Abstract
Dextrocardia with situs inversus totalis (SIT) is a rare congenital anomaly involving complete mirror-image reversal of thoracic and abdominal organs, which may complicate diagnostic evaluation and management despite often being benign. We report a 45-year-old Ethiopian woman with type 2 diabetes mellitus and hypertension who presented with dry cough, right-sided pleuritic chest pain, dyspnea, and low-grade fever. Examination revealed respiratory distress and a right-sided cardiac apex, while laboratory workup demonstrated anemia, impaired renal function, and proteinuria. Imaging confirmed SIT with right lower lung consolidation and incidental cholelithiasis, and electrocardiogram (ECG) showed extreme right-axis deviation with absent R-wave progression; echocardiography revealed normal intracardiac anatomy and preserved function. Although SIT is commonly discovered incidentally, reversed anatomy can create diagnostic challenges and requires careful clinical correlation, particularly in settings with limited imaging availability. This case highlights the importance of recognizing SIT to prevent misinterpretation, guide safe management, and contribute to the limited regional epidemiological data.
Introduction
Dextrocardia with situs inversus totalis (SIT) is a rare congenital anomaly characterized by complete mirror-image transposition of the thoracic and abdominal organs. In this condition, the cardiac apex points to the right side of the chest, and the arrangement of major visceral organs is reversed, with the liver and gallbladder positioned on the left and the spleen and stomach located on the right.1-3 Embryologically, it results from abnormal left–right body patterning during early organogenesis, often involving defects in motile cilia and NODAL signaling pathways. 4 The condition is generally inherited in an autosomal recessive pattern and has an estimated prevalence of approximately 0.01% in the general population, making it a rare clinical entity.1,5
Most individuals with dextrocardia and SIT remain asymptomatic throughout life, and the condition is frequently discovered incidentally during imaging studies conducted for unrelated clinical indications. 6 By itself, SIT is usually benign, with structurally normal cardiac and visceral anatomy in the majority of patients; congenital heart defects occur in only 5% to 10% of cases. 5 When associated with other anomalies, the condition can have significant clinical implications. For instance, primary ciliary dyskinesia (PCD), a genetic disorder affecting motile cilia, can present in combination with SIT, chronic sinusitis, and bronchiectasis, forming the triad known as Kartagener syndrome. Other reported associations include renal anomalies such as unilateral agenesis, ectopic kidneys, and biliary malformations, although these remain uncommon.1,4 The presence of such anomalies can influence clinical management and long-term outcomes.
SIT can also complicate the interpretation of physical examination findings, electrocardiography, and imaging studies, potentially leading to diagnostic errors or procedural complications. Awareness of this anatomical variation is therefore crucial, particularly in emergency, surgical, or invasive diagnostic settings.
In Ethiopia, dextrocardia with SIT has been rarely documented. The limited number of reported cases in sub-Saharan Africa likely reflects underdiagnosis, due to low clinical suspicion and limited access to advanced imaging. One recent report described a 30-year-old Ethiopian woman who presented with right-sided chest pain and was subsequently diagnosed with SIT after clinical assessment and imaging confirmed the mirror-image arrangement of her thoracoabdominal organs. 3 Given its rarity and potential clinical implications, reporting such cases contributes to improved awareness, diagnostic accuracy, and tailored management strategies, especially in resource-limited settings.
Case Presentation
A 45-year-old married woman from Worka, Oromia Region, Ethiopia, presented to the Department of Internal Medicine with a 4-day history of dry cough, right-sided pleuritic chest pain, shortness of breath, and intermittent low-grade fever. She denied orthopnea, paroxysmal nocturnal dyspnea, palpitations, or urinary symptoms. There was no family history of similar illnesses.
Her medical history was significant for type 2 diabetes mellitus diagnosed 10 years ago and hypertension diagnosed 1 year ago. She had been on Metformin 500 mg orally twice daily, Glimepiride 2 mg orally once daily, and Amlodipine 5 mg orally once daily. She had 5 children and no known drug or food allergies.
On physical examination, she appeared in marked respiratory distress with use of accessory muscles and intercostal retractions. Her vital signs were: blood pressure 137/74 mmHg (right arm, sitting), pulse rate 80 beats per minute (regular and full), respiratory rate 24 breaths per minute (deep and regular), peripheral oxygen saturation (SpO2) 88% on room air, and temperature 35.1°C (axillary). A repeat measurement was 36.7°C, suggesting the initial reading was likely a measurement artifact rather than true hypothermia. The conjunctivae were pink. Chest examination revealed decreased air entry over the right lower third posteriorly, coarse crepitations in the same area, and relative dullness to percussion. Cardiovascular examination showed an absent apical impulse in the usual left-sided location, with heart sounds best heard on the right side, raising suspicion of dextrocardia.
Complete blood count (CBC) showed: WBC 7.7 × 109/L, hemoglobin 8.8 g/dL, mean corpuscular volume (MCV) 87.9 fL, and platelets 195 × 109/L. Renal function tests revealed elevated creatinine (3.74 mg/dL) and urea (112 mg/dL). Estimated glomerular filtration rate (eGFR) was approximately 17 mL/min/1.73 m2, consistent with stage 4 to 5 chronic kidney disease (CKD). Renal impairment was recognized as a major comorbidity and considered in immediate clinical decision-making, including careful medication selection and avoidance of nephrotoxic exposure. Urinalysis showed proteinuria (+2). Serum sodium (135-145 mmol/L), potassium (3.5-5.0 mmol/L), and chloride (98-106 mmol/L) were within reference ranges. Liver function tests, including AST (10-40 U/L), ALT (7-56 U/L), and total bilirubin (0.2-1.2 mg/dL), were normal. Lipid profile values were also within normal limits according to institutional reference standards (total cholesterol < 200, LDL < 130, HDL > 40, and triglycerides < 150 mg/dL). Blood film and malaria rapid diagnostic tests were negative.
Chest X-ray (posteroanterior view) demonstrated dextrocardia, with the cardiac apex and aortic knob on the right side, along with right lower lung opacity (Figure 1). Abdominal ultrasound revealed situs inversus, with the liver in the left upper quadrant, the spleen in the right upper quadrant, and incidental cholelithiasis. There was also evidence of right lower lung zone consolidation. Renal ultrasound demonstrated findings suggestive of CKD, including increased cortical echogenicity and reduced corticomedullary differentiation, without evidence of hydronephrosis. Abdominal CT scan confirmed situs inversus with a mirror-image arrangement of intra-abdominal organs (Figure 2).

CXR with cardiac apex and aortic knob located on the right side, and an opacity observed in the right lower lung on the posteroanterior (PA) view of the chest X-ray.
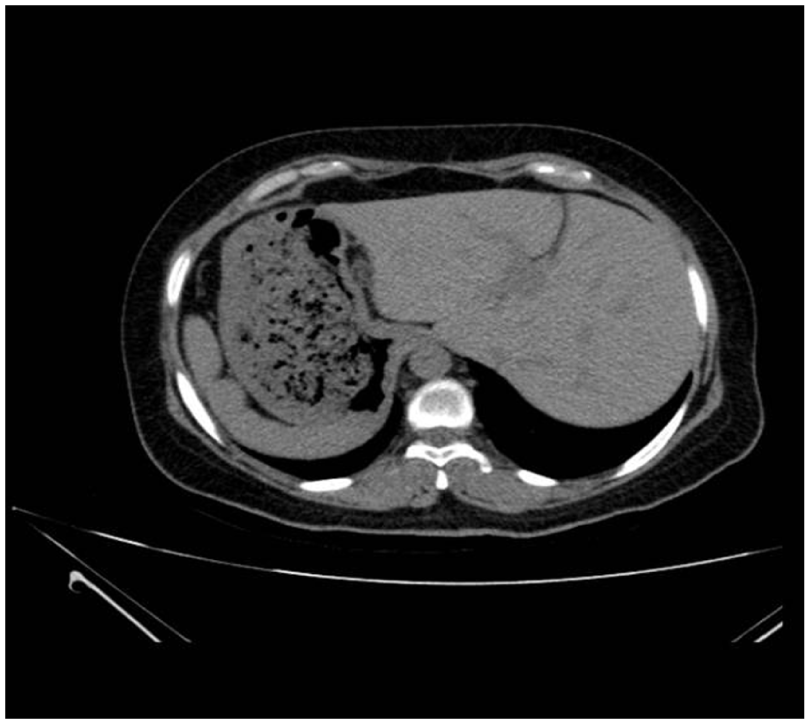
Axial non-contrast computed tomography (CT) scan of the upper abdomen demonstrating SIT. Note the liver positioned in the left upper quadrant and the stomach and spleen located in the right upper quadrant.
Electrocardiogram (ECG) showed sinus rhythm, extreme right axis deviation, global negativity in lead I, and absent R-wave progression in the precordial leads (Figure 3). Transthoracic echocardiography demonstrated a right-sided cardiac apex, D-looped ventricles, and atrioventricular (AV) and ventriculoarterial (VA) concordance. Systemic and pulmonary venous connections were normal, and all cardiac chambers had normal internal dimensions, with an estimated left ventricular ejection fraction of 60%.

Electrocardiogram (ECG) demonstrating sinus rhythm, extreme right axis deviation, global negativity in lead I and absent R wave progression in precordial leads.
The patient was admitted and started on supplemental oxygen via nasal cannula to maintain SpO2 above 92%. Empiric intravenous antibiotics (Ceftriaxone 1 g daily) were initiated for community-acquired pneumonia, alongside standard supportive care including analgesics and hydration. Her blood glucose and blood pressure were monitored and adjusted as needed. Over the course of 5 days, her respiratory symptoms gradually improved, oxygen requirements decreased, and repeat chest imaging showed partial resolution of the right lower lung consolidation. The incidental cholelithiasis was asymptomatic. She was advised on lifestyle modifications, including dietary changes, and scheduled for outpatient follow-up with the surgical team to monitor for any future biliary complications. No immediate surgical intervention was required.
She was discharged on oral antibiotics to complete a 7-day course, with instructions for follow-up in the internal medicine clinic for ongoing management of diabetes, hypertension, gallbladder disease, and CKD. She was counseled on the importance of close outpatient monitoring, including periodic assessment of serum creatinine and eGFR, optimization of blood pressure and glycemic control, avoidance of nephrotoxic medications, and referral to nephrology for long-term management and risk stratification. At 2-week follow-up, she remained clinically stable, with improved oxygen saturation, stable renal function, and no biliary symptoms. Continued outpatient follow-up by the treating team demonstrated sustained clinical improvement, with no recurrence of respiratory symptoms, stable kidney function without further deterioration, and good adherence to medical therapy and lifestyle recommendations.
Discussion
SIT is a rare congenital anomaly characterized by complete mirror-image transposition of thoracic and abdominal organs. Its estimated prevalence is approximately 1 in 10 000 live births, with most cases discovered incidentally due to its asymptomatic nature. SIT has no strong sex predilection, and is typically inherited in an autosomal recessive pattern.1,2 This unusual anatomical configuration arises from abnormal embryologic rotation during early organogenesis, which is often influenced by defects in left–right body patterning genes and the function of motile cilia, including disruptions in NODAL signaling pathways. 4
Imaging is the definitive diagnostic tool. Chest X-ray, ultrasound, and CT clearly demonstrate mirror-image organ placement and can identify associated anomalies. In this patient, CT confirmed complete SIT and right lower lung consolidation. 7
The clinical significance of SIT is generally minimal; however, it may complicate the interpretation of physical findings and imaging studies. For instance, right-sided heart sounds, reversed liver and splenic dullness, and altered gastric tympany can mislead clinicians. This underscores the need for careful clinical evaluation and awareness of anatomical variations, particularly in emergency or surgical settings. 8 Electrocardiographic changes, such as right-axis deviation, negative lead I complexes, and absent R-wave progression in precordial leads, are characteristic and should prompt imaging to confirm organ transposition. 9
The presence of comorbidities in our patient, including diabetes mellitus, hypertension, and advanced CKD, does not appear directly related to situs inversus, as most individuals with this anomaly have structurally and functionally normal organs. 2 However, stage 4 to 5 CKD represented a clinically significant condition that substantially influenced both risk stratification and management decisions. Severe renal impairment alters pharmacokinetics, necessitates careful medication selection and dose adjustment, increases vulnerability to electrolyte disturbances and cardiovascular complications, and limits the use of contrast-based imaging. Although renal anomalies such as agenesis or dysplasia have been described in some patients with situs inversus, the patient’s renal impairment was most consistent with CKD secondary to long-standing diabetes mellitus and hypertension, supported by proteinuria and ultrasound evidence of chronic parenchymal disease. 10 The relatively stable renal parameters during hospitalization, in the absence of features suggestive of abrupt renal insult, favored an underlying chronic process with possible acute-on-chronic worsening related to infection and dehydration. Importantly, the coexistence of advanced CKD required careful clinical monitoring, renal-protective management, and structured nephrology follow-up, underscoring the need to address major comorbid conditions alongside anatomical anomalies.
Respiratory symptoms in situs inversus should raise suspicion for PCD or Kartagener syndrome, particularly when recurrent sinusitis or bronchiectasis is present. 1 Our patient had no history suggestive of PCD, and imaging did not show bronchiectasis, supporting the diagnosis of uncomplicated SIT. The incidental cholelithiasis in this patient is notable but asymptomatic. Such biliary anomalies are uncommon in SIT; follow-up and lifestyle modification were recommended, with no immediate surgical intervention.
SIT can present unique clinical challenges in emergency and surgical settings. Misinterpretation of symptoms, such as left-sided abdominal pain in appendicitis, or reversed auscultatory findings can delay diagnosis and lead to procedural errors. Awareness of the anatomical variation, careful interpretation of ECGs, imaging confirmation, and meticulous procedural planning are critical to ensure patient safety and effective clinical management. 11
Compared with previously reported cases of SIT, this case illustrates the practical implications of reversed anatomy on clinical assessment and management. While prior reports from Ethiopia and sub-Saharan Africa have largely documented incidental detection of SIT during imaging for unrelated complaints,3,6,10 in our patient, the mirror-image anatomy directly influenced the initial physical examination and interpretation of diagnostic studies. Right-sided heart sounds and the cardiac apex prompted early recognition of dextrocardia, while the reversed positions of the liver and spleen could have complicated abdominal assessment and potentially delayed identification of coexisting conditions such as right lower lung consolidation and incidental cholelithiasis.
This case also highlights a clear clinical takeaway: in patients with SIT, awareness of organ reversal is essential for accurate interpretation of ECG and imaging studies, guiding safe and effective management. Extreme right-axis deviation and absent R-wave progression on ECG required careful correlation with imaging to avoid misdiagnosis. In resource-limited settings where advanced imaging may not be immediately available, SIT can pose a genuine risk of diagnostic and management error, including mislocalization of pulmonary pathology, misinterpretation of electrocardiographic findings as primary cardiac disease, and potential wrong-side procedural planning. In our patient, early recognition of reversed anatomy prevented erroneous cardiac workup and ensured accurate localization of pneumonia, allowing timely and appropriate treatment. Recognizing the anatomical variation therefore not only prevented misdirected diagnostic procedures but also illustrates an underemphasized clinical pitfall: failure to identify SIT during acute presentations may lead to diagnostic delay or inappropriate intervention. This case adds to existing reports by illustrating how SIT, though often benign, can meaningfully influence diagnostic reasoning and management decisions, particularly in patients with comorbidities and in resource-limited settings.
The scarcity of documented SIT cases in sub-Saharan Africa likely reflects underdiagnosis rather than true rarity. Contributing factors include limited access to imaging modalities, low clinical suspicion for rare anatomical anomalies, and underreporting in the medical literature.11,12 Systematic reporting of such cases is vital to enhance clinician awareness, improve epidemiological understanding, and guide safe management practices in settings with constrained resources. Encouraging case documentation and sharing regional experience will also help inform local clinical guidelines and optimize patient outcomes.
Conclusion
This case of a 45-year-old woman with dextrocardia and SIT highlights the incidental discovery of this rare congenital anomaly during evaluation for respiratory symptoms. While SIT is typically benign, it can complicate clinical assessment, imaging interpretation, and procedural planning. Awareness of such anatomical variations is essential to avoid diagnostic errors and ensure safe management. In resource-limited settings like sub-Saharan Africa, underdiagnosis is likely due to limited imaging access and low clinical suspicion. Reporting cases like this 1 not only informs clinicians but also contributes to regional epidemiological knowledge and guides patient-centered care.
Footnotes
Acknowledgements
We thank the patient for agreeing to give their consent to publish their clinical record for this report.
Ethical Considerations
IRB review and approval were waived for this case report.
Consent for Publication
Written informed consent of the clinical details and accompanying images was obtained from the patient.
Author Contributions
Tsegaab Mesele and Kebron W. Aweke contributed equally to this work and share first authorship. Tsegaab Mesele, Kebron W. Aweke, and Samuel Dereje Bedada were involved in direct patient care and clinical management of the case. Tsegaab Mesele, Kebron W. Aweke, and Gelila S. Kassa contributed to data collection and case documentation. Mikiyas G. Teferi and Muluken Bekele Wondimagegnehu contributed to the literature review, interpretation of findings, and drafting of the manuscript. Leul M. Manyazewal contributed to critical revision of the manuscript for important intellectual content. Samuel Dereje Bedada also contributed to clinical interpretation and manuscript review. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data underlying the results presented in this work are available within the manuscript.
