Abstract
Nasal nitric oxide (nNO) measurement is a diagnostic test for primary ciliary dyskinesia (PCD). Here, we have shown the development of unilateral PCD-like symptoms associated with low nNO. A 60-year-old man had been previously healthy but developed unilateral, severe pansinusitis. He required surgical drainage of all left sinuses, and biopsies showed loss of the ciliated epithelium. At 4 weeks, he had unilateral (left-sided), profuse, clear rhinorrhea characteristic of PCD, and his surgical ostia were all patent endoscopically. His left-sided nNO was less than the right side by 37 ± 1.2 nL/min; this difference decreased to 18 ± 0.87 nL/min at 5 weeks and was gone by 6 weeks when his symptoms resolved. Measurements of 2- and 10-second measurements, in addition to standard nNO measurements, identified this discordance. We conclude that nNO reflects, in part, the production of NO by the ciliated epithelium, not just in the absence or occlusion of sinuses. Early (nasal/sinus volume) measures may be better for diagnosing PCD in than standard, steady-state assays in certain populations.
Introduction
In our center, we have been measuring nasal nitric oxide (nNO) production rate because of data suggesting that it is an important marker for the presence of primary ciliary dyskinesia (PCD). Here, we report an unusual case with unilateral symptoms and left-right nNO discordance that may provide new insights and opportunities regarding nNO measurement. Primary ciliary dyskinesia is a genetically heterogeneous recessive disorder of motile cilia that leads to a multitude of manifestations notably pulmonary, ear, and sinus diseases and organ laterality defects in approximately 50% of cases.1–4 The estimated incidence of PCD is approximately 1 per 15 000 births, but the prevalence of PCD is difficult to determine, primarily because of limitations in diagnostic methods that focus on testing ciliary ultrastructure and function.5,6 The use of these diagnostic approaches shows that at least 30% of patients with PCD have normal ciliary ultrastructure.7–9 More accurate identification of patients with PCD has also allowed definition of a strong clinical phenotype, which includes neonatal respiratory distress in >80% of cases, daily nasal congestion and wet cough starting soon after birth, and early development of recurrent/chronic middle ear and sinus disease.4,10,11–13
Several studies suggest that nNO measurement is a diagnostic test for PCD.14–17 Production of NO by the airway epithelium has been extensively studied in a number of diseases.14–16,18–20 Exhaled levels of NO from the lungs and sinuses are quite high, in general, in allergic asthma but paradoxically low in other inflammatory conditions, including cystic fibrosis and PCD. 18 Several mechanisms have been proposed to account for the different levels of NO, but no mechanism to date has accounted for all of the clinical variation.
In older children, the nNO value of <77 nL/min is considered diagnostic of PCD if the patient has been confirmed not to have cystic fibrosis (CF); although levels can be low in CF, they are lower still in PCD.14–16,21 Ironically, however, NO synthase expression in the airway epithelium is not a determinant of these differences.14,18,19,22 It is thus hypothesized that nNO reflects measurement of sinus NO production, 21 and that nNO cannot be adequately sampled in many patients with PCD because of chronic sinus obstruction. 23 However, lower airway NO production rates are also significantly impaired in PCD, 18 suggesting a primary epithelial issue rather than simple mucoid impaction. Nasal NO is higher in CF than in PCD, although sinus occlusion is often worse in CF than in PCD; and patients with PCD without severe sinus disease still have very low nNO. Therefore, it is likely that the cause of low nNO is both sinus occlusion and a defect in airway epithelial NO production that is not directly dependent on NOS expression. 19
Methods and Case Report
A 60-year-old man developed fevers, nausea, and emesis and progressed to septic shock. Blood cultures grew methicillin-sensitive Staphylococcus aureus. Because of left-sided facial swelling, a computed tomography was obtained demonstrating left-sided pansinusitis; his right-sided sinuses were essentially normal (Figure 1). Despite intravenous antibiotic therapy, his symptoms persisted and he was taken to the operating room for left-sided maxillary, ethmoid, sphenoid, and frontal sinus surgery. At the time of his operation, the mucosa was very diseased and biopsies and cultures were obtained. Sinus cultures, like the blood cultures, grew methicillin-sensitive S aureus. The biopsies of the left sinus mucosa revealed epithelial necrosis and no cilia (Figure 2). Follow-up endoscopies after antibiotics and isotonic sinus irrigations revealed patent ostia and normalizing mucosa, but the improvement was very slow taking months for complete resolution. The patient began experiencing continuous, clear rhinorrhea—the type of chronic, unremitting rhinorrhea experienced by patients with PCD—but only unilaterally on the left side, and not in conjunction with any other PCD symptoms 1 week after surgery. He had no, right-sided symptoms 1 week after surgery. Serial bilateral nNOs were measured (PCD Foundation protocol 14 ; Sievers NOA 280i) until 6 weeks after the onset of infection, at which point the continuous left-sided rhinorrhea resolved. Gas mixes rapidly between left and right nostrils at 5 mL/s flow. The early [NO], before mixing, best distinguished absent cilia from present cilia. [NO]2sec was measured: the first second (5 mL) clears dead space in the tubing and instrument and, given the volume of the sinuses and nose, the second samples the relevant volume. The nasal NO production curve display on the NOA was time fractionated using the NOA software display. Each assay was performed in triplicate.
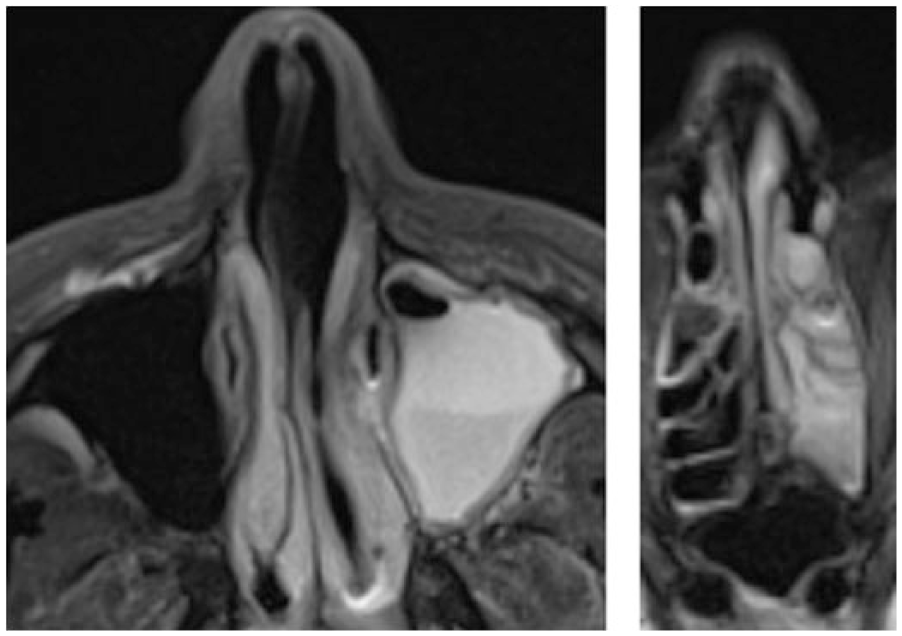
Acute infection of all left-sided sinuses, with no infection on the right. MRI, Time 0. Left panel, maxillary; right panel, ethmoid.
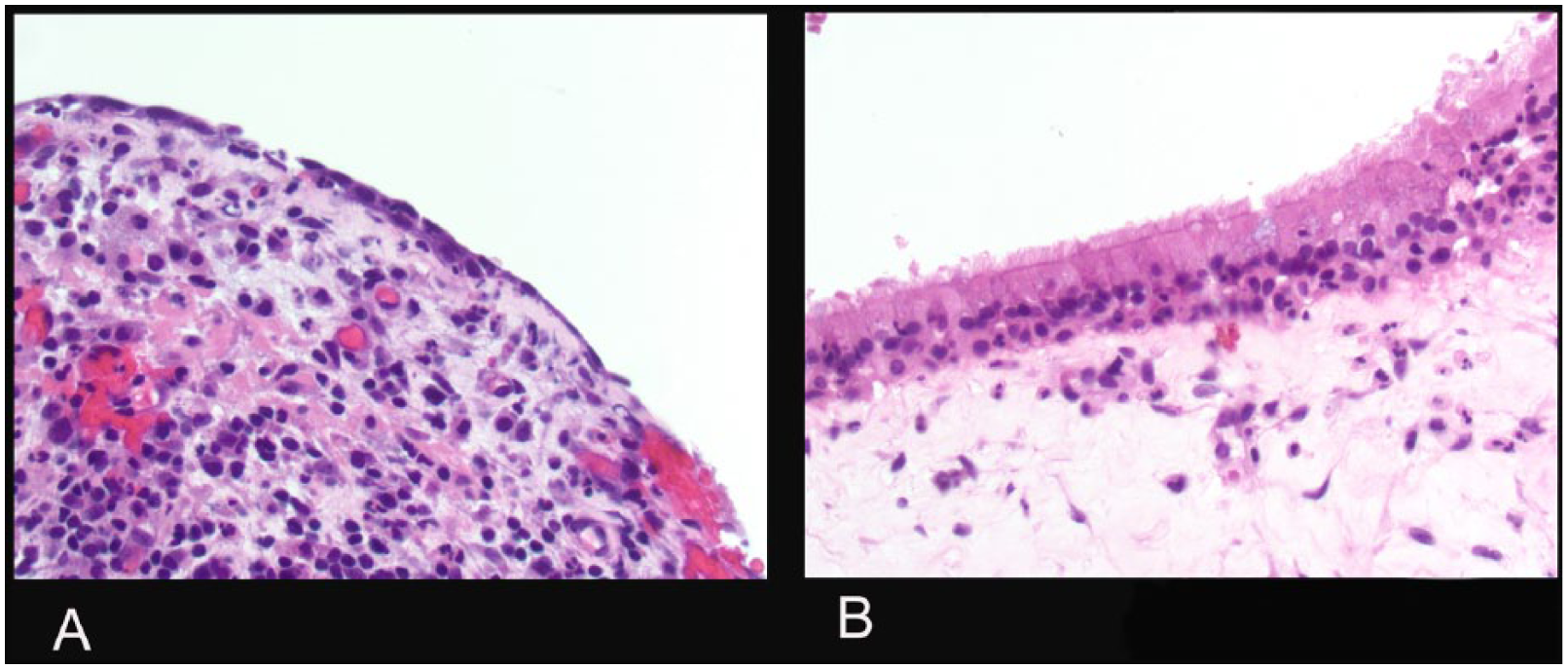
A. Left Maxillary sinus exhibit denuded epithelium. B - Healthy control. H&E staining 100x magnification.
Statistical analysis
Analysis of variance (ANOVA) followed by Student t test was used for data analysis. All data are expressed as mean ± SD unless otherwise specified. Results with P < .05 were considered to be significant.
Results
The subject had served previously as a normal control and had equal left and right premorbid nNO flow rates (142 nL/min). After 4 weeks, the acute infection—when he had unilateral left-sided PCD-like symptoms—endoscopy revealed continued necrosis of the sinus mucosa but patent sinus meatuses and no further purulent fluid. By 6 weeks, the chronic left-sided rhinorrhea had resolved entirely.
The nNO measurements paralleled the symptoms. At 4 weeks, nNO was 137.4 ± 2.3 nL/min on the right side and 100.7 ± 1.1 nL/min on the left side, so at 4 weeks, the left-sided nNO was less than the right-sided nNO by 37 ± 1.2 nL/min (26% lower).
At 5 weeks, nNO was 111.7 ± 3.4 nL/min on the right side and 93.14 ± 4.3 nL/min on the left side, so the right-left side difference decreased to 18 ± 0.87 nL/min.
At 6 weeks, nNO was 92.4 ± 3.1 nL/min on the right side and 99.1 ± 6.7 nL/min on the left side, so the right-left side difference becomes −6.7 ± 3.6 nL/min at 6 weeks (mean ± SD; n = 3-4 samples; P = .006 by ANOVA; pairwise [Holm-Sidak], P < .05 each) (Figure 3A).
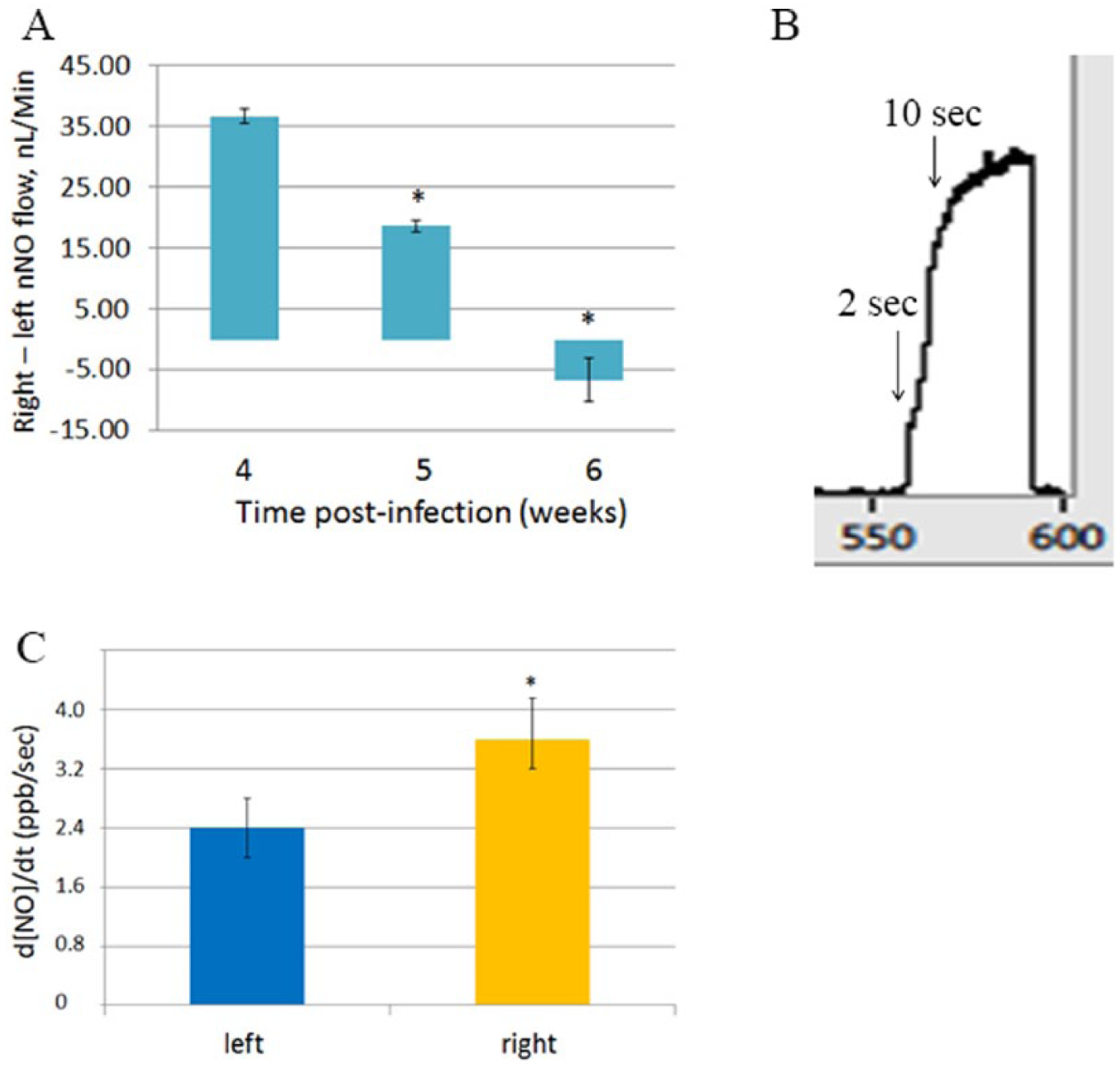
A. After complete resolution of the infection with endoscopically clean sinuses and meatuses, the subject had unilateral chronic rhinorrhea and left/right discordance in nNO flow. P = 0.006 overall; *p < 0.05 compared to the previous week. B. ΔNO10sec during standard nNO measurement. Change in [NO] between the first and second arrow. C. Left-right discordance in ΔNO10sec at week 5 from the beginning of symptoms, with no purulence in the sinuses or occlusion of the meatuses. *p = 0.027
Gas mixes rapidly between left and right nostrils at 5 mL/s flow. The early [NO], before mixing, best distinguished absent cilia from present cilia. [NO]2sec may be ideal: the first second (5 mL) clears dead space in the tubing and instrument and, given the volume of the sinuses and nose, the second samples the relevant volume.
Early measurements occurred before left-sided and right-sided measurements were mixed because of flow. At week 4, the initial increase in [NO]2sec was constantly <20 ppb (<5 nL/min) on the left and >100 ppb (>20 nL/min) on the right. At week 5, the increase in [NO] over the first 10 seconds (Figure 3B) was 2.5 ± 0.4 ppb/s on the left and 3.8 ± 0.7 ppb/s on the right (n = 3 each; P = .027) (Figure 3C).
Discussion
This case of unilateral PCD-like symptoms in a patient with severe, unilateral sinusitis provided a unique opportunity to understand mechanisms causing nNO values to be low. This is important because diagnostic criteria for PCD for patients more than 5 years old include specific clinical symptoms and nNO production rate of <77 nL/min in the context of a normal sweat chloride and/or a diagnostic genotype. 16 Electron microscopy (EM) is an important diagnostic tool for ciliopathy. Infection can cause inflammatory changes visible on EM as reported by pathologists.24,25 However, EM results are now understood often to be falsely positive or negative, and they are no longer the sine qua non for PCD.10,14,16,26 The nNO, however, needs to be performed strictly in accordance with the PCD Foundation standards to be considered diagnostic. 14 In this sense, nNO is the “sweat chloride of PCD.”
The reason for low nNO production in PCD remains controversial.7,19 It is held that levels are low because of thick mucus plugging the sinus ostia: NO concentrations are higher in the sinuses than in the nose. Data from Walker et al, 18 demonstrate that NO production by the pulmonary airway wall itself is decreased in PCD, suggesting that plugging of the sinus ostia may not, alone, account for decreased nNO in PCD. Our case has provided a unique insight. Specifically, a patient developed unilateral PCD symptoms (chronic, profuse clear rhinorrhea) and had ipsilateral low nNO. His sinus ostia were widely patent and his cilia were injured or absent on the affected side, suggesting that the symptoms of low nNO were not because of mucous plugging but because of loss of normal, ciliated epithelium.
This case also demonstrates that gas mixes rapidly between left and right nostrils at the flow used for nNO measurement (5 mL/s). The data suggest that early [NO], before mixing, best distinguished absent cilia from present cilia in this patient. Standard nNO measurement procedure requires that a steady plateau of NO production be recorded.14,21,26 However, this requirement is not suitable in younger children who do not hold still and in whom these plateau measures do not adequately distinguish patients with PCD from controls.
Our data suggest, however, that [NO]2sec might be studied as a test to distinguish the presence from the absence of cilia in nasal and sinus epithelium itself: the first second (5 mL) clears dead space in the tubing and instrument and, given the volume of the sinuses and nose, the second samples the relevant volume. Alternatively, change in [NO] in the first 10 seconds may distinguish ciliated from nonciliated better than the equilibrium nNO.
Conclusions
Decreased nNO is caused, in part, by loss or dysfunction of airway epithelium, independently of mucus obstruction. Measuring early fraction of nNO may be useful for distinguishing epithelial ciliary dysfunction.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fundings: NHLBI, PPG 1 P01 HL128192.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
KR collected data and prepared the manuscript. BG analysed the data and prepared the manuscript. JW collected and analysed the data. NM collected and analysed data and prepared the manuscript.
Disclosures and Ethics
As a requirement of publication, author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality, and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
