Abstract
Introduction:
Pseudomonas aeruginosa is a frequent cause of healthcare-associated infections and is increasingly encountered in extensively drug-resistant (XDR) or pan-drug-resistant (PDR) forms. Such strains leave clinicians with virtually no therapeutic options and carry high mortality.
Case Presentation:
We describe a 66-year-old man with chronic hypoxic respiratory failure, chronic kidney disease, and recurrent infections who was admitted with septic shock. Initial broad-spectrum antibiotics were ineffective, and cultures yielded PDR Pseudomonas aeruginosa, resistant to cefiderocol and with borderline colistin susceptibility (MIC 2 µg/mL). Antimicrobial susceptibility testing was performed by broth microdilution on a Thermo Fisher Sensititre™ GNX3F panel, with colistin MIC confirmed by reference broth microdilution; results were interpreted per EUCAST v13.0 (2025) and CLSI M100 (2025). Despite high-dose intravenous colistin, cefiderocol, and adjunctive amikacin, the infection persisted, leading to progressive multi-organ dysfunction. Colistin dosing was adjusted for renal insufficiency and subsequently for continuous renal replacement therapy (CRRT). Meropenem (MIC 8 µg/mL; resistant) was trialed in combination with colistin for potential pharmacodynamic synergy. After 12 days of intensive care, and following discussion with family, care was redirected to comfort measures, and the patient died. The cause of death was refractory septic shock due to PDR Pseudomonas aeruginosa with multi-organ failure.
Discussion:
This case illustrates the therapeutic futility posed by pan-drug-resistant (PDR) Pseudomonas aeruginosa. Colistin’s limited efficacy at borderline MICs, compounded by renal impairment, constrained dosing. Even newer agents such as cefiderocol proved ineffective. Experimental therapies, including bacteriophages, novel β-lactamase inhibitors, and antimicrobial peptides, remain unavailable in most clinical settings.
Conclusion:
PDR Pseudomonas aeruginosa represents an urgent global threat. Until novel therapies become accessible, stringent stewardship, infection control, and preventive strategies remain the most effective defenses.
Keywords
Introduction
Pseudomonas aeruginosa accounts for a sizeable proportion of hospital-acquired infections, including those observed in intensive care settings. 1 It benefits from intrinsic traits, such as low membrane permeability and robust efflux pumps, thereby reducing the impact of many antimicrobial classes. 2 Horizontal transfer of resistance determinants and rapid genetic mutations add to the organism’s capacity to withstand pharmacologic interventions. 3 Individuals with protracted hospital stays, mechanical ventilation, repeated exposure to antibiotics, and conditions such as chronic kidney disease are at a particularly high risk of these infections. 4 Extensive drug resistance (XDR) in Pseudomonas aeruginosa is difficult to manage. 5 When resistance extends to virtually all available antimicrobial agents, as in pan-drug resistance (PDR), treatment options shrink to a critical level. 6 Following the CARE (CAse REport) guidelines, 7 this report reviews the trajectory of a patient who presented with septic shock caused by PDR Pseudomonas aeruginosa and received high-dose colistin and cefiderocol for several days without improvement.
Case Report
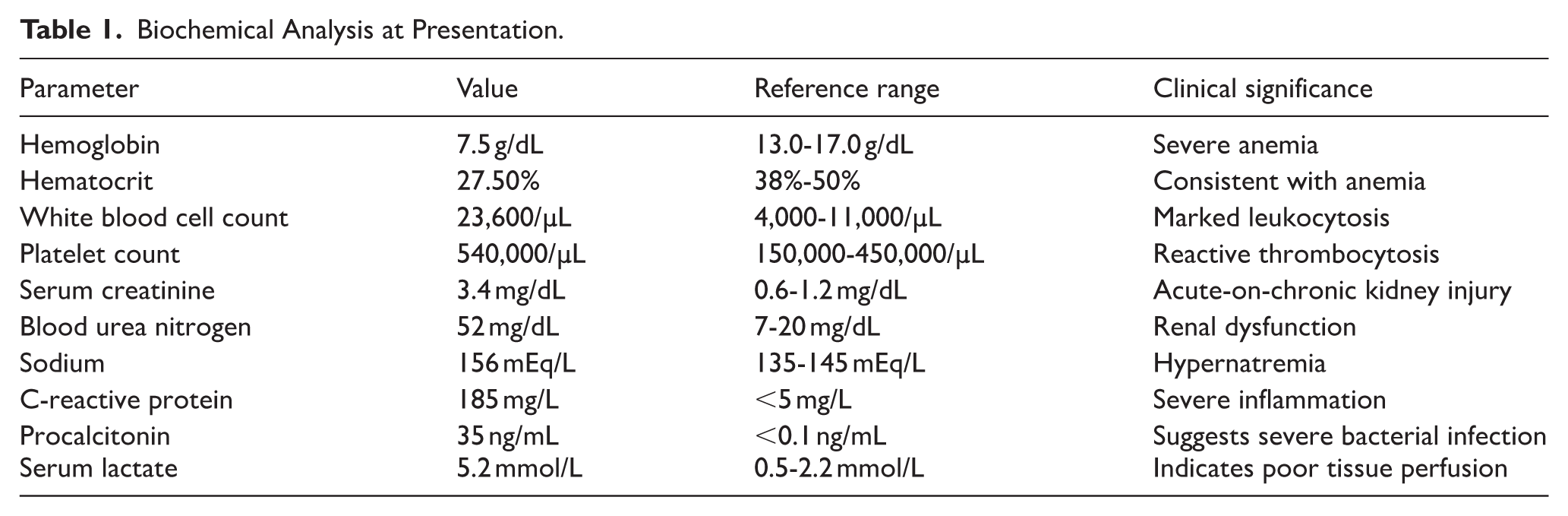
A 66-year-old man was transferred from a rehabilitation facility to a tertiary care hospital because of an uncontrollable fever, hypotension, and escalating ventilator requirements. His medical history included chronic hypoxic respiratory failure requiring prolonged mechanical ventilation, chronic kidney disease (stage 4), hypertension, hyperlipidemia, multiple prior infections with resistant bacteria, and an inoperable aortic root abscess that had been diagnosed a year earlier. On arrival, his blood pressure was 76/44 mmHg, temperature was 40°C, and Glasgow Coma Scale score was 10/15. Initial laboratory results indicated acute-on-chronic kidney injury, with a serum creatinine of 3.4 mg/dL, severe leukocytosis at 23,600/µL, hemoglobin of 7.5 g/dL, and a sodium level of 156 mEq/L. Serum lactate was 5.2 mmol/L, consistent with tissue hypoperfusion in septic shock. These findings are detailed in Table 1, which shows the principal biochemical values at the time of admission.
Biochemical Analysis at Presentation.
Chest radiography revealed bilateral pleural effusions and cardiomegaly, and a subsequent chest computed tomography scan showed a chronic penetrating aortic ulcer flanked by inflammatory changes (Figure 1). The patient was administered empiric broad-spectrum antibiotics based on prior resistant infections. These comprised ceftazidime at 2 g intravenously every 8 hours, trimethoprim-sulfamethoxazole at 320 mg/1600 mg intravenously every 12 h, fosfomycin at 4 g intravenously every 6 hours, and linezolid at 600 mg intravenously every 12 hours. He also received norepinephrine infusion to maintain adequate perfusion. Two sets of blood cultures (aerobic/anaerobic) and an endotracheal aspirate were obtained before antimicrobials. Organism identification was performed by MALDI-TOF mass spectrometry.

Chest CT scan showing the penetrating aortic ulcer with adjacent inflammatory changes and bilateral pleural effusions.
Antimicrobial susceptibility testing was performed by broth microdilution using a Thermo Fisher Sensititre™ GNX3F panel; colistin MIC was confirmed by reference broth microdilution. Interpretations followed EUCAST v13.0 (2025) primary breakpoints and CLSI M100 (2025) where applicable. Internal and external quality controls were within specification. Laboratory cultures soon yielded Pseudomonas aeruginosa, which was resistant to most β-lactams and quinolones. As the patient remained febrile with intensifying leukocytosis (more than 30,000/µL), therapy was switched to ceftazidime/avibactam (2.5 g) intravenously every 8 hours for 48 hours; however, no clinical stabilization was observed. When comprehensive susceptibility results were available, the isolate was resistant to cefiderocol and showed an MIC of 2 µg/mL for colistin according to new interpretive standards, effectively signifying a pan-drug-resistant phenotype (Table 2).
Antibiotic Susceptibility Testing Results for the Pseudomonas aeruginosa Isolate.

AST performed by broth microdilution; colistin MIC confirmed by reference broth microdilution; instrument platform Thermo Fisher Sensititre™ GNX3F.
Interpretations based on current Clinical and Laboratory Standards Institute (CLSI) and European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines.
High-dose intravenous colistin was administered with a loading of 9 million IU once, followed by 4.5 million IU every 12 hours over 9 days. Given progressive renal dysfunction, the colistin maintenance dose was reduced to 2.25 million IU every 12 hours on day 3, with subsequent adjustment to 2.0 million IU every 12 hours after initiation of continuous renal replacement therapy (CRRT; continuous veno-venous hemodiafiltration, effluent dose 25-30 mL/kg/h) on day 5. The addition of amikacin at 15 mg/kg intravenously once daily was limited by the patient’s deteriorating renal function and did not affect the progression of the infection. Despite in-vitro resistance to meropenem (MIC 8 µg/mL), meropenem 2 g every 8 hours via 3-hour extended infusion was combined with colistin to explore potential pharmacodynamic synergy; serial blood cultures remained positive. Repeat cultures persisted positive for Pseudomonas aeruginosa.
The patient’s respiratory failure advanced, and renal replacement therapy was initiated because of refractory metabolic derangement. Vasopressor support escalated to norepinephrine up to 0.4 µg/kg/min with adjunctive vasopressin 0.03 U/min; hydrocortisone 50 mg every 6 hours was used for catecholamine-refractory shock. By day 10 of ICU stay, it was apparent that no viable therapies were capable of containing the infection. His hemodynamic instability worsened, and following a comprehensive dialogue with his family, a joint decision was made to shift management toward comfort-based measures. He died on day 12. The immediate cause of death was refractory septic shock due to PDR Pseudomonas aeruginosa, with multi-organ failure involving renal, respiratory, and circulatory systems.
Discussion
Pan-resistant Pseudomonas aeruginosa creates a clinical environment in which standard antibiotic regimens offer no therapeutic success. Multiple factors make this organism impervious to pharmacological intervention. These factors include constitutive traits that impede drug entry into bacterial cells, efflux mechanisms that expel antibiotics before they can exert their killing effects, and widespread use of broad-spectrum antimicrobials in hospital settings that foster an ecosystem where resistance genes circulate rapidly. 6 In this case, the patient’s background of recurrent infections and repeated antibiotic exposure set a stage where a PDR strain was able to multiply, and the presence of chronic renal impairment complicated the dosing strategies for agents such as colistin and amikacin. Even cefiderocol, designed to bypass traditional resistance pathways by harnessing siderophore-mediated entry, 8 was found to be inadequate here. A colistin MIC of 2 µg/mL lies within the resistant range by EUCAST criteria and is associated with reduced probability of pharmacodynamic target attainment in critical illness, particularly when extracorporeal clearance by CRRT is present and toxicity limits dose escalation.
Reports in the literature illustrate that the efficacy of colistin may be reduced in patients with borderline MIC values of 2 µg/mL, 9 especially when complicated by the limited renal function observed in this patient. Previous analyses have noted that certain Pseudomonas aeruginosa isolates develop resistance despite combination therapy if the pathogen harbors multiple resistance determinants, including enzymatic degradation of drug molecules and structural mutations in porin channels. 10 Colistin-based regimens can still be beneficial in some XDR infections, yet the success rate drops significantly when the pathogen crosses into the PDR category, 11 as confirmed in this case. Several studies have compared ceftazidime/avibactam, cefiderocol, and older polymyxin-based regimens.12,13 While these therapies have been applied in XDR scenarios with variable degrees of effectiveness, there are accounts of treatment failures once deeper synergy is required and the organism has multiple advanced defense mechanisms.12,14,15 Other investigations contrast bacteriophage-based modalities against conventional antibiotics, finding potential in synergy but underscoring that phage formulations remain experimental.16,17 In this patient, combining meropenem with colistin despite a meropenem MIC of 8 µg/mL did not achieve clearance, in line with reports that synergy is inconsistent once multiple mechanisms of resistance co-exist.
Emerging approaches for managing PDR infections include bacteriophage cocktails that target specific Pseudomonas strains, such as those based on Myoviridae (ie, PAK_P1 and PAK_P2), which have been used on a compassionate-use basis. 18 Novel β-lactamase inhibitors are being developed and tested in combination with established agents, such as zidebactam and taniborbactam, to overcome class B and other resistant β-lactamase types.19,20 Engineered antimicrobial peptides, such as murepavadin (POL7080), have been studied for their ability to disrupt bacterial membrane formation. 21 Immunotherapy-based modalities, such as the bispecific monoclonal antibody MEDI3902, have been explored in animal models to reduce lung infection severity. 22 Nanoparticle drug delivery systems, including liposomal formulations of antibiotics, have been examined for their potential to enhance tissue penetration and limit toxicity. 23 Although these ideas are promising, most of them remain limited to clinical trials or are not yet licensed for widespread adoption. Given the regulatory timeline involved in validating new agents or methods, urgent preventive tactics are still pivotal, including early screening for resistant organisms, strict isolation protocols, thorough disinfection measures, and the measured use of existing antimicrobials. Operationally, early detection of colonization in high-risk ICU patients, device-minimization strategies, and antimicrobial gatekeeping for carbapenems and polymyxins are pragmatic priorities while novel agents remain inaccessible.
Infection control restrictions have been compared across various institutions, and programs combining universal surveillance with robust stewardship interventions can decrease the spread of resistant organisms.24,25 Yet even these combined tactics are not foolproof once a PDR organism has become entrenched. In our case, the patient’s clinical course shows that once standard regimens fail, the cascade into progressive organ dysfunction is difficult to contain. Given the continuous emergence of new resistant strains, the impetus for ongoing research cannot be overlooked. The clinical trajectory here emphasizes the narrow therapeutic window of last-line agents and the need for trial frameworks enabling emergency access to investigational modalities when standard options are exhausted.
The limitations of this case report include its nature as a single instance, which inherently restricts its generalizability. Additional limitations include the patient’s underlying comorbid conditions and chronic organ dysfunction, which made treatment of the infection extraordinarily challenging. Furthermore, the unavailability of advanced diagnostic options such as rapid genomics or phage susceptibility testing added to these constraints. Access to novel experimental agents was likewise not feasible in an urgent setting. Previous antibiotic exposure and suboptimal infection control measures in multiple healthcare facilities have likely contributed to the emergence of this intractable strain. These factors reflect the real-world obstacles faced by many centers that care for patients with prolonged complex hospitalizations.
Conclusion
Pan-resistant Pseudomonas aeruginosa can defy current antimicrobial agents to a degree that renders standard treatments ineffective, as seen in this case of a patient with a history of chronic respiratory failure, renal disease, and prior resistant infections. The failure of high-dose colistin in combination with cefiderocol demonstrates the urgent need for pioneering anti-infective strategies, and reaffirms the importance of preventing colonization and infection at every possible juncture. Progressive international cooperation and funding are critical for streamlining the approval of investigational treatments, reinforcing stewardship protocols, and adopting best-practice infection-prevention measures. This case can serve as a warning that practical solutions must be found before PDR organisms can spread further in healthcare environments.
Footnotes
Acknowledgements
The authors extend their gratitude to Medicos In Research, Nautanwa, UP, 273164, India, an initiative by Dr. Amogh Verma, for their unwavering guidance and support throughout the development of this manuscript. Their invaluable contributions provided a platform for us to learn research methodologies, collaborate effectively, expand our professional network, and gain additional insights that significantly enriched this work. We deeply appreciate their efforts in fostering an environment of academic growth and scientific inquiry.
Ethical Considerations
No formal ethical approval was required for this case report. All identification details have been anonymized to maintain confidentiality.
Consent to Participate
Written informed consent was obtained from the patient prior to death, and additional consent was secured from the next of kin after the patient’s passing for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal upon request.
Author Contributions
AS and AV: Conceptualization, validation, supervision, project administration, writing—original draft, writing—review & editing. RB: Data curation, methodology, validation, writing—review & editing. HJ: Supervision, conceptualization, visualization, writing—review & editing. GB: Investigation, resources, formal analysis, writing—review & editing. PS, AK, SL, PS, RA, and RN: writing—original draft, writing—review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in the published article. Further details can be obtained by contacting Dr. Arihant Surana (
Generative AI Use Statement
Paperpal and ChatGPT, were utilized solely for language refinement, grammar enhancement, and stylistic refinement. These tools had no role in the conceptualization, data analysis, interpretation of results, or substantive content development of this manuscript. All intellectual contributions, data analysis, and scientific interpretations remain the sole work of the authors. The final content was critically reviewed and edited to ensure accuracy and originality. The authors take full responsibility for the accuracy, originality, and integrity of the work presented.
