Abstract
Acute lymphoblastic leukemia (ALL) rarely presents with initial liver failure. We describe a 9-year-old boy with no past medical history who presented with jaundice, hyperbilirubinemia (total bilirubin 13.1 mg/dL), and significantly elevated transaminases (ALT 1283 U/L, AST 1325 U/L). Bone marrow aspiration confirmed precursor B-cell ALL. Despite severe hepatic dysfunction, full-dose induction chemotherapy was administered following corticosteroid therapy, leading to rapid improvement in liver function and normalization by day 29. No complications were observed, and at the end of induction, minimal residual disease was 0.001%. This case of successful unadjusted chemotherapy, with leukemic infiltration identified as the etiology of hepatitis, underscores the importance of early diagnosis, multidisciplinary management, close monitoring, and highlights the need for further investigation into safe treatment protocols in such cases.
Keywords
Introduction
Acute Lymphoblastic Leukemia (ALL) is the most prevalent type of cancer in children. 1 Over 80% of ALL cases represent B-cell Acute Lymphoblastic Leukemia (B-ALL), where genetic mutations disrupt differentiation at the precursor B-cell stage, sustaining survival and proliferation of malignant cells. 2 The infiltration and proliferation of malignant lymphoid cells may extend beyond the bone marrow (BM) to affect extramedullary organs, like the central nervous system, spleen, lymph nodes, kidneys, testes, and liver, showing the systemic nature of ALL.2-4
Liver involvement is a common manifestation in ALL patients, with presentations ranging from asymptomatic hepatosplenomegaly in about 68% of cases to severe fulminant hepatitis.4-6 However, Acute Liver Failure (ALF) as initial ALL presentation is rarely reported. 3 While drug-induced hepatotoxicity is a known complication of chemotherapy in ALL patients, initial liver failure can complicate treatment and may require dose modification to mitigate toxicity.3,7
In this case report, we describe a rare presentation of B-ALL manifesting as ALF with jaundice and significantly elevated transaminases, underscoring the therapeutic challenges in managing ALL with concurrent hepatic impairment.
Case Presentation
A 9-year-old Caucasian boy presented to the pediatric clinic with a 3-day history of anorexia, nausea, non-bilious and non-bloody vomiting, jaundice with scleral and skin icterus, and 2 months of fatigue. He reported no abdominal pain, fever, diarrhea, recent bleeding, bruising, travel, or trauma. The patient, an only child born from a normal pregnancy, had a BMI of 15.9 (44th percentile), normal developmental milestones, no nutritional or vitamin deficiency, and was fully vaccinated, including against tuberculosis and hepatitis B. He had no history of chronic diseases such as chronic liver failure, hospitalization, tuberculosis exposure, or use of potentially hepatotoxic agents in the last month. There was no family history of liver disease, hereditary disorders, or malignancies.
On examination, the abdomen was soft and non-tender, with a liver span of approximately 7 cm, measured by percussion. The spleen was not palpable, and no masses, ascites, or signs of peritonitis were noted. There was no lymphadenopathy, spider angiomas, palmar erythema, or pedal edema. Testicular and neurological exams were normal, and vital signs were age-appropriate.
The patient was hospitalized for further evaluation. Initial differential diagnoses included viral hepatitis, obstructive biliary diseases, hemophagocytic lymphohistiocytosis (HLH), autoimmune hepatitis, hemolytic anemia, and malignancies.
Abdominal ultrasound revealed a normal common bile duct, no organomegaly of the liver or spleen, no focal hepatic lesions, and para-aortic lymph nodes measuring up to 15 mm in diameter. Initial laboratory tests revealed significant liver dysfunction with: total bilirubin 13.1 mg/dL, direct bilirubin 6.43 mg/dL, alanine aminotransferase (ALT) 1283 U/L, aspartate aminotransferase (AST) 1325 U/L, alkaline phosphatase (ALP) 1150 U/L, and γ-glutamyl transferase 44 U/L. Coagulation results were also prothrombin time at 14.5 seconds (normal: 11-13.5 seconds) and partial thromboplastin time at 28.1 seconds (normal: 25-35 seconds). Lactate dehydrogenase (LDH) and uric acid levels were also elevated, while triglycerides, fibrinogen, and albumin levels were normal. Autoimmune autoantibodies, viral serologies, and interferon-gamma release assay (IGRA) were all negative, excluding autoimmune hepatitis, viral hepatitis, and tuberculosis. Hematologic evaluation revealed pancytopenia with WBC 2100/μL (differential: 13% neutrophils, 56% lymphocytes, 1% monocytes, 3% immature cells), platelets 18,000/μL, RBC 3.8 million/μL, hemoglobin 10.1 g/dL, Ferritin 121 ng/mL, and reticulocyte count 0.5%, findings that also rule out hemolytic anemia. Additional investigations, including blood cultures, urinalysis, and stool examination, were unremarkable. All laboratory results are shown in Table 1.
Overview of Patient’s Initial Laboratory Tests.

Abbreviations: Ab, antibody; CMV, cytomegalovirus; EBV, Epstein-Barr virus; HAV, hepatitis A virus; HBs Ag, hepatitis B surface antigen; HBV, hepatitis B virus; HIV, human immunodeficiency virus; IgM, immunoglobulin M; IGRA, interferon-gamma release assay; PT, prothrombin time; PTT, partial thromboplastin time.
Standard ranges represent age-specific pediatric values per hospital standards.
Bone marrow aspiration (BMA), performed due to pancytopenia, showed 80% blasts, positive for CD19, CD10, CD45, CD38, TdT, HLA-DR, CD123, and CD66c on flow cytometry, confirming precursor B-ALL. No common ALL translocations were found, and cytogenetics were normal (Figure 1). HLH was considered initially but ruled out due to absent hemophagocytosis on BMA, normal ferritin, fibrinogen triglyceride levels (Table 1), and lack of fever or splenomegaly. After excluding other etiologies, leukemic infiltration was identified as the cause of hepatic dysfunction. However, a liver biopsy to confirm leukemic infiltration was avoided due to bleeding and infection risks associated with pancytopenia. Magnetic resonance cholangiopancreatography (MRCP) also ruled out biliary obstruction and supported the findings, suggesting hepatitis secondary to leukemic infiltration.

The bone marrow smear of the patient reveals cells that are 3 to 4 times larger than mature red blood cells, characterized by a high nuclear-to-cytoplasmic ratio. The cytoplasm is scant, light blue, and lacks granules, indicative of lymphoblasts (Leishman stain, ×100).
Treatment was initiated with daily divided doses of dexamethasone (6 mg/m²), with regular monitoring of vital signs and laboratory tests. Within 24 hours, Liver Function Tests (LFTs) showed significant improvement. Following 3 days of corticosteroid therapy, LFTs decreased to: ALT 529 U/L, AST 362 U/L, total bilirubin 4.61 mg/dL, and direct bilirubin 2.58 mg/dL. With rapid improvement in LFTs following steroid administration and exclusion of other hepatitis etiologies, leukemic infiltration was confirmed as the cause of ALF. Following consultation with a pediatric gastroenterologist, chemotherapy was initiated after 3 days of dexamethasone administration to reduce leukemic burden and cytokine release, thereby minimizing the risk of tumor lysis syndrome. The Children’s Oncology Group (COG) protocol AALL0932 for standard-risk B-ALL induction therapy was administered. High disease burden and persistent blastemia necessitated urgent and unadjusted treatment including intrathecal methotrexate and cytarabine, intravenous vincristine, and PEG-asparaginase with daily LFTs monitoring. The LFTs timeline and treatment regimen details are presented in Table 2.
Timeline of LFT Trends and Chemotherapy Regimen During the Induction Phase.
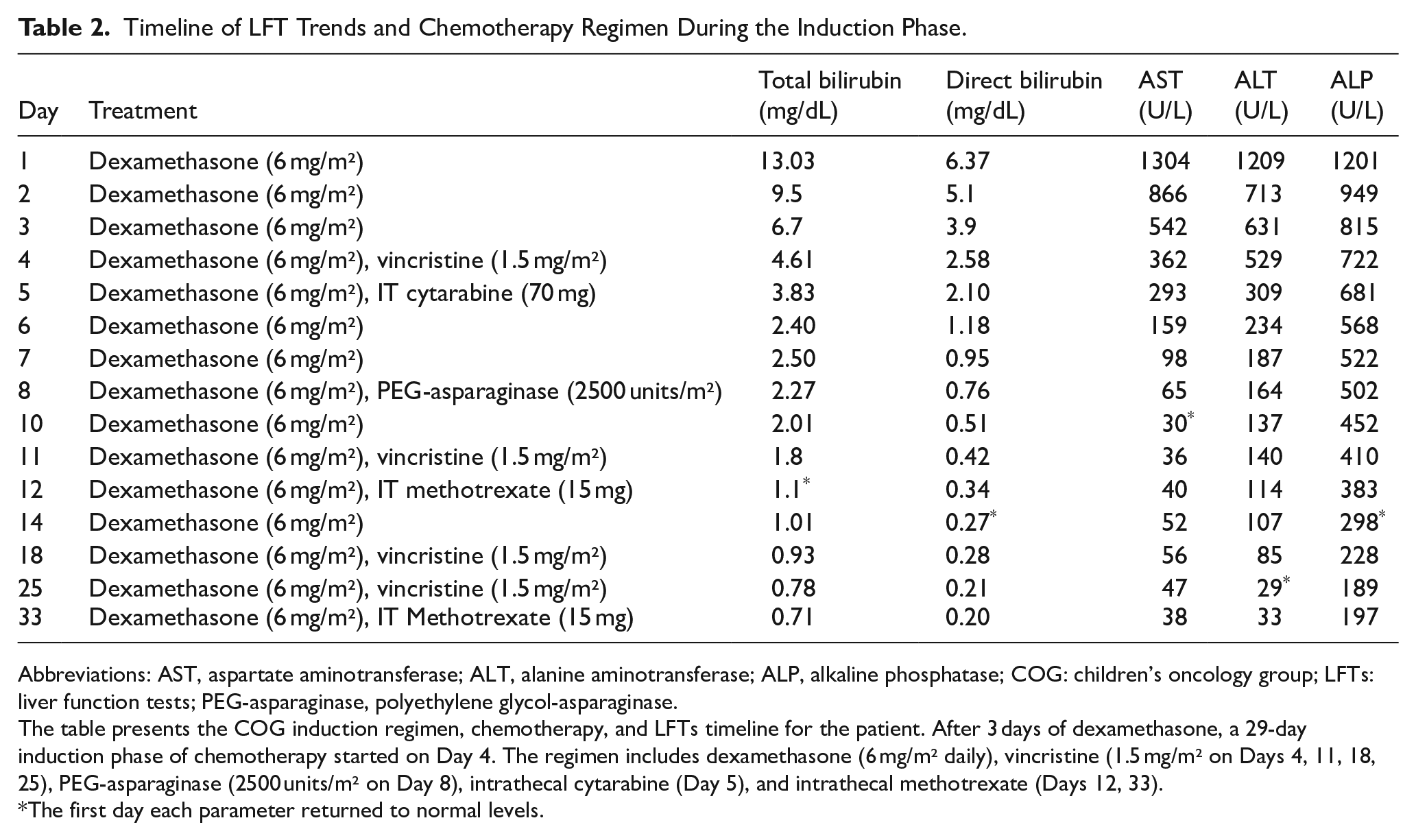
Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; COG: children’s oncology group; LFTs: liver function tests; PEG-asparaginase, polyethylene glycol-asparaginase.
The table presents the COG induction regimen, chemotherapy, and LFTs timeline for the patient. After 3 days of dexamethasone, a 29-day induction phase of chemotherapy started on Day 4. The regimen includes dexamethasone (6 mg/m² daily), vincristine (1.5 mg/m² on Days 4, 11, 18, 25), PEG-asparaginase (2500 units/m² on Day 8), intrathecal cytarabine (Day 5), and intrathecal methotrexate (Days 12, 33).
The first day each parameter returned to normal levels.
Follow-up showed a gradual decline in LFTs. By day 10, AST was 30 U/L, ALP was 298 on day 14, and by day 25, ALT was 29 U/L. Jaundice resolved by day 12, with total bilirubin at 1.1 mg/dL and direct bilirubin at 0.27 mg/dL by day 14, all within normal ranges. On day 29 of chemotherapy, at the end of the induction phase, repeat BMA revealed minimal residual disease (MRD) of 0.001%, correlated with decreasing LFTs, supporting continuation of chemotherapy. Subsequent improvement following chemotherapy may also suggest a multifactorial etiology, with drug-induced liver injury potentially contributing; however, leukemic infiltration remains the primary cause, as evidenced by the rapid improvement in LFTs with steroid therapy.
The patient is currently in the maintenance phase under regular hematology follow-up. After 1 year of treatment, chemotherapy has been successful, with no organomegaly, significant LFTs elevation, or jaundice observed.
Discussion
ALL is a hematologic malignancy representing approximately 25% of all pediatric cancers, characterized by uncontrolled lymphocyte proliferation.1,2 ALL typically manifests with cytopenia (anemia, neutropenia, and thrombocytopenia) due to malignant cell replacement in BM, often includes systemic symptoms such as fever, fatigue, weight loss, and night sweats. 8 The proliferation of immature lymphoid cells can infiltrate extramedullary sites, potentially affecting the central nervous system, testicles, liver, spleen, thymus, and lymph nodes, which can cause symptoms related to the affected organs.4,5,9
Hepatomegaly is the most common symptom in children with ALL, with over 50% presenting with a palpable liver. However, initial ALF is rarely reported.3,6,10,11 The mechanism of liver dysfunction is not clear but can be attributed to paraneoplastic syndrome, concomitant viral infections, sepsis, chemotherapeutic drug-induced liver injury, and lymphocyte infiltration, which is more susceptible in cases with normalized LFTs with early steroid intake.3,11 Leukemic liver infiltration is more common in T-cell ALL, older patients, those with a high number of WBCs, and bulky disease. It is rare for B-ALL to present with ALF at the initial diagnosis.5,12
Although Initial liver damage in acute leukemia is not typically evident in laboratory tests, postmortem studies have observed liver infiltration in more than 95% of patients with ALL. 13 Despite the necessity of liver biopsy for early diagnosis, it is not recommended due to the increased risk of bleeding and infections.5,11 In pediatric ALL, mild liver dysfunction is common, with about 30% of patients showing abnormal aminotransferase levels. Direct hyperbilirubinemia is seen in 3.4% of cases, but severe elevations are rarely reported.5,14 In addition, elevated levels of LDH and uric acid are often observed in ALL-associated hepatitis. 5
Contemporary ALL Treatment phases consist of 4 phases: induction, consolidation, intensification, and maintenance guided by risk stratification according to clinical factors. The induction phase, which generally lasts 4 to 6 weeks, has a success rate of nearly 98%.1,15 Chemotherapeutic agents used during the induction phase could exhibit hepatotoxicity. Specifically, asparaginase and vincristine can induce liver injury due to their hepatic detoxification systems. 16 In contrast, liver injury from intrathecal methotrexate and cytarabine is uncommon.17,18
In cases with elevated conjugated bilirubin or transaminase levels, dose reduction or delayed chemotherapy, combined with initial steroid administration, is recommended.5,19 However, some studies have reported that, in cases similar to ours, initiating full-dose chemotherapy after corticosteroid therapy can lead to a rapid and significant decline in transaminase and bilirubin levels, without major complications and with leukemia cure rates comparable to those in patients without hepatitis.3,5,11 Glucocorticoids are a key part of induction, inducing leukemic cell apoptosis and providing prognostic insights based on treatment response. 20 as observed in our case during chemotherapy. Preventive measures, including ursodiol administration and LFTs monitoring, should also be considered, with plans to consider withholding chemotherapy if an elevation in LFTs occurs.13,21
Pediatric ALL presenting with ALF has not been widely reported. Four cases with similar presentations are summarized in Table 3. All patients presented with jaundice, markedly elevated transaminases, and hyperbilirubinemia. A liver biopsy was only performed in the report by Ford et al when the etiology remained unidentified during the initial evaluation. Despite variations in clinical presentation, each case received corticosteroids prior to chemotherapy. However, no consensus exists regarding dose reduction or the optimal timing for initiating chemotherapy following steroid therapy. Notably, all cases achieved successful treatment outcomes accompanied by improvement in LFTs.3,11,21,22
Previous Cases of Childhood ALL Presenting as ALF.

Abbreviations: ALL, acute lymphoblastic leukemia; ALF, acute liver failure; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ICiCLe, Indian Collaborative Childhood Leukemia Group; IVIG, intravenous immunoglobulin; LFTs, liver function tests; TPOG, Taiwan Pediatric Oncology Group.
Although chemotherapy-induced toxicity may cause morbidity and mortality, changes in total bilirubin, AST, and ALT levels during diagnosis and post-induction in patients with acute leukemia can lead to treatment delays, potentially increasing the risk of hepatotoxicity-related death. 16 However, early treatment has been associated with improved long-term survival rates.11,23 Given the high leukemic burden and persistent blastemia, the medical team opted for early initiation of therapy to prevent further hepatic injury.
Conclusion
A rare pediatric B-ALL case presenting initial symptomatic hepatitis with transaminases over 1000 U/L and significant hyperbilirubinemia highlights the diagnostic challenge of initial hepatitis in ALL, where leukemic infiltration, drug toxicity, and infections must be considered. The significant LFTs improvement following steroids and MRD results supported leukemic infiltration as the etiology, justifying chemotherapy continuation. This allowed full-dose induction therapy without hepatic complications despite initial risks and underscores the need for multidisciplinary monitoring. Future studies should optimize specific regimens and explore biomarkers to predict hepatotoxicity risk in similar patients.
Footnotes
Acknowledgements
We thank the Department of Clinical Research Development Unit of Amirkabir Hospital in Arak for their support in the preparation of this report.
Ethical Considerations
This case report was approved by the Research Ethics Committee of Arak University of Medical Sciences (Approval ID: IR.ARAKMU.REC.1403.318, Date: 2024-12-29).
Consent to Participate
Written informed consent was obtained from the parents, with consideration of the child’s rights and autonomy. The parents had a prospective understanding of the treatment plan based on their child’s condition prior to study initiation, and the patient provided age-appropriate verbal assent.
Consent for Publication
Written informed consent was obtained from the patient and his parents. No identifiable information or images were included.
Author Contributions
AL: Conceptualization, Supervision, Validation. MS: Investigation; Writing—original draft; Writing—review and editing, Project Administration. MSZ: Data Curation, Methodology. NP: Validation, Investigation, Writing—Review and Editing. AHR: Resources, Writing—Review and Editing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available to other authors upon reasonable request.
Presentation(s) or Awards at a Meeting
None.
Use of Artificial Intelligence (AI)-Assisted Technologies
During the preparation of this work, the authors used ChatGPT-4.0 for language editing and improving the readability based on Sage policy on AI usage.
