Abstract
Background:
Aconitine, a highly toxic diterpenoid alkaloid derived from the Aconitum genus, continues to be utilized in traditional Chinese medicine. Aconitine poisoning is common in China and often causes malignant arrhythmias and cardiac arrest, with a mortality rate of approximately 15.1%. Using venous-arterial extracorporeal membrane oxygenation (VA-ECMO) in this context has been rarely reported, and associated microcirculatory changes have not been described.
Case Report:
We present a case of a 59-year-old man who ingested a topical medicinal wine prepared from Aconitum brachypodum Diels. Despite the conventional treatment administered, the patient developed a cardiac arrest 2 hours after admission. VA-ECMO was promptly initiated alongside repeated cardiopulmonary resuscitation and electrical defibrillation. After 6 hours of VA-ECMO initiation, the heart rhythm of the patient stabilized to sinus without further malignant arrhythmias. Microcirculatory monitoring was performed using sublingual microcirculation during VA-ECMO support, which was used for a total of 46 hours. The patient was transferred out of the intensive care unit (ICU) after 7 days of hospitalization and was eventually discharged without any sequelae.
Conclusion:
Early initiation of VA-ECMO support may help stabilize cardiac electrical storms in patients with aconitine poisoning. Sublingual microcirculation monitoring may play a role in predicting VA-ECMO weaning success and patient but this requires further investigation.
Introduction
Aconitum brachypodum Diels is a species of the Aconitum genus, with aconitine as its primary active compound, and is predominantly found in the Sichuan and Yunnan provinces of China. Aconitine is extensively used to treat bruises and injuries in traditional Chinese medicine and in medicinal wine. However, it possesses marked cardiotoxicity and neurotoxicity properties, with cardiac arrhythmia being the most frequent clinical manifestation. Malignant arrhythmia is also the main cause of death, with a mortality rate of 15.1%.1,2
Currently, there are no specific antidotes, and treatment is mainly symptomatic, involving pharmacological therapy and organ support such as blood purification and venous-arterial extracorporeal membrane oxygenation (VA-ECMO).
VA-ECMO is an advanced life-support technique that temporarily replaces cardiopulmonary function and is commonly used to provide essential respiratory and circulatory support in patients with impaired cardiac hemodynamics. 3 Given that the pathophysiological process of acute poisoning is reversible, VA-ECMO has been increasingly used to rescue patients experiencing circulatory collapse or cardiac arrest secondary to drug or alkaloid poisoning. 4 Therefore, researchers have attempted to monitor microcirculatory changes during therapy and to guide VA-ECMO withdrawal and predict patient outcomes. However, the findings remain heterogeneous and controversial.5,6
We present a case of severe aconitine poisoning due to the consumption of medicinal wine made from A. brachypodum Diels. The patient presented with life-threatening malignant cardiac arrhythmia and cardiac arrest, and was successfully treated with VA-ECMO, hemoperfusion, and other therapies.
Case Presentation
A 59-year-old man, previously in good health, was admitted to the Emergency Department 3 hours after accidentally taking medicinal wine prepared with A. brachypodum Diels (approximately 200 mL). On arrival, the patient had numbness, palpitations, and chest tightness. Physical examination revealed somnolence, a heart rate of 193 beats/min., and a blood pressure of 51/33 mmHg. Electrocardiograph (ECG) revealed bidirectional ventricular tachycardia. The patient was suspected of having aconitine poisoning. After initial management, including blood pressure support, antiarrhythmia therapy, and gastric lavage, the patient was urgently transferred to the intensive care unit (ICU).
Following ICU admission, the patient developed alternating episodes of various malignant arrhythmias, including atrial fibrillation, ventricular tachycardia, and ventricular fibrillation (Figure 1), resulting in cardiogenic shock. Lidocaine and amiodarone were administered, along with synchronized electrical cardioversion and defibrillation. Hemoperfusion was initiated to eliminate toxins, and intermittent chest compressions were performed to maintain adequate perfusion. Blood samples were collected for toxin analyses. The patient suffered cardiac arrest 2 hours after admission. Endotracheal intubation and mechanical ventilation were then performed. Blood pressure was supported with high-dose vasopressors, and VA-ECMO was initiated alongside repeated chest compression and continuous defibrillation.

ECG results: (A) atrial fibrillation (in the emergency department); (B) polymorphic ventricular tachycardia (before VA-ECMO initiation); (C) ventricular premature beat (VA-ECMO initiation for 6 hours); (D) sinus rhythm (after VA-ECMO withdrawal).
VA-ECMO was initiated within 30 minutes (initial parameters: blood flow, 4 L/min; pump speed, 3770 rpm; air flow, 2 L/min). Subsequently, blood flow was maintained between 3 and 3.5 L/min. Despite VA-ECMO support, repeated defibrillation and chest compression were required. Fortunately, no further lethal arrhythmias occurred after 6 hours. ECG at this stage demonstrated a sinus rhythm with occasional premature ventricular contractions. Toxicology testing confirmed aconitine.
Management and monitoring during VA-ECMO treatment: (1) Anticoagulation: We adopted a low-heparin anticoagulation strategy, maintaining activated partial thromboplastin time at 35.9 to 47.7 seconds. (2) Ultrasound: Real-time monitoring of tube position, ejection fraction (EF), and velocity-time integral (VTI; Table 1). (3) Microcirculation: Monitoring sublingual microcirculation, the indexes including microvascular flow index (MFI), total small vascular density (TSVD), perfused small vascular density (PSVD); proportion of perfused vessels (PPV; Table 1 and Figure 2) and lactate levels; (4) Internal environment: Blood gas analysis revealed that compensatory metabolic acidosis was corrected 12 hours after VA-ECMO initiation. The initial serum potassium level was 3.36 mmol/L, which returned to normal within 2 hours. (5) Hemoperfusion: After 4 rounds of hemoperfusion, the repeat toxicity test was negative, and hemoperfusion was stopped.
Macrocirculatory and Microcirculatory Monitoring Indicators After VA-ECMO Initiation.

Abbreviations: EF, ejection fraction; MAP, mean arterial pressure; MFI, microvascular flow index; PPV, proportion of perfused vessels; PSVD, perfused small vascular density; TSVD, total small vascular density; VA-ECMO, venous-arterial extracorporeal membrane oxygenation; VIS, vasoactive inotropic score; VTI, velocity-time integral.
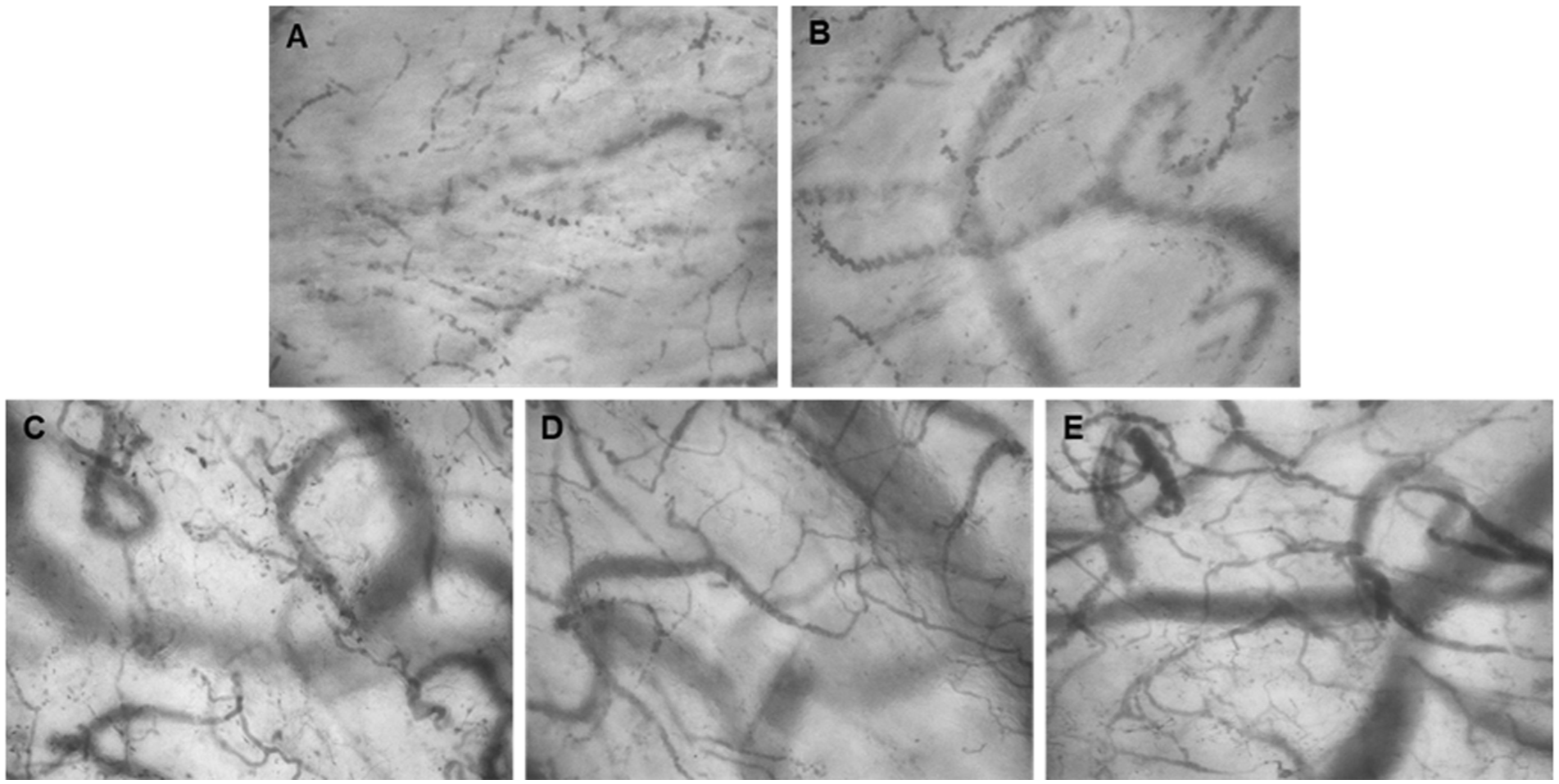
Changes in sublingual microcirculation. VA-ECMO initiation for (A) 4 hours; (B) 12 hours; (C) 24 hours; (D) 45 hours; (E) VA-ECMO withdrawal for 24 hours.
Sublingual microcirculation: The MicroSee V100 Sublingual Microcirculation Imaging System Management Software (MicroSee V100; Guangzhou Medsoft Intelligent Technology Co., Ltd., Guangzhou, China) was used to capture and analyze images. The most commonly selected site at present was the sublingual area. Two trained operators observed at least 3 sublingual regions for a minimum of 4 seconds after cleaning the mouth and then analyzed the images.
VA-ECMO withdrawal: VA-ECMO was successfully discontinued after 46 hours. The decision was based on the stabilized hemodynamics of the patient, cardiac ultrasound parameters (VTI, 16 cm; EF, 54%), and normal lactate levels. On day 7 of hospitalization, the patient was transferred to the ICU and later discharged from the hospital without any residual complications.
Discussion
In this case, the patient consumed medicinal wine prepared with A. brachypodum Diels, resulting in severe aconitine poisoning. Aconitine can cause arrhythmia and cardiac arrest, with ventricular arrhythmia being the most common, accounting for approximately 78.57%. 7 VA-ECMO is commonly used to provide circulatory support to patients with cardiac arrest. 8 However, VA-ECMO treatment for aconitine poisoning has mostly been reported in case reports, as severe cases are relatively rare. In the present case, VA-ECMO was administered because of severe circulatory failure. The electrical storm in the heart of the patient subsided after VA-ECMO support. Zhao reported a case with aconitine toxicity and cardiac arrest treated with prompt VA-ECMO, and the patient reverted to sinus rhythm after 1 hours of support. 9 Another patient with refractory arrhythmia and cardiogenic shock was discharged successfully after <48 hours of VA-ECMO support. 10 These reports demonstrate the therapeutic potential of VA-ECMO in patients with aconitine poisoning. Consequently, early initiation of VA-ECMO may be critical in controlling cardiac electrical storms.
Macrocirculation and microcirculation are interdependent. In patients with cardiogenic shock, both impaired macrocirculation and microcirculatory dysfunction are associated with mortality and poor prognosis. Furthermore, decoupling between them may occur.11,12 Additionally, to relevant macrocirculatory indicators, microcirculation was also monitored.
Sublingual microcirculation can be successfully visualized using handheld microscopy. Sublingual microcirculation and lactate levels were monitored in this case to assess microcirculatory characteristics and trends. Recently, there have been no reports analyzing the microcirculation in patients with aconitine toxicity treated with VA-ECMO. Sublingual microcirculation combined with ultrasound were used to guide vasoactive drug adjustment, and the EF rised to 53% within 12 hours of VA-ECMO initiation. By comparison, another patient with ultrasound guidance alone took 60 hours to reach the same EF. 13 Comparisons across individual cases could have occasionality, but these findings still suggested a potential benefit from sublingual microcirculation monitoring.
As the condition of the patient improved, PSVD increased while lactate levels decreased, indicating recovery of microcirculatory function. Chommeloux et al also observed that 24 hours after the initiation of VA-ECMO, lactate levels normalized and sublingual microcirculatory parameters, such as MFI and PSVD, demonstrated an improvement trend. 5 Furthermore, a report by Li revealed significant improvements in sublingual microcirculation indices (PSVD, PPV, and MFI) in cardiogenic shock survivors. 14 Another study demonstrated that survivors exhibited significantly higher values for TSVD, PSVD, and PPV than non-survivors 24 hours post-VA-ECMO. 15 These studies suggest that sublingual microcirculation could be used to predict the outcome for patients undergoing VA-ECMO. However, whether this conclusion applies to patients with cardiogenic shock caused by poisoning requires further research.
Although low-dose pressor medications were used, the overall condition and microcirculation of the patient stabilized, allowing for safe discontinuation of ECMO. Whether sublingual microcirculation reliably predicts successful weaning from ECMO remains controversial. An early report suggested that TSVD >14.3 mm/mm2 could predict successful weaning when VA-ECMO flow was reduced by 50% for patients with cardiogenic shock. 16 The 2018 consensus stated that MFI >2.9 scores were considered normal, whereas MFI <2.6 scores indicated microcirculatory dysfunction. 17 In this case, the MFI score of the patient stabilized at approximately 2.9 during weaning. However, in pediatric patients, microcirculation monitoring at the weaning phase did not predict the ECMO outcomes. 18 The differences between the findings could be due to variations in the study populations. Therefore, more evidence is needed for confirmation.
In conclusion, for patients with severe cardiac electrical storms, early VA-ECMO support combined with recovery of microcirculation may improve the prognosis. However, whether sublingual microcirculation can serve as a predictive indicator for ECMO weaning and prognosis in aconitine poisoning patients requires further investigation.
Footnotes
Acknowledgements
Special appreciation to Dr Fang Xu, MD and Dr Minkang Guo, MD who provided clinical guidance to ensure accuracy of the report and final draft.
Ethical Considerations
Writtem Informed consent form was obtained from the patient for publication of this case report.
Author Contributions
Yu Yang and Xuan Yao contributed to performing data curation and drafting the manuscript. Bei Ma performed the analysis and partly the manuscript preparation. Xi Chen participated in revising the manuscript and providing important intellectual content. All authors have provided approval of the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing Municipal Health Commission, under Grant number 2024WSJK022.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All available data are provided in this submission.
