Abstract
Statins are a class of drugs that are vital for lowering blood lipid levels and reducing the risk of both primary and secondary cardiovascular diseases. These drugs are generally considered safe; however, some individuals may experience side effects, such as muscle pain, headache, fatigue, myopathy, and gastrointestinal disorders. One of the less frequently reported side effects of statins is visual disturbances. We report the case of a 51-year-old woman who developed persistent headache, blurred vision, and diplopia shortly after initiating Atorvastatin. These visual disturbances recurred when treatment was switched to Rosuvastatin and Lovastatin. Comprehensive neurological evaluation, including brain MRI, was unremarkable. All symptoms resolved completely after permanent discontinuation of statin therapy, indicating a rare but significant statin-associated visual adverse event. Due to the significance of reporting this complication, this study aims to present a case of a patient who experienced visual impairment associated with the use of atorvastatin, rosuvastatin, and, to a lesser extent, lovastatin.
Introduction
Statins are a class of medications widely prescribed to reduce blood lipid profiles and lower the risk of both primary and secondary cardiovascular disease by inhibiting the enzyme HMG-CoA reductase, which plays a significant role in cholesterol synthesis in the liver. 1
According to the previous studies, statins are generally considered safe; however, they can cause side effects in some people. The most frequently reported complications include: muscle pain, headache, fatigue, myopathy, and gastrointestinal disturbances. 2 Although these side effects are usually manageable, they can lead to discontinuation of treatment and may negatively affect patient outcomes.
Visual disturbances, which are among the less frequently reported and under-recognized side effects of statins, significantly impact lifestyle, quality of life, psychosocial well-being, and mobility in patients. 3 The prevalence of visual impairment associated with statins varies among different studies, with estimates ranging from 0.5% to 3%. 4 This variation may be due to differences in study populations, statin types, and methodologies. Patients have reported experiencing various forms of visual disturbances, including blurred vision, dry eyes, diplopia, changes in color perception, and even transient vision loss. 5 For instance, 1 investigation involving over 70 000 patients found that a history of statin use was associated with an approximately 40% increased risk of developing dry eye disease. 6 Some studies suggest that these disorders may be dose-dependent, with more visual impairment observed at higher doses of certain statins. 7 Studies have demonstrated that atorvastatin and simvastatin are associated with a higher risk of visual complications than rosuvastatin. 8 Risk factors that contribute to the development or exacerbation of this complication include older age, a history of eye diseases, and concomitant use of medications with visual side effects. 9 Managing this side effect is challenging due to concomitant diseases and medications; the risks associated with polypharmacy and comorbidities may further complicate clinical management. 10 To reduce the risk of statin-related visual impairment, strategies such as regular eye examinations, educating patients about potential side effects, and considering alternative lipid-lowering therapies are recommended. 11 Given the significance of this issue, we present a case involving a patient who experienced visual disturbances after using atorvastatin, rosuvastatin, and, to a lesser extent, lovastatin.
Case Presentation
A 51-year-old woman with no significant past medical history presented to the clinic due to severe anxiety related to non-significant proteinuria observed in routine tests. Based on her condition, Losartan 25 mg daily and Atorvastatin 10 mg daily were prescribed. At her initial visit, the patient’s weight was 84 kg; however, she lost 2 kg after following a dietary regimen and exercise program.
Approximately 2 months later, she returned to our facility, complaining of headaches, blurred vision, and double vision. These symptoms were persistent, with no identifiable factors that relieved or exacerbated them. Consequently, atorvastatin was replaced with rosuvastatin 10 mg daily, and spironolactone 25 mg (half a tablet in the morning) was added to her treatment plan. However, the patient continued to experience similar symptoms, which resolved upon discontinuation of the medication.
A trial of lovastatin 20 mg daily was then considered; however, the patient experienced visual disturbances again, although less severe, leading to discontinuation of this statin as well. The patient denied any history of head trauma, preexisting visual disturbances, recent infections, headaches, migraines, burning dry eyes, or slurred speech.
At the time of examination, vital signs were within normal limits. Laboratory investigations revealed a normal creatinine level (0.85 mg/dl), an erythrocyte sedimentation rate (ESR) of 5 mm/h, and a C-reactive protein (CRP) level of 1 mg/dl. All other laboratory findings were unremarkable. A comprehensive neurological and psychiatric evaluation conducted by a neurologist displayed normal findings. Brain CT and MRI scans revealed no acute pathology. The patient’s weight loss was not significant, and anxiety significantly decreased following the receipt of appropriate medication after the first visit to the doctor. Apart from the mentioned medications, no other drugs were taken, and symptoms of decreased vision and blurriness only appeared after the resumption of statin therapy.
Although a formal ophthalmological consultation was not obtained, initial clinical assessment revealed slow extraocular muscle movements accompanied by diplopia and blurred vision. Ultimately, statin therapy was permanently discontinued. The patient’s regimen was maintained with losartan alone, and ophthalmological follow-up was recommended.
Patient consent for the publication of this information has been obtained.
Discussion
A clear temporal relationship was observed between the administration of each statin and the onset of visual disturbances. The causality assessment was performed using the Naranjo Adverse Drug Reaction Probability Scale. 12 This 10-item questionnaire standardizes the clinical assessment of adverse drug reactions by evaluating factors such as the temporal sequence of the reaction, improvement upon drug discontinuation (dechallenge), and recurrence upon re-administration (rechallenge). According to the scale’s interpretation, the patient’s total score was 8, indicating a “probable” causal relationship between the statin therapy and the visual disturbances_ detailed results of the Naranjo scale assessment are presented in Table 1.
Adverse Drug Reaction Probability Scale (Naranjo Scale) Assessment.
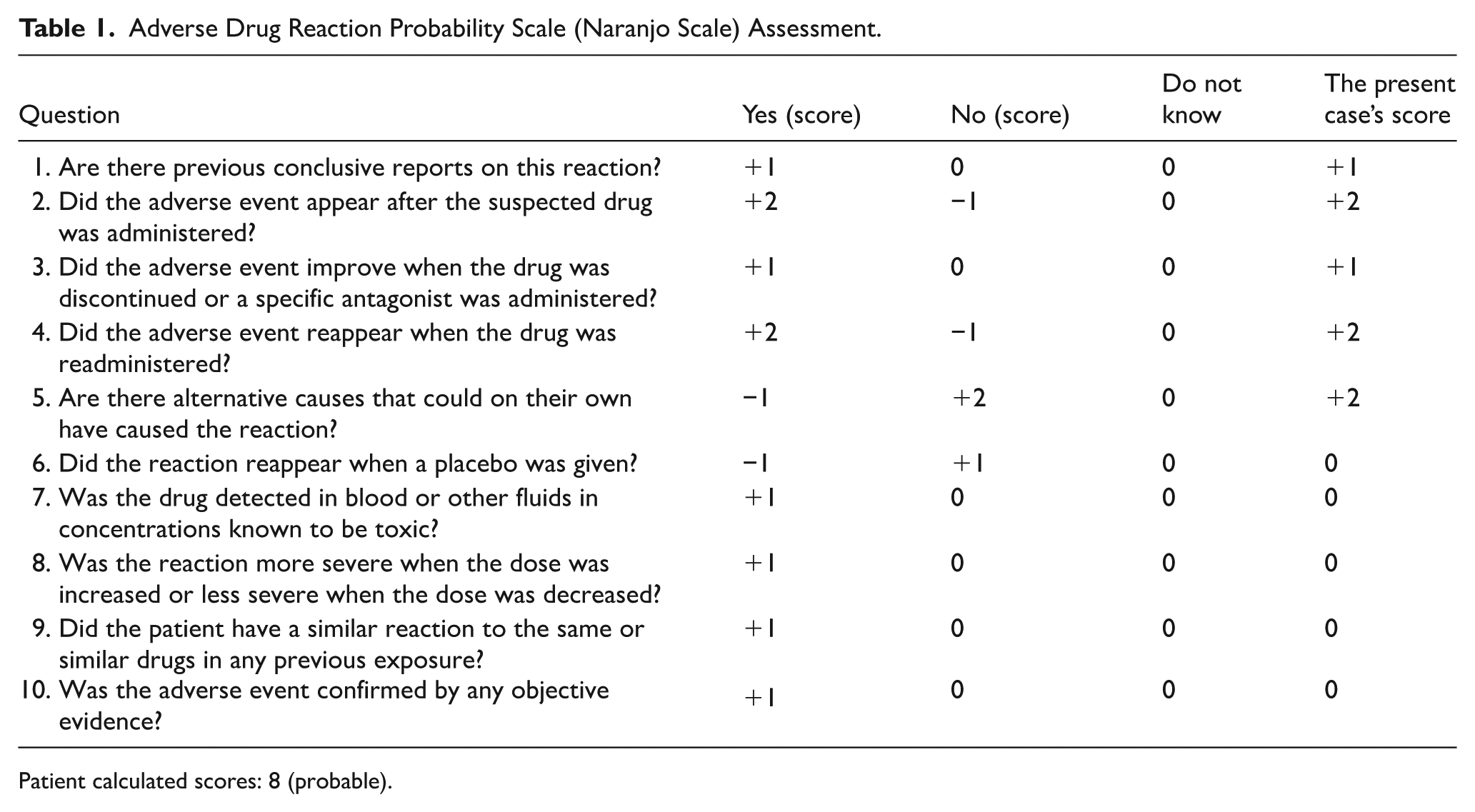
Patient calculated scores: 8 (probable).
Complete resolution of symptoms after stopping statin use, followed by a recurrence upon re-initiating the medication, suggests a probable causal relationship between statin use and visual disturbance. This dechallenge-rechallenge pattern strongly indicates that the patient’s visual disturbance was a drug-induced adverse effect of statins. 13 This case underscores the importance of physicians being aware of rare ocular side effects of statins. Statin generally has a high safety profile, and many patients tolerate it well. 14 Nevertheless, some patients experience several side effects, which are mostly mild and manageable. According to existing reports, the incidence of visual disturbances during statin therapy is low; in most studies, less than 1% to 2% of patients experienced such effects. 15 While this occurrence is rare, the high number of statin users in the general population means that even rare side effects can affect a substantial number of patients. 13
Diplopia (double vision) is another significant symptom, often presenting in the form of extraocular muscle palsy. Ptosis (drooping of the eyelid) and restricted ocular motility (ophthalmoplegia) have been observed alongside diplopia. Although changes in color perception and transient visual loss are infrequent and their mechanisms are not well understood, they have been mentioned in isolated case reports. Overall, most of these visual disturbances are reversible and resolve upon discontinuation of the drug. 13 In our patient’s case, the ocular symptoms improved after the statins were withdrawn. Their known actions on other tissues may explain the mechanism behind statin-induced ocular side effects.
Growing evidence suggests that systemic vascular instability can affect ocular structures. A recent study in hypertensive patients demonstrated a significant association between short-term blood pressure variability and reduced choroidal thickness, supporting its role as a marker of systemic vascular injury. These findings suggest that statin-related visual disturbances might involve similar microvascular mechanisms, possibly through transient alterations in ocular perfusion leading to structural and functional changes. 16 By inhibiting HMG-CoA reductase, statins not only reduce cholesterol synthesis but also affect the biosynthesis of other important compounds, one of which is coenzyme Q10 (ubiquinone). 5 Coenzyme Q10 is essential for mitochondrial function and plays a crucial role in the electron transport chain. A decrease in its levels can lead to mitochondrial dysfunction. There is evidence indicating that reduced coenzyme Q10 levels and mitochondrial damage in muscles contribute to the pathogenesis of statin-induced myopathy. 17 Therefore, it can be hypothesized that weakness or inflammation of the ocular muscles may potentially occur through the same mechanism. In fact, the well-known adverse effect of statin-induced myopathy may also affect the extraocular and eyelid muscles, resulting in diplopia or ptosis. 6
Fronkfelder et al conducted a study involving 256 cases of ocular adverse events associated with statin use. They found evidence of myositis (inflammation) in the extraocular muscles or the levator palpebrae superioris muscle in these patients, identifying this as the most likely cause of statin-related diplopia and ptosis. 6 Furthermore, MRI imaging in 1 report confirmed inflammation of the extraocular muscles in a patient experiencing statin-induced Diplopia. 15 Notably, in most patients with statin-related diplopia or ophthalmoplegia, symptoms completely resolved upon discontinuation of the drug, and in some cases, they reappeared with statin re-administration. 6
Another possible explanation for the pathophysiology of statin-induced diplopia is the induction of an autoimmune neuromuscular disorder, such as myasthenia gravis. In recent years, several cases of generalized or ocular myasthenia gravis have been reported following the initiation of statin therapy. 18 Furthermore, in patients already diagnosed with myasthenia gravis, statins may exacerbate muscle weakness. 19 It should be emphasized that statin-induced myasthenia is rare, and most patients with myasthenia gravis tolerate statin therapy without major issues. 20
In the current patient, there were no signs of generalized muscle involvement or ptosis, and symptoms were limited to impaired eye movement. Although specific tests for myasthenia (such as acetylcholine receptor antibodies and edrophonium test) were not conducted in this case, the transient and reversible nature of the symptoms following drug discontinuation is more suggestive of a temporary toxic-inflammatory myopathy due to statins rather than a sustained autoimmune process. Additionally, an examination of predisposing factors indicates that certain patients may be more susceptible to such adverse effects than others. The average age of patients experiencing statin-induced ocular disorders has been relatively high in reported cases, and older age can generally be considered a risk factor. A possible explanation may be the greater sensitivity of periorbital muscles to metabolic stress or the presence of comorbid conditions found in older individuals. Moreover, the presence of diabetes or vascular problems may also increase the risk of statin-induced muscular and ocular side effects. 6 In a study conducted by Fraunfelder, 9 out of 256 patients had diabetes, and 5 were simultaneously taking gemfibrozil. It is well established that the concomitant use of fibrates, such as gemfibrozil, alongside statins increases the risk of myopathy and rhabdomyolysis. 6 This drug interaction can elevate statin levels, increasing the risk of muscular side effects, including those affecting the ocular muscles. In addition, a history of ocular disorders (such as subclinical ocular myasthenia, ptosis, or ocular circulatory problems) can make patients more vulnerable to the adverse visual effects of medications. While our patient had no prior history of visual or neurological disorders, it is important to note that in other cases, underlying ocular conditions may make statin tolerance more difficult for the visual system. 14 The research also demonstrates the difference in the likelihood of these adverse effects among various types of statins. These differences stem from their pharmacological properties, including whether they are lipophilic or hydrophilic, their potency in enzyme inhibition, and their dosage.
Atorvastatin and simvastatin are classified as lipophilic statins, which means they have greater tissue penetration capabilities and can cross the blood-retinal barrier. In contrast, rosuvastatin and pravastatin are hydrophilic and are less likely to enter extrahepatic tissues. Mizranita et al revealed that the proportion of ocular adverse events to total reported adverse effects was higher with atorvastatin compared to other statins (2.1% vs lower rates for others).15,21 Therefore, it can be hypothesized that lipophilic statins may have a greater potential for causing ocular or central nervous system effects. Indeed, some studies have reported a higher incidence of visual disturbances with atorvastatin and simvastatin compared to rosuvastatin. 15 In our patient, however, despite the theoretical expectation of better tolerability with rosuvastatin, diplopia occurred with this medication as well. However, the severity of symptoms on rosuvastatin did not differ from that on atorvastatin and only slightly diminished with lovastatin (an older and less potent statin). This finding suggests that in specific sensitive individuals, all drugs in this class – regardless of pharmacological differences– can trigger adverse effects. In such cases, it may be necessary to completely discontinue the use of statins and explore alternative therapies.
Fortunately, high-quality studies (primarily randomized clinical trials) have so far revealed no evidence of severe ocular damage caused by statins. In a recent systematic review analyzing a collection of randomized trials, it was concluded that statins do not hurt the eye’s lens and are unlikely to increase the risk of cataracts. In fact, several studies have provided evidence suggesting a protective role for statins against certain ocular diseases. For example, some observational studies have associated the use of statins with reduced incidence or slower progression of cataracts. On the other hand, its anti-inflammatory and antioxidant properties may be beneficial for the lens. Moreover, some population-based studies have suggested that statins may reduce the risk of age-related macular degeneration (AMD) and severe diabetic retinopathy. 22 However, these findings are not yet conclusive, and some other studies have produced conflicting results.23,24 The presence of such data highlights that statins can have complex effects on the eye – potentially both beneficial (in retinal and vascular diseases) and harmful (involving the ocular muscles or optic nerves). It depends on the patient’s conditions and the drug characteristics. Physicians should be aware that any new visual complaint in patients taking statins, though rare, may be related to the drug. Initially, more common causes of visual disturbances (such as refractive errors, cataracts, glaucoma, retinal disease, and central nervous system lesions) should be investigated. If no significant findings suggest these conditions, the possible role of statins should be considered.
As observed in our patient, drug discontinuation can serve as both a diagnostic and therapeutic trial; if symptoms improve upon stopping the statin and recur upon reintroduction, a drug-induced adverse effect is almost confirmed. 13 In these situations, the next step is to manage the patient’s hyperlipidemia. One option is to try a different statin (either switching to an equivalent lower dose or one that has different pharmacologic properties). However, if all statins cause problems, alternative non-statin lipid-lowering agents should be used. These alternatives include ezetimibe, bile acid sequestrants, or, in high-risk cases, PCSK9 inhibitors. In our patient, after discontinuation of the statin, lipid-lowering therapy was switched to ezetimibe or other agents (as recommended) in order to control the patient’s lipid profile without recurrence of visual symptoms. Preserving visual function in patients at risk of statin-induced visual impairment is of particular importance. Recent innovations in ophthalmic surgery further highlight ongoing efforts to enhance visual outcomes through minimally invasive and well-centered intraocular lens implantation techniques (Yamane, Agarwal, Full-reverse). 25 Furthermore, periodic ophthalmologic examinations in patients taking statins, particularly the elderly or those with preexisting ocular conditions, may help detect any changes early.
Conclusion
This report is one of the few existing case reports concerning statin-induced visual disturbances. It confirms that although ocular side effects from statins are rare, they are real and can involve several drugs within this class in the same patient. The likely mechanism is primarily related to weakness or inflammation of the extraocular muscles, which aligns with the known myopathic nature of statins. Additionally, in some patients, genetic predisposition or autoimmune responses_ such as myasthenia_ may contribute to these issues. The practical message for physicians is to consider statins as a potential cause in patients with unexplained visual complaints during treatment and, if necessary, temporarily discontinue the drug and monitor the patient’s condition. Increased awareness of this rare adverse effect within the medical community can lead to timely diagnosis of similar cases, avoid costly diagnostic interventions, and prevent continuation of a potentially harmful treatment. In conclusion, the risk-benefit balance still favors the significant cardiovascular benefits of statins. Therefore, identifying and managing rare side effects such as visual disturbances can improve treatment adherence and ensure the ongoing safe use of these valuable medications.
Footnotes
Consent to Participate
Written informed consent was obtained from patient prior to study initiation.
Consent for Publication
The patient provided informed consent for publication of the medical details.
Author Contribution
AA: Conceptualization; investigation; project administration; MM: investigation; writing – original draft; writing – review and editing; MP: investigation; writing – original draft; writing – review and editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All the available data are provided in this submission.
