Abstract
Biliary tract carcinomas (BTC) are malignant epithelial neoplasms subdivided anatomically into: gallbladder carcinomas and carcinomas of the bile duct or cholangiocarcinomas (CCA); including intrahepatic, hilar/perihilar, and distal CCA. Adenocarcinoma accounts for the most common BTC (over 90% of all carcinomas), while other histological subtypes represent rarer forms including: poorly cohesive/signet ring cell carcinoma, which has a greater malignant potential than conventional BTC and a poorer prognosis. Only few cases have been reported in the literature to date. The positive diagnosis remains on histology. Herein, we describe a new case of poorly cohesive carcinoma of bile duct extending to the gallbladder in a 60 years old women with a fatal outcome, to raise awareness of this rare entity and to provide data for larger series.
Introduction
Biliary tract carcinomas (BTC) are malignant epithelial neoplasms subdivided anatomically into: gallbladder carcinomas and carcinomas of the bile duct or cholangiocarcinomas (CCA); including intrahepatic, hilar/perihilar, and distal CCA. 1 BTCs are rare cancers with an estimated incidence of less than 0.6 cases per 100 000 inhabitants/year. 1 Adenocarcinoma accounts for the most common BTC (90% of all carcinomas), while other histological subtypes represent rarer forms including: adenosquamous carcinoma, clear cell carcinoma, lymphoepithelioma-like carcinoma, sarcomatous CCA, and poorly cohesive/signet ring cell carcinoma. 2 The latter is extremely rare and accounts for only 1.3% of BTC, showing a greater malignant potential than conventional BTC. Only few cases have been reported in the literature to date. 3 Herein, we describe a new case of poorly cohesive carcinoma of bile duct extending to the gallbladder in a 60 years old women with fatal outcome, to raise awareness of this rare entity and provide data for larger series. The authors shed light on the pathological aspects of this rare histotype as well as on its poor prognosis.
Case Presentation
A 60 years old women was referred to the department of surgery for a 2 months history of obstructive cholestasis and weight loss. She had a history of type 2 diabetes without previous hepatobiliary complaints. Physical examination revealed jaundice and tenderness of the right hypochondrium without lymph nodes. The general condition was moderately deteriorated, with stable vital signs.
Blood analysis showed a slight anemia (hemoglobin at 10 g/dL), cytolysis, and cholestasis. Tumor markers (carcinoembryonic antigen, CA 19-9, alpha-fetoprotein) and other parameters were within normal ranges.
Computed tomography revealed a tissular mass in the hepatic hilum with heterogeneous enhancement measuring: 65 × 59 × 29 mm3 (Figure 1), infiltrating the inferior vena cava and extending to the gallbladder which shows a diffuse parietal irregular thickening measuring 12 mm. These findings were suggestive of hilar CCA extending to the gallbladder diffusely. Routine staging (Thoraco-abdomino-pelvic CT scan and fibroscopy) were negative for metastasis, or primary gastric carcinoma. Since the tumor was locally advanced, palliative drainage was indicated. Initially, endoscopic retrograde cholangiopancreatography (ERCP) drainage was unsuccessful due to altered biliary anatomy. The patient then underwent surgical biliary drainage. After a right subcostal laparotomy, surgical exploration revealed a tumor in the gallbladder with infiltration of the common hepatic duct and cystic duct. Dissection of the hilar plate demonstrated the extension of the tumor process to the hepatic convergence and the right duct. A hepaticojejunostomy was performed on the left hepatic duct using a Roux-en-Y technique. Bile samples and transductal biopsies of the tumor were obtained. Histology revealed a carcinomatous proliferation consisting of isolated cells focally arranged in a trabecular pattern without well-formed glands. No signet cell component was present. Neoplastic cells were embedded in a an abundant edematous or mucinous stroma (Figure 2). No biliary epithelium was found in all fragments. Immunohistochemistry showed positive CK7 and muc1, and confirmed as well the poorly cohesive feature. CK20 was negative. The definitive diagnosis was primary poorly cohesive carcinoma of bile duct with gallbladder involvement. Unfortunately, the patient succumbed to a massive pulmonary embolism the fifth day after surgery. Thus the patient couldn’t receive any anti-cancer treatment (Figure 3).
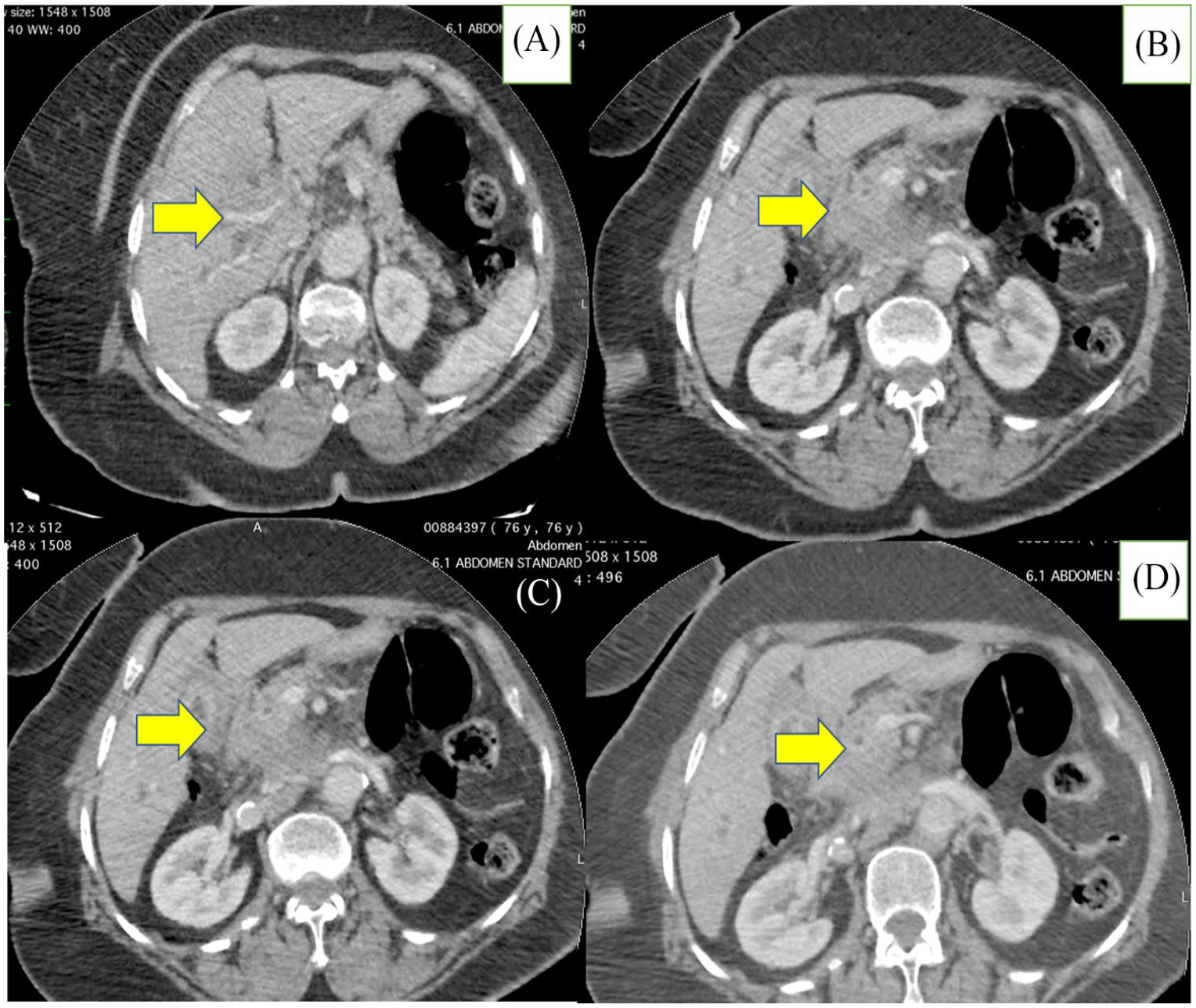
Computed tomography revealed a tissular mass in the hepatic hilum with heterogeneous enhancement measuring :65 × 59 × 29 mm3 (Figure 1), infiltrating the inferior vena cava and extending to the gallbladder which shows a diffuse parietal irregular thickening measuring 12 mm. (A) Hepatic hilum; (B-D) Gallbladder.

Poorly cohesive cell carcinoma histological features: fibro adipous tissue infiltrated by PCC, no biliary epithelium was identified. (A) hematoxylin and eosin, ×100; (B) hematoxylin and eosin, ×400; (C, D) hematoxylin and eosin, ×400 (angio invasion yellow arrow).

Immunohistochemsirty findings: (A, B) positive for CK7, (C) positive for muc1, and (D) negative for CK20.
Discussion
CCA is a heterogeneous group of BTC, developing from cholangiocytes and hepatic stem or progenitor cells (for intra hepatic CCA). 4 The spectrum of these tumors include several histotypes: adenocarcinoma, which is by far the most common; adenosquamous carcinoma, sarcomatous CCA, mucinous carcinoma, mucoepidermoid carcinoma, lymphoepithelioma-like carcinoma, clear cell carcinoma, and poorly cohesive/signet ring cell carcinoma. These histological subtypes show different epidemiological aspects, survival, and prognosis although having the same anatomic origin. In fact, poorly cohesive carcinoma/signet cell carcinoma is a rare histological type, reported mainly in gallbladder and representing only 1.3% of all cases of BTC (reported in 525 cases including gallbladder and BTC).2,3 These tumors are more frequent in women, with mean age of 66 years old, 5 as in our reported case (60 years old women). There are 2 pathogenetic hypothesis regarding the histogenesis of PCC/SCC, it can be secondary to a gastric metaplasia, or to a preexisting ectopic gastric mucosa. 3 Clinically, CCAs are commonly asymptomatic at early stages. When symptomatic, the most common presentation is jaundice, in extra hepatic forms, because of the obstruction of bile duct. Advanced stages, include other symptoms like: asthenia, abdominal pain asthenia, and weight loss, as for our patient. 6 Radiology (CT scan and MRI) plays a pivotal role, in diagnosing, staging, follow-up, and assessment of treatment response. Given the lack of specific imaging features, pathological analysis (by cytology or histology) is required for an accurate diagnosis.6,7 However, these tumors might be difficult to access for sampling, resulting in inadequate or insufficient samples. 4 Diagnostic tools include, cholangioscopy-guided biopsies, and brush cytology. As most of the cases present with advanced, unresectable disease, surgical biopsies can be performed during surgical palliation. 7
The diagnosis requires a histological proof, PCC/SCC are defined as their GI counterparts, by the presence of “ individual cells and cord like patterns forming the diffuse infiltrative growth in which the cells dissect through the tissue planes.” 2 The presence of signet ring cells, characterized by abundant intracytoplasmic mucin pushing the nucleus to the periphery; is not required for the diagnosis. The diagnosis of SCC requires more than 50% of signet ring cells. 3 Although being rare, the differential diagnosis include metastatic signet ring carcinoma of other sites, especially stomach and colon. Immunohistochemical profile, in addition to a complete routine staging, confirms the biliary primary origin of the tumor. 8 In our patient, immunohistochemistry showed positivity for CK7 and MUC1 with negative CK20 and MUC2. Commonly altered genes in CCA are TP53, SMAD4, KRAS, ARDI1A, and GNAS. In fact genomic testing plays important role in understanding responses to chemotherapy as well as finding new potential therapeutic targets. PCC have only been reported in few cases, with a 5-year survival rate worse than typical CCA. Thus it represents a more aggressive tumor showing less favorable prognosis than conventional adenocarcinoma.2,4
This type of carcinoma is aggressive tumor with a poor prognosis, the main prognostic factors are the stage at the presentation. Histoprognostic factors include: poor differentiation, agressive subtypes, vascular, and perineural invasion. 2 Recently, prognostic molecular markers have been identified to predict the aggressiveness of the disease and select the appropriate treatment regimen. 9 Therapeutical options include radical or palliative surgery, chemotherapy, radiotherapy, targeted therapy, and immunotherapy.10,11
Conclusion
In conclusion, the authors report a new case of PCC of the bile duct to raise awareness about this entity and to highlight its poor prognosis.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors read and approved the final manuscript.
Informed Consent
Informed consent was obtained from the patient’s guarantor.
Guarantor
Corresponding author.
