall, bromhexine showed a modest efficacy, but its use was associated with a clinically consistent and subjectively perceived improvement in mucus clearance. 23
As with ambroxol, antibiotic penetration seems to be enhanced by the concomitant administration of bromhexine.24–26
The safety of bromhexine has been demonstrated by clinical studies and long-term use, but the PRAC recommendations issued for ambroxol apply to bromhexine as well. 21 Bromhexine should not be used in children younger than 2 years.
Carbocysteine and NAC
N-acetyl cysteine and, to a lesser extent, carbocysteine (S-carboxymethyl-
Cysteine derivatives have been shown in vitro to break disulphide bridges between the macromolecules present in the mucus, thus leading to a reduced mucus viscosity. 28 They are also endowed with antioxidant activities that, for instance, have been linked to their beneficial action in the pharmacologic treatment of COPD.29–32 In adult patients with COPD or chronic bronchitis, both drugs cause a small reduction in acute exacerbations;5,10,33,34 2 studies have shown beneficial effects of carbocysteine on the quality of life of patients with COPD.35,36 N-acetyl cysteine appears of no use in cystic fibrosis. 37 According to a review by Shen et al, 10 long-term high-dose NAC treatment may lead to a lower rate of exacerbations in patients with COPD.
Regarding paediatric patients with acute bronchopulmonary disease, a recent Cochrane review described the efficacy of both drugs as limited. 38
N-acetyl cysteine and carbocysteine are generally recognised as safe in adults and children, but there have been reports of respiratory paradoxical adverse drug reactions associated with their systemic use in paediatric patients. 39 Two case reports have suggested that carbocysteine may cause pneumonia in predisposed patients.40,41 This would question the risk/benefit ratio of these drugs in children and in selected groups of patients.
Erdosteine
Erdosteine is a thiol derivative with mucolytic 42 and antioxidant activity. 43 It has been on the market for more than 20 years for the treatment of chronic obstructive bronchitis, including acute infective exacerbation of chronic bronchitis and COPD.42,44 In clinical trials, erdosteine reduced frequency and severity of cough and sputum viscosity more effectively than placebo and was more efficient in reducing sputum adhesion than ambroxol. 44 Less (approximately 30) trials have been conducted with this drug than with the other mucolytics so far discussed, 45 but in recent well-designed, blinded, placebo-controlled trials, erdosteine has shown reduction in the frequency and duration of exacerbations in patients with COPD,6–9 and, when administered concomitantly to standard treatment, symptom improvement and reduction in exacerbation of chronic bronchitis/COPD, associated with a reduction in hospitalisation rate and an improved quality of life.42,44,45
Data on the use of erdosteine in indications other than COPD are very scant. Nevertheless, 2 studies have shown beneficial effects of an erdosteine/amoxicillin combination in the treatment of paediatric patients with acute RTI.46,47 Erdosteine alone, however, had no effects on paediatric patients with rhinosinusitis. 48
The safety profile of erdosteine is generally quite good, the most common side effect being heartburn. No specific interactions with other drugs have been described.
Sobrerol
Sobrerol has been on the market of mainly European countries for almost 50 years. It has been shown in vivo to increase mucus production and ciliary motility, thus improving mucociliary clearance, 49 and to reduce the viscosity of tracheobronchial mucus without causing any alterations of the alveolar surfactants. 50 Radical scavenging activities have also been reported. 51
Although recent, specific reviews exist for ambroxol, 15 bromhexine, 23 NAC and carbocysteine,38,52,53 and erdosteine,44,45,54,55 sobrerol has not received much attention, probably because most studies have been published in Italian and often in difficult-to-access journals. Only 1 old, outdated review on sobrerol has so far been published. 49 For this reason, we have decided to compile a tabular presentation of all available studies conducted with this compound (Table 1). A total of 10 double-blind and 15 open (controlled and non-controlled) studies conducted on adults and children have been published, covering acute and chronic airways diseases.56,58–70,72–80,82,83 Sobrerol has been shown to be safe and effective also in paediatric patients and infants.56,66–68,77,80,83 One study conducted on paediatric patients with whooping cough 83 suggests a synergistic effect of sobrerol with antibiotics similar to that observed for other mucolytics; no negative interactions were observed.63,77,78
Synopsis of clinical studies conducted with sobrerol.
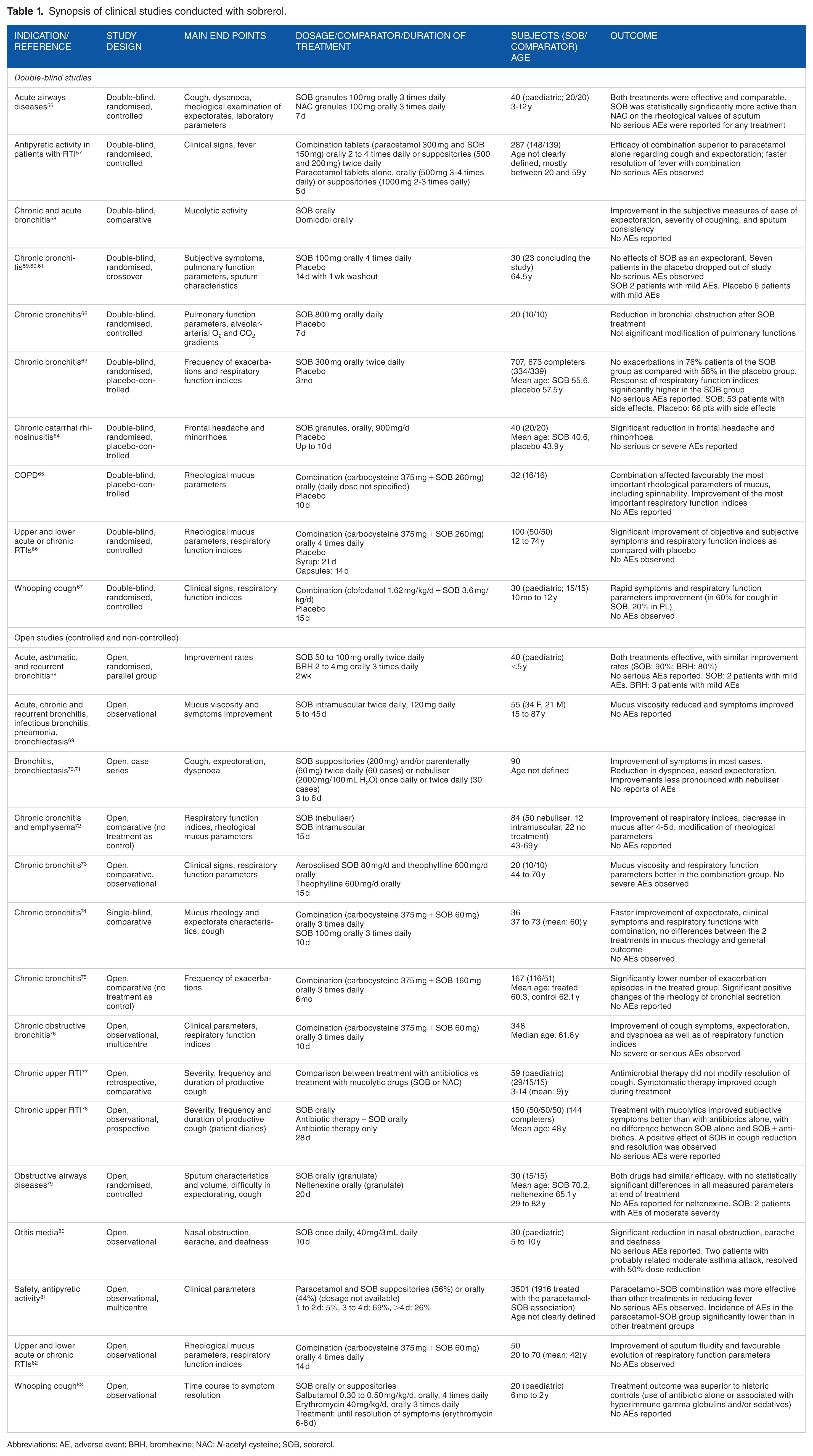
Abbreviations: AE, adverse event; BRH, bromhexine; NAC: N-acetyl cysteine; SOB, sobrerol.
In 2 studies, concomitant administration of sobrerol and paracetamol led to a better febrifuge action than the use of paracetamol alone.57,81
No clinical study reported any noteworthy AEs for sobrerol; the most common AEs reported (gastrointestinal problems and allergic reactions) were mostly of mild nature.
Discussion and Conclusions
Observational studies and real-life use have shown that mucolytics have an important place in the relief of upper respiratory tract symptoms by easing the elimination of mucus, even though the published literature regarding their effectiveness has been defined as inconsistent. 84 All drugs reviewed have shown comparable efficacy in the symptomatic treatment of productive cough, with some shared characteristics and some more specific features (Table 2).
Main features of the mucolytic drugs reviewed.
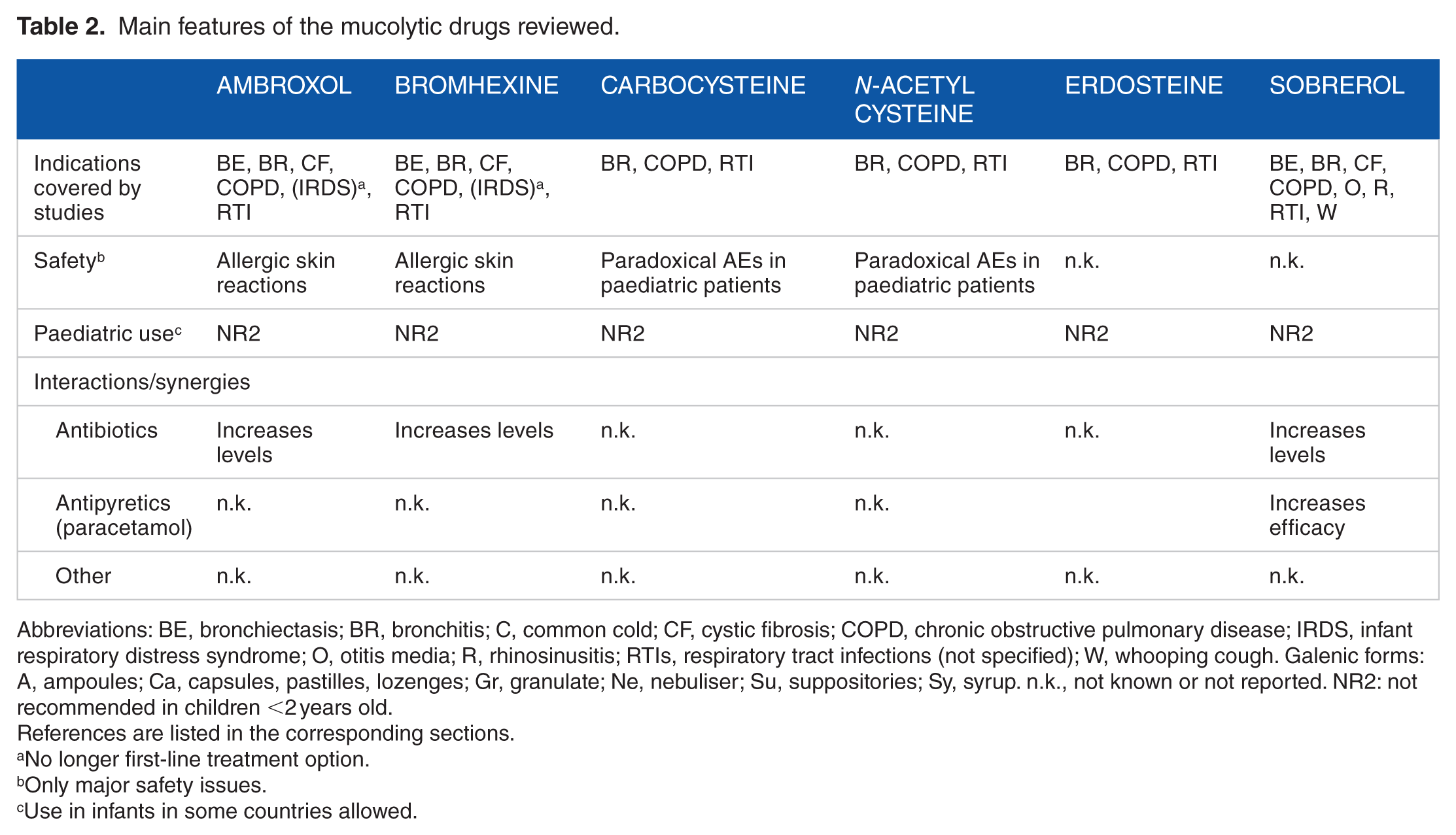
Abbreviations: BE, bronchiectasis; BR, bronchitis; C, common cold; CF, cystic fibrosis; COPD, chronic obstructive pulmonary disease; IRDS, infant respiratory distress syndrome; O, otitis media; R, rhinosinusitis; RTIs, respiratory tract infections (not specified); W, whooping cough. Galenic forms: A, ampoules; Ca, capsules, pastilles, lozenges; Gr, granulate; Ne, nebuliser; Su, suppositories; Sy, syrup. n.k., not known or not reported. NR2: not recommended in children <2 years old.
References are listed in the corresponding sections.
No longer first-line treatment option.
Only major safety issues.
Use in infants in some countries allowed.
Current guidelines and reviews of non-bacterial RTI recommend symptomatic cough relief treatment only.85–87 The primary goal of treatment in case of productive cough is to support expectoration and thereby to indirectly reduce cough. To improve the patient’s general condition, the treatment should also aim at a rapid recovery from symptoms secondary to cough such as sleep disturbance, impaired well-being, dyspnoea, and chest pain. 87
All mucolytics reviewed here provide symptomatic cough relief and possibly shorten the duration of symptoms. They are also endowed with additional pharmacologic properties that may well contribute to their clinical benefit in the treatment of respiratory diseases such as bronchitis and COPD. The conflicting clinical evidence mentioned in recent reviews11–14,33,88 is probably the consequence of several factors, such as the study designs used, the self-limiting nature of the disease, as well as the lack of well-defined study standards. There is also no clear consensus on end points to be used in clinical efficacy trials with mucolytics; for instance, the correlations between some efficacy end points are very poor, the acute or long-term effects of mucoactive therapy cannot be measured reliably, and the intra- and interpatient variability can be very high. 84
The galenic form may also influence the effectiveness of a mucolytic drug but only little comparative evidence has been gathered with this drug class. One trial featuring a well-designed head-to-head comparison has studied nebulised products containing ambroxol, NAC, or sobrerol and has shown substantial differences among the 3 apparently identical galenic forms, with the sobrerol concentration in the aerosolised form being larger than that of ambroxol or NAC. 89 This may suggest, for instance, that a shorter sobrerol nebulisation could achieve the same results obtained with a longer nebulisation with ambroxol or NAC.
Positive interactions with antibiotics have been reported for ambroxol,17–20 bromhexine,24–26 and sobrerol. 83 In the context of the indications for which mucolytics are used, however, this property is of limited usefulness.
However, 2 studies report a positive interaction of sobrerol with an antipyretic.57,81 This synergistic effect should be further investigated. Paracetamol is often used by patients with common cold symptoms to reduce fever, and if the concomitant treatment of both drugs leads to a better antipyretic action of paracetamol than administration of paracetamol alone, the paracetamol dosage could be decreased without loss of efficacy and possibly with a reduction in potential AEs. 57
All mucolytics discussed in this review have a good safety profile, although some precautions should be taken when using ambroxol and bromhexine 21 and the use of NAC and carbocysteine should be monitored in special patient groups.21,39–41
Ambroxol, bromhexine, carbocysteine, erdosteine, NAC, and sobrerol may alter the volume of secretions or their composition; therefore, they can effectively ease symptoms of respiratory tract diseases such as productive cough. Their mucolytic, anti-inflammatory, and antioxidant properties all contribute to their clinical benefit. In patients with COPD, they may help to reduce frequency and duration of exacerbations. Upper RTIs being a multifaceted disease, treatment must rely on the simultaneous treatment of all symptoms. In addition, the overall treatment success is dependent on a number of additional factors. For instance, in indications such as COPD or chronic bronchitis, adherence to treatment and simultaneous targeting of different pathological mechanisms are crucial to achieve symptoms resolution. Overall, the available evidence from randomised, controlled, and observational trials, as well as pragmatic, real-life experience, suggest that these mucolytics, taken at the recommended dosages, are useful in the therapy of lower respiratory diseases such as COPD and bronchiectasis, as well as of upper respiratory airways infections, including bronchitis, sinusitis, and rhinosinusitis.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The preparation of this article was possible by an unrestricted grant by Pharmaidea s.r.l., Travagliato, Italy.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Both authors contributed equally to the design, preparation and writing of this review.
