Abstract
Background:
Pseudoaneurysm of the superior lateral genicular artery (SLGA) is a rare vascular complication following total knee arthroplasty (TKA), with an incidence ranging from 0.03% to 0.2%. Most cases occur in the immediate postoperative period while delayed presentation months after surgery can be often misdiagnosed as infection or hemarthosis.
Case Presentation:
A 62-year-old male smoker with previous TKA presented to the emergency department with knee pain and a palpable pulsatile mass on the lateral side of the right knee. Clinical evaluation with doppler ultrasonography and CT scan showed the presence of a 1.5 cm pseudoaneurysm of the SLGA. Initial conservative management with manual compression proved ineffective thus given the high risk of rupture, definitive surgical exclusion was performed with no post-procedural complications.
Conclusion:
Delayed SLGA pseudoaneurysms typically present with nonspecific symptoms like pain, swelling and limited ROM. Diagnosis relies on imaging while surgical ligation proved to be an effective treatment. Pseudoaneurysm should be considered in post-TKA patients with unexplained knee swelling and pain, even months after surgery. Early diagnosis through second level imaging, multidisciplinary collaboration and timely surgical intervention are crucial for preventing complications.
Keywords
Introduction
Total knee arthroplasty (TKA) is among the most frequently performed orthopedic procedures, with annual volumes approaching 500 000 cases in the United States alone. Global projections indicate continued growth to an estimated 3.48 million procedures by 2030, primarily driven by aging demographics and the rising prevalence of osteoarthritis. 1 Over the past several decades, advances in surgical techniques, implant design, and perioperative care have contributed to excellent long-term outcomes with long-term implant survival exceeding 90% at 15 years in contemporary series. 2 Nevertheless, postoperative complications remain a major cause of morbidity, reoperation, and healthcare burden. 3
The most common causes of TKA failure include periprosthetic joint infection, aseptic loosening, instability, bearing surface wear, and periprosthetic fractures.4,5 Beyond these, mechanical and infectious etiologies alongside systemic and local medical complications may occur. Thromboembolic events, such as deep vein thrombosis and pulmonary embolism, are well-recognized risks in the early postoperative period. 6
In addition to these systemic vascular complications, patients may also experience local neurovascular complications, although less frequent, are potentially limb- or life-threatening and may arise from direct arterial injury, intimal dissection, thrombosis, embolization or secondary mechanisms, including vasospasm and vasoconstriction associated with tourniquet application, retractor placement or periarticular dissection.7,8
Notably, functional outcomes after TKA may be further compromised by postoperative structural or mechanical impairments, highlighting the multifactorial nature of postoperative morbidity. Postoperative stiffness, malalignment and extensor mechanism disruption represent additional significant sources of functional impairment. 9 Stiffness can result from arthrofibrosis, inadequate rehabilitation or technical factors such as improper component positioning, 10 and it may severely restrict ROM, potentially hindering recovery and patient satisfaction. 11 Malalignment in the coronal, sagittal or rotational plane not only compromises joint kinematics, but also predisposes to accelerated polyethylene wear and early prosthetic loosening. 12 In addition, disruption of the extensor mechanism, including quadriceps tendon rupture, patellar fracture or patellar tendon rupture, though uncommon, can have profound clinical consequences, often resulting in substantial functional limitation, impaired ambulation, and the potential need for complex revision or reconstructive procedures. 13
Although vascular complications following TKA are rare, with an incidence of 0.03% to 0.2%, 14 their consequences can be severe, with amputation reported in up to 28% of affected patients depending on delays in diagnosis and intervention. 15 The popliteal artery, due to its anatomical proximity to the posterior capsule, is most commonly affected. 16 In contrast, vascular injuries to the genicular branches are exceedingly rare and largely reported in case studies. 17 Among these, pseudoaneurysms involving the superior genicular arteries, particularly the SLGA, represent an exceptional occurrence. 18
We report a 62-year-old male who developed a delayed SLGA pseudoaneurysm 7 months following total knee arthroplasty. This case highlights the subtle and delayed clinical presentation of SLGA pseudoaneurysm, the associated diagnostic challenges, and the critical role of early multidisciplinary management, involving orthopedic, vascular, and radiology teams, in preventing potentially limb-threatening complications such as rupture, hemorrhage, or ischemia. Documentation and reporting were conducted in accordance with the CARE guidelines to ensure comprehensive and transparent presentation of clinical data, interventions, and outcomes
Case Presentation
A 62-year-old Caucasian male smoker (20 pack/year) with no known comorbidities complained of severe right knee pain for about 1 year with range of motion (ROM) limitation. The patient worked as a truck driver and manual laborer. He was diagnosed with severe right knee osteoarthritis, associated with 11° valgus deformity and treated in mid-2024 with TKA (Figure 1A-C).

(A) Anterior-posterior (AP) X-Ray of right knee showing severe osteoarthritis. (B) Lateral X-Ray of the knee revealing joint space narrowing and patello-femoral degeneration. (C) Weight-bearing full-length lower extremities X-Ray demonstrating 11° valgus deformity of the right lower limb.
The surgical procedure was performed under spinal anesthesia, using a midvastus approach without a tourniquet. Cemented components (Zimmer Biomet: Persona femoral component #11 CR, tibial component #H, and a #10 UC polyethene articular surface) were implanted (Figure 2A-C).

(A) X-Ray axial view of the patella, showing correct alignment between the femoral component and the posterior facet of the patella. (B) AP X-ray of the right knee demonstrating TKA with proper positioning of the cemented femoral and tibial components. (C) Lateral X-ray of the same knee revealing the posterior profile of the prosthetic components and fixation elements.
At the end of surgery, despite no evidence of macroscopic signs of hemorrhages or vascular injuries, an intra-articular drain was placed that was removed the day after surgery with no complications. The patient underwent a structured postoperative rehabilitation program, including daily supervised physiotherapy focusing on early quadriceps activation, progressive range-of-motion exercises, and gait training, supplemented by cryotherapy to control swelling. Upon discharge on postoperative day 6, the patient was in good general condition, with stable vital signs and a hemoglobin level of 9.3 g/dL, and was provided with a home-based continuation of the rehabilitation protocol. Outpatient follow-up visits were scheduled at 1, 3, and 6 months postoperatively to monitor recovery, functional outcomes, and wound healing.
At 1 month postoperatively, the patient exhibited a ROM of 90°-5°-0°, with mild residual joint effusion. By 3 months, ROM had increased to 120°-0°-0°, and the surgical incision showed progressive healing without evidence of infection or delayed closure aligning with anticipated postoperative improvement. Functional recovery was evaluated using both the Knee Society Score (KSS) and the Forgotten Joint Score (FJS). At 1 month, the KSS showed a clinical score of 75 and a functional score of 72, reflecting good pain control, preserved stability, and partial restoration of walking and stair-climbing ability. By 3 months, the KSS improved to a clinical score of 92 and a functional score of 90, indicating substantial gains in pain relief, ROM, stability, and activities of daily living. Parallel assessment with the FJS-12 demonstrated an increase from a raw score of 70 at 1 month to 75 at 3 months, reflecting progressive reduction in joint awareness, enhanced joint-related quality of life, and improved patient satisfaction.
Approximately 7 months postoperatively, the patient presented to the emergency department with intermittent acute swelling, redness, and pain in the right knee, with functional limitation persisting for about 7 days. Clinical examination revealed no abnormalities in the other parts of the limb, with no evidence of thigh or calf swelling, edema, or tenderness. After initial laboratory investigations (reported in Table 1) the patient underwent knee plain radiographs (Figure 3) that showed joint effusion with no abnormalities in the positioning or integrity of the TKA components.
Initial Laboratory Investigations at Emergency Department Presentation (7 Months Post-TKA).


(A) AP and lateral projections. (B) X-Rays negative for osteolysis, aseptic loosening and periprosthetic fractures, yet showing joint effusion (C) as highlighted by the yellow rectangle.
Orthopedic evaluation was then performed. Given the combination of pain, swelling, and elevated inflammatory markers, the differential diagnosis initially prioritized periprosthetic joint infection versus hemorrhagic synovitis. Diagnostic arthrocentesis was subsequently attempted to rule out the presence of infection; however, aspiration was unsuccessful with only scant blood obtained, presumably due to the presence of intra-articular clot formation hindering synovial fluid retrieval. As the patient remained clinically stable and afebrile, he was discharged with instructions to return for reassessment. In light of these findings, repeat laboratory testing and imaging were scheduled within 5 days. Supportive therapy was prescribed, including
Three days following discharge, the patient re-presented to the emergency department with persistent knee pain and the new finding of a pulsatile mass over the lateral aspect of the right knee. Laboratory evaluation revealed a decrease in hemoglobin (11.2 g/dL) and partial normalization of C-reactive protein (14 mg/L) (Table 2).
Blood Tests at the Time of the Patient’s Second Emergency Department Admission Showing a Decrease in Hemoglobin Levels and Improvement of C-Reactive Protein (CRP) Values.

At this stage, the differential diagnosis was narrowed to hemorrhagic synovitis or vascular lesions. The pulsatile nature of the mass, coupled with the history of TKA, raised early suspicion for a vascular etiology, further reinforced by the lack of systemic symptoms, absence of inflammatory signs at the wound site and negative radiological findings.
Accordingly, a vascular surgery consultation was obtained. Duplex Doppler ultrasonography (DUS) of the lower limb (Figure 4A) identified a 1.5 cm saccular lesion arising from the SLGA. The lesion was sharply circumscribed and focal, located immediately adjacent to the femoral epicondyle. Color Doppler imaging indicated a characteristic swirling “yin-yang” pattern within the sac, and spectral Doppler revealed the classic “to-and-fro” flow at the neck, consistent with bidirectional blood movement between the artery and the cavity. On further evaluation, the parent vessel showed no diffuse dilatation, and all 3 arterial wall layers were preserved outside the sac, indicating a contained disruption of the intima and media rather than a true aneurysm involving the full vessel wall. These findings were subsequently validated by contrast-enhanced CT angiography (CTA), which additionally identified intra-articular hemarthrosis, evidenced by hyperdense fluid accumulation within the joint space, and depicted a saccular outpouching with a narrow neck, clearly separated from the parent vessel, without involvement of the surrounding arterial wall, thereby confirming the diagnosis of an iatrogenic pseudoaneurysm (Figure 4B).
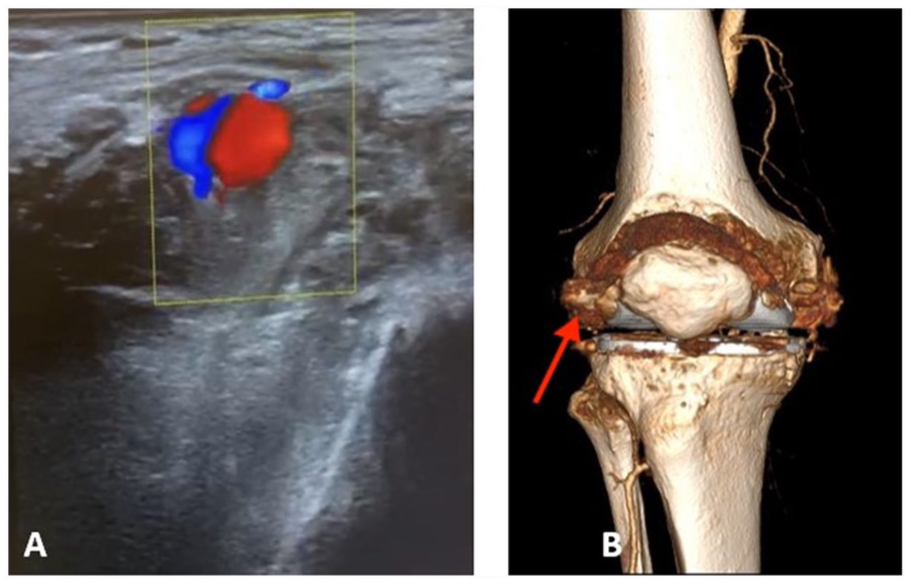
(A) On DUS, the yin-yang sign indicates bidirectional flow due to the swirling of blood within the true or false aneurysm suggestive for SLGA. (B) 3D CTA illustrating the pseudoaneurysm of the SLGA (red arrow).
Immediate manual compression was attempted to exclude the vascular lesion; however, this maneuver proved ineffective. As a result, the following day, under general anesthesia, a 3 cm lateral capsulotomy was performed to access the region of the SLGA (Figure 5).
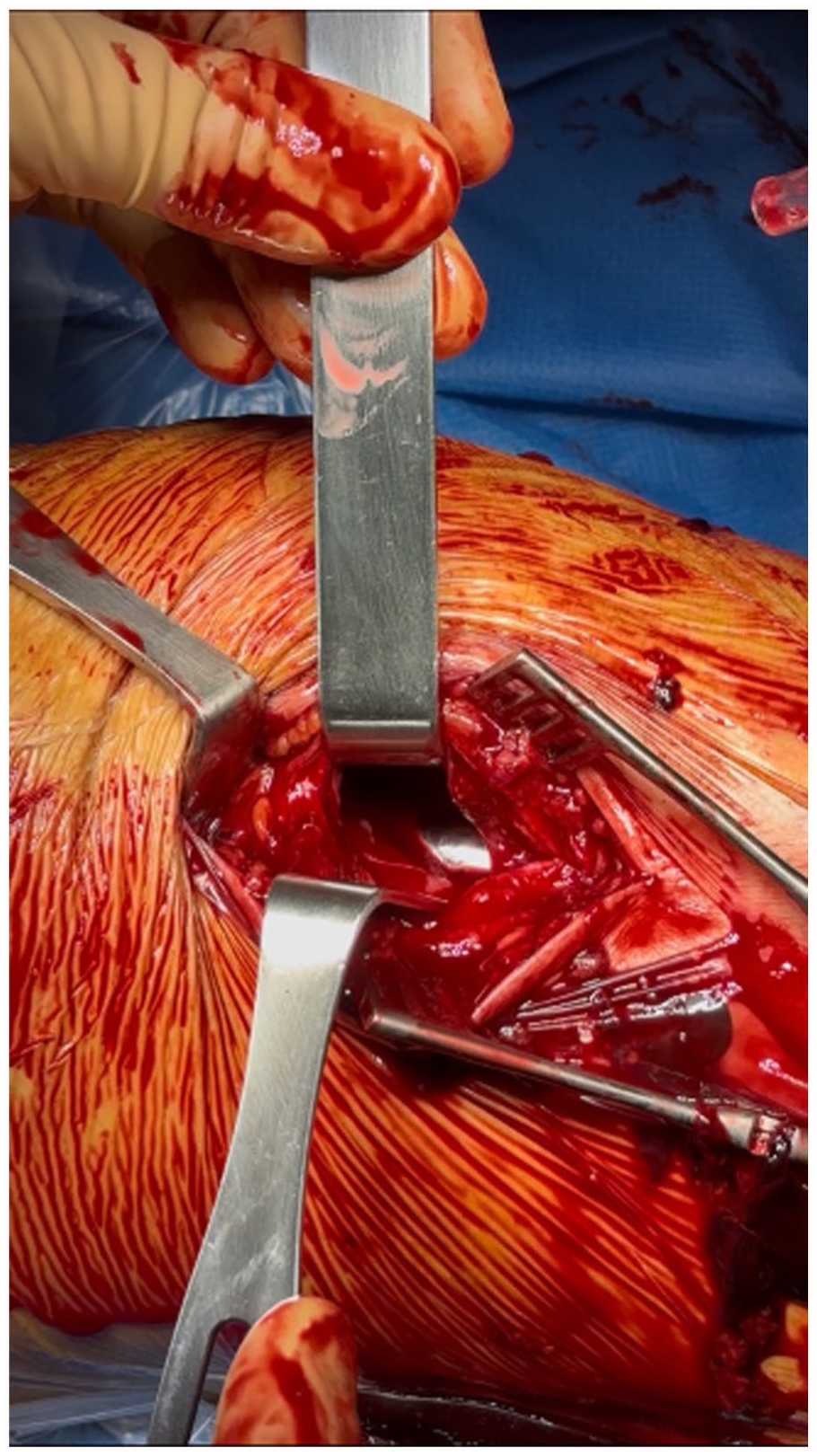
Lateral capsulotomy exposing the TKA prosthetic components adjacent to the lateral femoral condyle and the prosthetic components.
Upon exposure, a well-defined 1.5 cm pseudoaneurysmal sac was identified adjacent to the lateral femoral condyle, with surrounding hematoma and evidence of recent intra-articular bleeding. The hematoma, approximately 300 mL in volume, was carefully evacuated (Figure 6), and was sent for microbiological culture, which returned negative for infection. The pseudoaneurysm was isolated, and meticulous dissection was carried out to ligate the feeding artery both proximally and distally with 5-0 polypropylene sutures (Figure 7). The aneurysmal sac was then excised in its entirety, with no residual vascular tissue. The joint capsule was irrigated thoroughly to remove any blood clots or debris. Hemostasis was achieved without difficulty, and the capsule was closed in layers using absorbable sutures. The skin was approximated with interrupted absorbable stitches. The postoperative course was uneventful and the patient was discharged on postoperative day 2 with double antiplatelet therapy consisting of aspirin 100 mg once daily and clopidogrel 75 mg once daily.

Evacuation of approximately 300 mL of hematoma from the pseudoaneurysmal cavity.
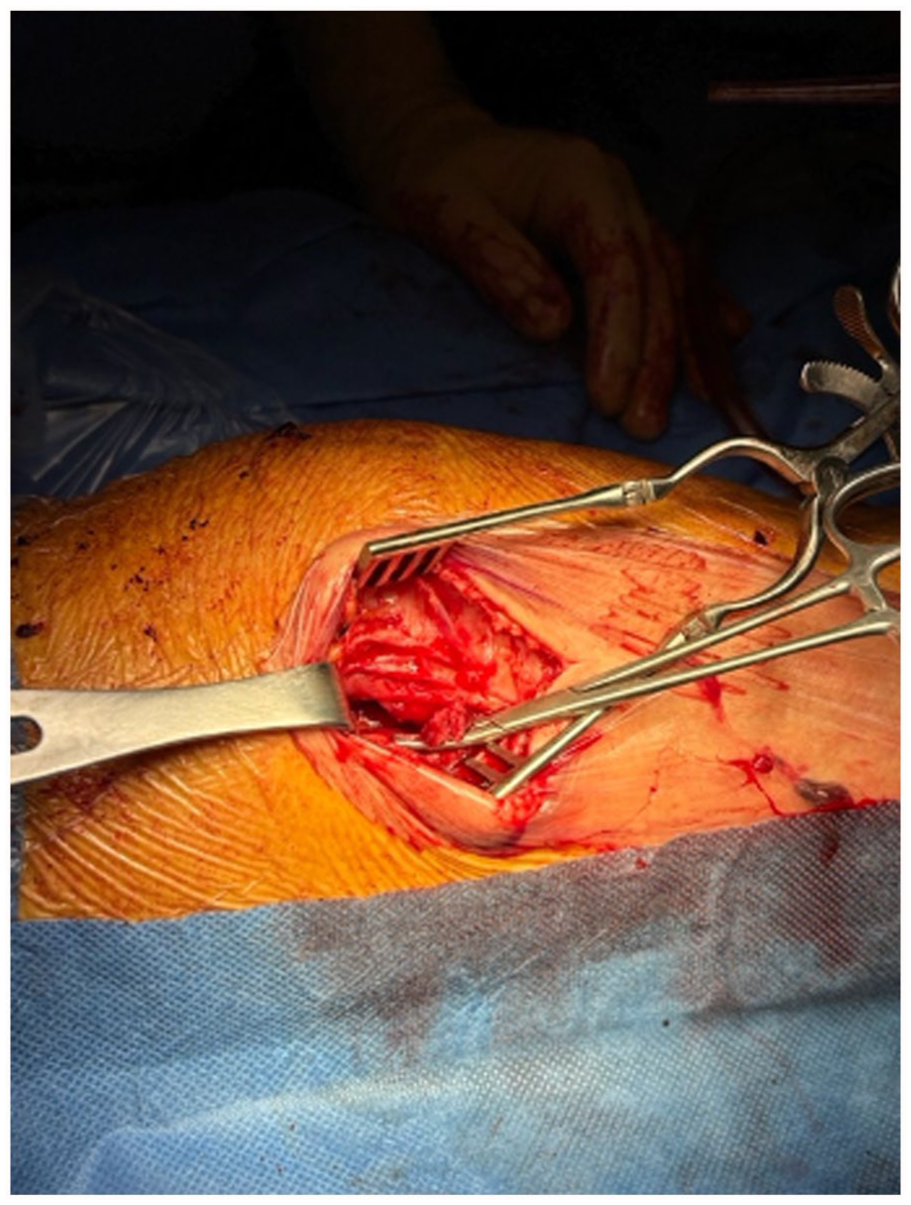
Ligation of the SLGA proximally and distally, followed by excision of the pseudoaneurysmal sac.
Following discharge, the patient was monitored in the outpatient clinic at 1 and 6 months, with the 6-month visit corresponding to the 1-year postoperative follow-up after TKA. At both visits, he demonstrated marked clinical improvement: the knee was non-tender, dry, and exhibited a full ROM (130°-0°-0°). There was no recurrence of joint effusion or tenderness along the lateral femoral condyle or the course of the SLGA, and the surgical incision remained well-healed. Functional evaluation, showed good outcomes, with a KSS clinical score of 95, functional score of 93, and an FJS-12 raw score of 90, indicating near-complete restoration of pain-free ROM, joint stability, and high patient-reported satisfaction.
Discussion
Vascular injuries following knee procedures, including TKA, typically involve the popliteal, medial and lateral inferior genicular, and descending genicular arteries, with only few reports describing pseudoaneurysms of the SLGA.18-21
SLGA is at significant risk of iatrogenic injury during TKA due to its anatomical course in close proximity to key surgical dissection planes. 22 Specifically, the anteromedial and anterolateral approaches for arthrotomy, medial and lateral parapatellar retinacular releases, subperiosteal dissection along the femoral metaphysis, and posterior capsular release. 23 During exposure and soft tissue balancing, particularly in varus and valgus deformity correction, as in our patient with 11° preoperative valgus, the artery may be injured by aggressive lateral dissection near the femoral epicondyles and the posterior aspect of the distal femur. Moreover, the deep positioning of retractors along the posterior capsule or adjacent to the supracondylar region can further predispose the vessel to traction or compression injury. 24
In case of acute pseudoaneurysms, most patients develop symptoms within 10 to 15 days postoperatively, although some cases may present as early as a few hours or days post-surgery.18,25 The most commonly reported symptoms of pseudoaneurysm can range from pain to hemarthrosis, but in some cases, patients may be mildly to asymptomatic. 26
However, delayed-onset pseudoaneurysms, presenting months after surgery, remain a rare entity and pose significant diagnostic challenges. They may exhibit more insidious symptoms, including non-specific pain, persistent and localized swelling associated with reduced ROM.27-29 These symptoms can mimic most common post-TKA complications such as infection, hematoma, or deep vein thrombosis, often leading to misdiagnosis and unnecessary invasive procedures including arthrocentesis.
The exact pathophysiology of delayed pseudoaneurysm formation remains unclear, but it is believed to involve subclinical arterial wall injury sustained during surgery, which gradually weakened the vessel and led to pseudoaneurysm development. Contributing factors may include mechanical stress from joint movement, chronic inflammation, and late-stage arterial wall degeneration. 30 In this case, the patient’s physically demanding job which involves continuous climbing up and down the truck stairs, may have contributed to increase mechanical stress on the knee joint and the SGLA wall, both in the preoperative and postoperative phase. 31 Additionally, the patient’s smoking history may have further compromised vascular integrity, increasing the risk. 32
The diagnostic process for suspected pseudoaneurysms should involve a stepwise approach that typically begins with DUS, a non-invasive modality capable of detecting abnormal vascular flow patterns indicative of pseudoaneurysms. DUS, as a first-line and widely available imaging modality, can rapidly detect abnormal vascular flow patterns, including the characteristic “yin-yang” sign of turbulent blood flow within the pseudoaneurysm sac and the “to-and-fro” waveform at the pseudoaneurysm neck.33,34 It allows for real-time hemodynamic assessment, helping to evaluate the patency of the affected vessel, detect the presence of an arterial neck, and assess potential thrombus formation within the pseudoaneurysm. Additionally, DUS enhances diagnostic accuracy up to 90% without exposing the patient to ionizing radiation or contrast agents. 34 However, overall image quality may be limited by patients with deep-seated lesions, obesity, or extensive soft tissue edema. 35
In cases requiring further anatomical delineation, CTA provides high-resolution cross-sectional imaging, offering detailed visualization of the size of the pseudoaneurysm, its morphology and relationship to adjacent nervous and vascular structures in sight of a surgical intervention. 36 Pseudoaneurysms typically appear as hyperdense saccular structures with an arterial-phase enhancement of both the sac and its feeding artery. Delayed-phase imaging can reveal contrast extravasation, suggesting active bleeding and an increased risk of rupture. 14 CTA remains critical also for surgical and endovascular planning, as it allows for precise localization of the lesion, identification of collateral circulation, and assessment of potential vascular complications including distal arterial insufficiency or distal embolization. 37 The complementary use of both imaging modalities may help prevent diagnostic delays, optimize surgical or endovascular strategies, and support individualized therapeutic decision-making, particularly in cases involving anatomically complex or deep-seated lesions 38
Accurate imaging therefore directly guides individualized therapeutic selection.
Ultrasound-guided compression therapy represents the least invasive option and has been proposed for the management of peripheral lower-limb arterial pseudoaneurysms, particularly in small lesions under 2 cm in diameter. 39 However, no specific reports are available regarding its use in genicular pseudoaneurysms. Moreover, recurrence rates have not been reported in this setting. Factors that may limit its success are likely those described in other locations, including incomplete thrombosis of the sac, patient intolerance due to pain during prolonged compression, and the continuous motion at the knee joint.40,41
When less invasive approaches are inadequate or unsuitable, endovascular therapy has emerged as a valuable minimally invasive option for the management of peripheral lower limb pseudoaneurysms, offering reduced surgical trauma, shorter hospitalization, and a lower risk of wound-related complications particularly in patients at higher surgical risk or in whom open repair is undesirable. 42 Several techniques have been described, including thrombin injection, coil embolization, liquid embolic agents (eg, glue, Onyx), or microparticles.43,44
Among these, guided thrombin injection, while well established for femoral access–site pseudoaneurysms, 45 has only limited application in the management of genicular artery lesions, with evidence restricted to isolated case reports.19,46
Coil embolization may offer effective exclusion of the pseudoaneurysm by occluding the feeding vessel, although recurrence may occur through collateral reconstitution. 47 while liquid embolic and microparticles provide the possibility of more distal penetration, but carry a higher risk of non-target embolization and ischemic complications.48-51
Nevertheless, surgical ligation, as performed in this case, remains a definitive treatment, particularly when the pseudoaneurysm is accessible and there is concern for rupture or neurovascular compromise. The open surgical approach offers several advantages, including direct visualization and control of the affected vessel, the ability to remove the pseudoaneurysm sac and the potential for arterial reconstruction if needed. 17 This approach is especially beneficial in cases of infected pseudoaneurysms, where debridement is necessary, or when endovascular access is challenging due to vessel tortuosity. 18 However, open surgery is associated with longer recovery times, increased surgical trauma and a higher risk of wound complications compared to minimally invasive techniques. 19
In our case, these considerations proved decisive. Given the ongoing intra-articular effusion, the need for joint evacuation and culture, and the anatomical location immediately adjacent to the implant in close proximity to the lateral femoral condyle, open surgical ligation through a lateral capsulotomy was undertaken. This approach provided direct access, definitive control of the lesion, and simultaneous management of the hemarthrosis. Moreover, given the complexity of diagnosis and management, a multidisciplinary approach involving both vascular and orthopedic surgeons showed to be crucial to achieve optimal outcomes in patients presenting with delayed complications following TKA.
Timely communication between orthopedic and vascular teams facilitates early recognition, accurate diagnosis, and intervention, reducing the risk of complications such as rupture, thrombosis, or distal ischemia. The orthopedic surgeon’s familiarity of the surgical anatomy and postoperative course facilitates the recognition of atypical presentations, while the vascular surgeon offers specialized expertise in imaging interpretation and in the selection of the most appropriate treatment strategy. Additionally, a collaborative approach can help minimize disruption to the rehabilitation process and optimize long-term functional recovery.
Limitations
The main limitation of this report is its single-patient design, which prevents broad generalization regarding the incidence or risk factors for pseudoaneurysm following total knee arthroplasty. Furthermore, the follow-up period, while sufficient to document short-term recovery, does not allow for evaluation of long-term vascular and functional outcomes. Additionally, certain variables including subtle anatomical variations, intraoperative technical factors, or perioperative vascular injury could not be systematically analyzed.
Take Home Messages
Pseudoaneurysm of the SLGA may present months after otherwise uncomplicated TKA. Persistent pain, swelling, or hemarthrosis should prompt consideration of vascular injury, even in patients with unremarkable early recovery.
DUS and CT angiography are essential for accurate diagnosis, localization, and characterization of pseudoaneurysms. Early imaging may reduce misdiagnosis and prevent complications such as pseudoaneurysm rupture or distal ischemia.
Individual patient factors, including preoperative valgus deformity, high mechanical stress from manual labor, and a history of smoking, may contribute to subclinical arterial injury and delayed pseudoaneurysm formation, supporting the need for tailored postoperative surveillance.
Open surgical ligation and excision can be effective, particularly when ongoing hemarthrosis, proximity to prosthetic components, or the need for joint debridement exists.
Coordinated management between orthopedic and vascular teams may ensure safe surgical exposure, effective hematoma evacuation, and preservation of knee function.
Conclusions
Maintaining a high index of suspicion for vascular complications is essential, even in delayed presentations following TKA. Pseudoaneurysm of the SLGA should be actively considered in the differential diagnosis of persistent knee pain and swelling, particularly when standard evaluations yield no clear cause. Prompt imaging and early intervention are critical to prevent severe complications such as rupture, thrombosis, or distal ischemia.
Given the potential severity of this condition, further research is imperative to clarify its incidence, risk factors, and optimal management. Raised awareness and close multidisciplinary collaboration among orthopedic surgeons, radiologists, and vascular specialists is crucial for the early recognition, accurate diagnosis, and timely treatment of this rare yet serious complication and may play a pivotal role in ensuring effective management.
Footnotes
Abbreviations
TKA – Total Knee Arthroplasty
SLGA – Superior Lateral Genicular Artery
ROM – Range of Motion
KSS – Knee Society Score
FJS – Forgotten Joint Score
ED – Emergency Department
DUS – Duplex Doppler Ultrasonography
CTA – Computed Tomography Angiography
NSAIDs – Non-Steroidal Anti-Inflammatory Drugs
Hb – Hemoglobin
WBC – White Blood Cell count
RBC – Red Blood Cell count
CRP – C-Reactive Protein
ESR – Erythrocyte Sedimentation Rate
PT – Prothrombin Time
aPTT – Activated Partial Thromboplastin Time
FEU – Fibrinogen Equivalent Units
Ethical Considerations
Ethical approval was not required for this case report, as it describes a single clinical case and does not involve identifiable patient data beyond standard clinical documentation.
Consent to Participate
Written informed consent was obtained from the patient for participation in this case report and for the use of relevant clinical data for publication purposes
Consent for Publication
Written informed consent was obtained from the patient for the publication of this case report and any accompanying images.
Authors Contributions
Conceptualization L.G., M.L.R, M.C., G.S. Methodology L.G., M.C. Validation L.G., M.C. Writing—original draft preparation, L.G., M.C., G.S. Writing—review and editing. L.G., M.C., G.S., G.M. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available on reasonable request from the authors.
