Abstract
In this paper, we present 2 patients with ST-elevation myocardial infarction in the course of Spontaneous Coronary Artery Dissection. The first patient, a female admitted with STEMI, was diagnosed with SCAD with intravascular imaging and treated with IVUS-guided PCI. In the second patient, a young male hospitalized due to angina and STEMI, SCAD was not identified initially, and the patient was treated conservatively. Based on these cases, we show different faces of the same disease that imply different diagnostics and management strategies. We draw attention to the fact that the disease affects not only young women without atherosclerotic risk factors and that it is not always possible to avoid generally not recommended invasive treatment and anticoagulant therapy. The paper also discusses the disease’s pathophysiology and its diagnosis methods.
Introduction
Spontaneous Coronary Artery Dissection (SCAD) is acute intramural bleeding that results in an intramural hematoma (IMH) in a coronary artery. This hematoma may separate any of the 3 layers of the coronary arterial wall, disrupting blood flow and leading to myocardial ischemia and infarction (MI).1,2
SCAD accounts for 1% to 4% of all cases of acute coronary syndrome (ACS).1,2 The incidence is higher in women than in men, with a ratio of approximately 9:1. It is estimated that SCAD is responsible for 35% of ACS cases in women under the age of 60.1,2 Recent studies suggest that the prevalence of atherosclerosis risk factors in these patients is not as low as previously believed; arterial hypertension, hyperlipidemia, and smoking may be present in up to 30% to 60% of patients.3,4
It is suggested that SCAD may occur through 2 different mechanisms: either a primary intimal tear, leading to the creation of a false lumen (the “inside-out” mechanism), or bleeding within the vessel wall (the “outside-in” mechanism). 5
In the second mechanism, considered more common, IMH arises de novo due to the rupture of vasa vasorum. 3 A growing hematoma causes the intima to bulge into the lumen, exposing sub-endothelium to flowing blood and shear stress. Thus, IMH may be the precipitating factor leading to intimal tear in most cases. 6 When intimal rupture is present, it is not certain if it is a result or a cause of IMH. Tearing of the intima may initiate platelet activation and promote thrombus formation. Studies show that lumen thrombi may be present in 36% of fenestrated and 14% of non-fenestrated cases. 7
The Yip-Saw classification categorizes angiographic patterns into 3 types (Figures 1–4). Type 1 SCAD is recognized by evident arterial wall contrast staining with a double lumen appearance. It is likely to develop later in the disease course, probably due to decompression of the false lumen hematoma into the true lumen. Type 1 SCAD is associated with a lower risk for clinical progression when managed conservatively and increased risk of complications when managed with percutaneous coronary intervention (PCI). Type 2, the most prevalent (more than 70% of all cases of SCAD), is characterized by a long (usually >20 mm) smooth stenosis. In type 2a, there is restoration of a normal vessel beyond the dissection area; in type 2b, the narrowing continues into the most distal angiographically visible segments. Type 3 mimics focal atherosclerotic disease, usually requiring intracoronary imaging for definitive diagnosis.5,6 The European Society of Cardiology (ESC) distinguishes type 4 of SCAD as complete vessel occlusion, usually in the distal section of the artery, which requires differentiation from peripheral embolization. 8
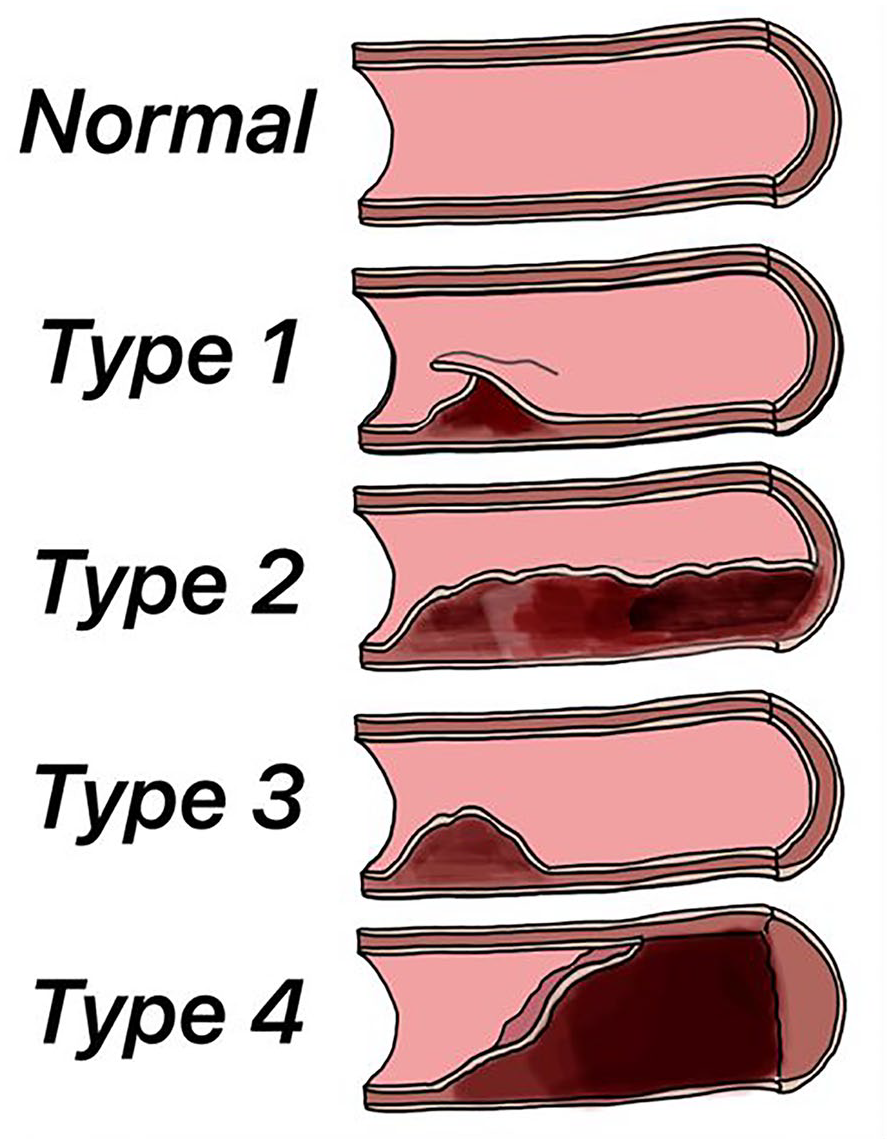
Classification of spontaneous coronary artery dissection.

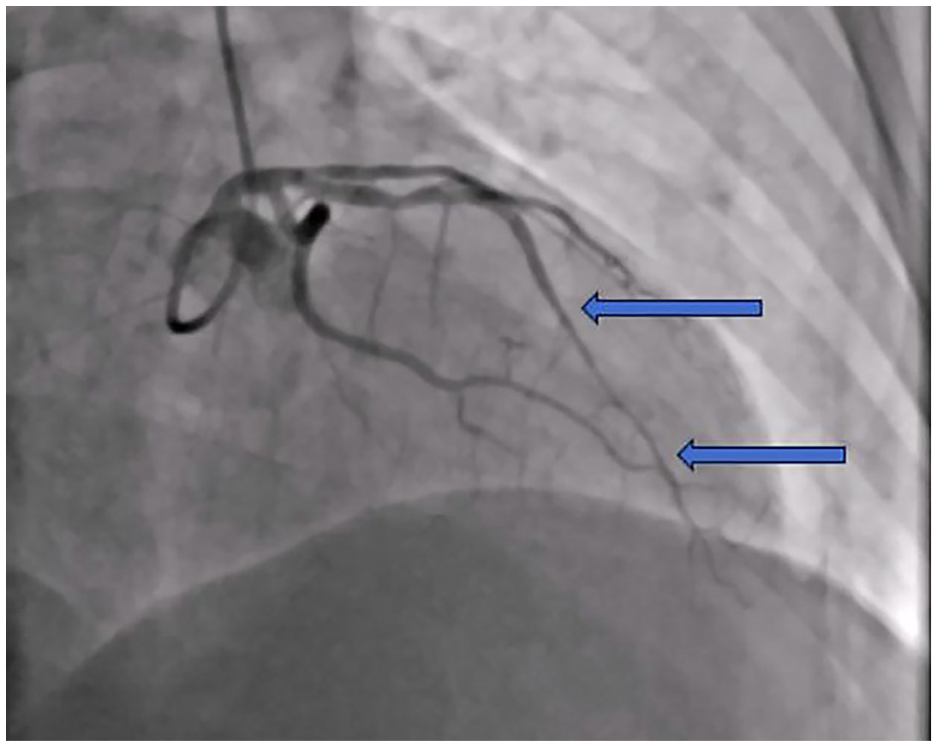
SCAD type 1 (patient 1)—right coronary artery dissection.
SCAD, type 2—long smooth stenosis in left anterior descending artery.
SCAD, type 3 (patient 2)—thrombus and a non-significant lesion in the left main coronary artery.
Various conditions, such as fibromuscular dysplasia, multiple pregnancy, post-partum, chronic systemic inflammatory diseases (Crohn’s disease, systemic lupus erythematosus, sarcoidosis), or connective tissue disorders (Marfan’s syndrome) are assumed to be associated with SCAD.1,5 Up to two-thirds of patients report potential precipitating stressors such as extreme emotional stress, exposure to physical exercise (isometric, especially heavy weightlifting), or drug use (steroids, cocaine, amphetamine). Men with SCAD are generally younger than women and more often have an isometric physical trigger prior to their SCAD event.6,9 The Valsalva maneuver performed during isometric exercises is probably the cause. Additionally, men tend to exhibit more frequent tears on the inner surface of coronary arteries. 10
SCAD frequently affects the left anterior descending coronary artery (LAD), occurring in approximately 50% of cases and in mid to distal coronary segments.2,5 Important differential diagnosis includes atherosclerosis, Takotsubo cardiomyopathy, myocardial infarction with non-obstructive coronary arteries (MINOCA), coronary artery vasospasm, and coronary embolism. 2
SCAD usually manifests as ACS. In the largest Canadian registry covering 750 patients, the ratio of Non-ST-Elevation Myocardial Infarction (NSTEMI) to ST-Elevation Myocardial Infarction (STEMI) was 70% up to 30%. 11 Diagnosing SCAD relies on imaging techniques. Coronary angiography plays a crucial role in identifying the patients suspected of SCAD. It is important to emphasize that coronary angiography should be approached with extreme caution, as there is a reported up to 10-fold higher risk of iatrogenic dissections in this group (2% vs 0.2%).8,12 Intravascular imaging is crucial in diagnosing SCAD and planning the treatment strategy. False lumen with a clear separation of intima and media and visible fenestrations support the typical presentation of SCAD. Patients with type 1 SCAD (multiple radiolucent lumens) do not require intra-coronary imaging to confirm the diagnosis. However, optical coherence tomography (OCT) or intravascular ultrasound (IVUS) may be helpful in diagnosing SCAD with suspected type 2 (occasionally) or type 3 (typically). In IVUS examination, distinguishing between false lumen and thrombus might pose some problems, so OCT is preferred for its higher specificity, despite the need for contrast injections. Also differentiating between SCAD and recanalized thrombus can be challenging. In addition to the clinical presentation of acute coronary syndromes without prior history indicative of SCAD, an irregular lumen with multiple cavities could suggest the presence of an old thrombus. 13
CT coronary angiography (CTCA) is a widely used diagnostic tool in low—and intermediate-risk patients. 1 It is also used as an imaging test during follow-up.
There is a notable scarcity of randomized controlled trials and large-scale prospective studies focusing on SCAD treatment.1,14 Thus, medical treatment poses a significant challenge. In most cases, dissected vessels tend to heal naturally over time. Complete angiographic resolution of the lesion is reported in most cases within 4 to 6 weeks. 1 A conservative approach is primarily favored, especially for clinically stable patients.1,5,15
PCI is associated with worse outcomes. Revascularization is advised only for patients at high risk associated with left main coronary artery dissection, persistent ischemia, severely limited blood flow (TIMI 0 and 1), hemodynamic instability, or refractory arrhythmia.1,9,15 If an invasive strategy is chosen, restoration of TIMI 3 flow with minimal intervention is favored. Intravascular imaging is advocated to avoid entering the false lumen and provide adequate stent size selection. There are reports of cutting balloon usage to fenestrate and drain intramural hematoma with a positive result of the treatment. 16 Difficulties in delivering and implanting stents should be anticipated due to increased shear stress, which may result in a geographic miss or even embolization of the stent. 17
Initial treatment should be identical to standard ACS patients with dual antiplatelet therapy (DAPT) and heparin. Glycoprotein IIb/IIIa inhibitors have also been used without complications. 15 Patients who undergo PCI should be administered DAPT consisting of aspirin and a P2Y12 inhibitor for 12 months. Intravascular imaging with OCT or IVUS is recommended for greater safety and better results in all PCI cases. 15
Recommendations for a conservative approach are based on expert opinion. DAPT with aspirin and clopidogrel is safe and generally accepted. There is no data on the advantage of any antiplatelet drug over the other. 15 In a cohort of 64 patients with SCAD, 59 (92%) received DAPT with aspirin plus either clopidogrel (69%), prasugrel (14%), or ticagrelor (9%). Of the 40 patients who underwent repeat angiography, dissection healing was demonstrated in all but 1. 18
There is no data on how long DAPT treatment should last. However, the available literature analysis shows that the duration ranges from 2-4 weeks to 12 months. Other researchers advocate discontinuing the P2Y12 inhibitor when dissection healing is confirmed.1,7,9,19
If PCI is not done, antiplatelet agents could theoretically be harmful by promoting further hemorrhage into the vessel’s false lumen, preventing intramural hematoma reabsorption and leading to dissection propagation. This theory is supported by the data from the DISCO trial, where DAPT was associated with a higher MACE rate than SAPT, mainly driven by an early excess of non-fatal MI or unplanned PCI. 20 On the other hand, DAPT can be helpful in preventing thrombus formation within the vessel’s true lumen, which seems particularly likely in the case of an intimal tear. Considering different forms of SCAD, it seems reasonable that treatment may depend on its type and cannot be generalized. Pure IMH (type 2 and 3) has a high risk of intramural progression, and here, SAPT may be the best option. On the other hand, when a tear communicating both true and false lumens is present (type 1), a drug-mediated increase of mural bleeding would not have such a clinical impact because the hematoma would be released into the circulation. 20 Randomized trials are needed to answer the questions of whether to use DAPT or SAPT, for how long, and whether the type of dissection should guide the treatment.
Beta-blockers (BB) are recommended for long-term therapy to minimize shear stress on the vessel wall and reduce the risk of IMH propagation. BB therapy prevents the recurrence of SCAD and should be maintained for a minimum of 6 to 12 months. It allows for the spontaneous healing of SCAD lesions. Although there is no firm evidence, statins can be used in SCAD due to their pleiotropic properties.1,14
Case Reports
Case 1
A 56-year-old Caucasian female with a history of diabetes, arterial hypertension, and a smoking habit, in menopause for 4 years, presented with symptoms of STEMI of the inferior wall (Figure 5). She experienced constant pain for an hour prior to admission, with no evidence of heart failure (Killip 1). The patient received saturating doses of dual antiplatelet therapy (aspirin 300 mg and clopidogrel 600 mg) and intravenous heparin bolus (5.000 IU).
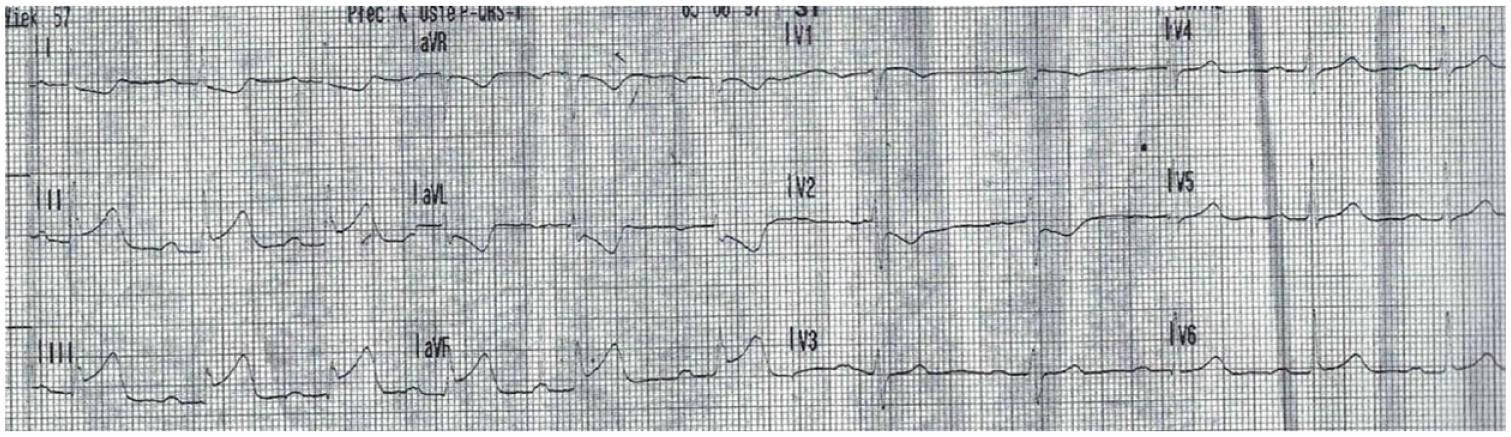
Patient 1—ECG with ST-elevation myocardial infarction of the inferior wall.
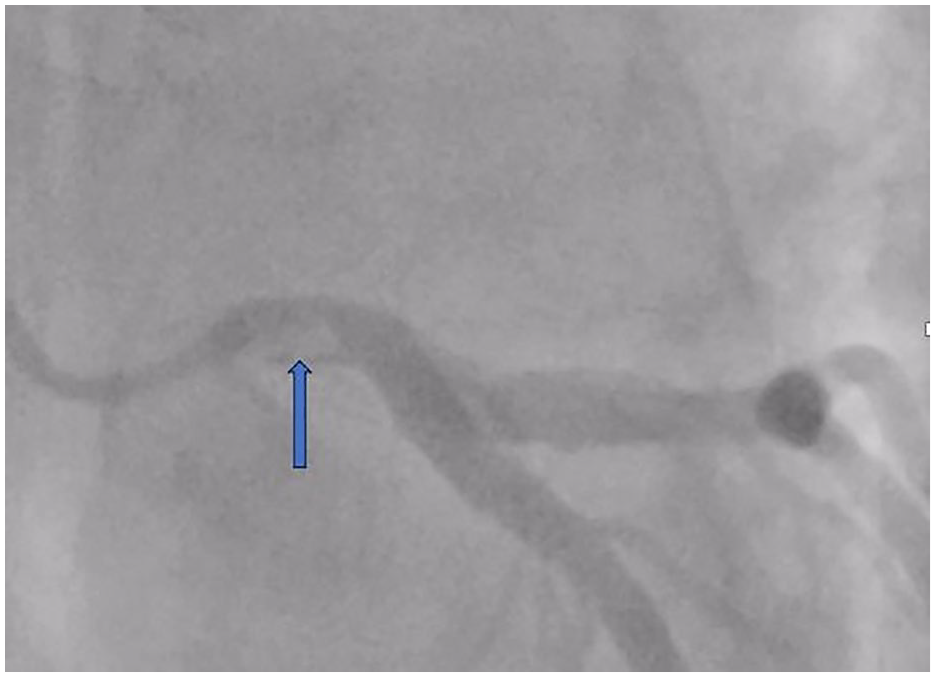
Urgent coronary angiography revealed dissection extending from the ostium to the second segment of the right coronary artery (Figure 2). Due to severe angina, ST-segment elevation, and significant blood flow disturbances, the patient underwent coronary angioplasty. Initial wire placement was within the false lumen, necessitating IVUS use to access the true lumen (Figure 6). Two drug-eluting stents (DES) were successfully implanted under IVUS guidance.
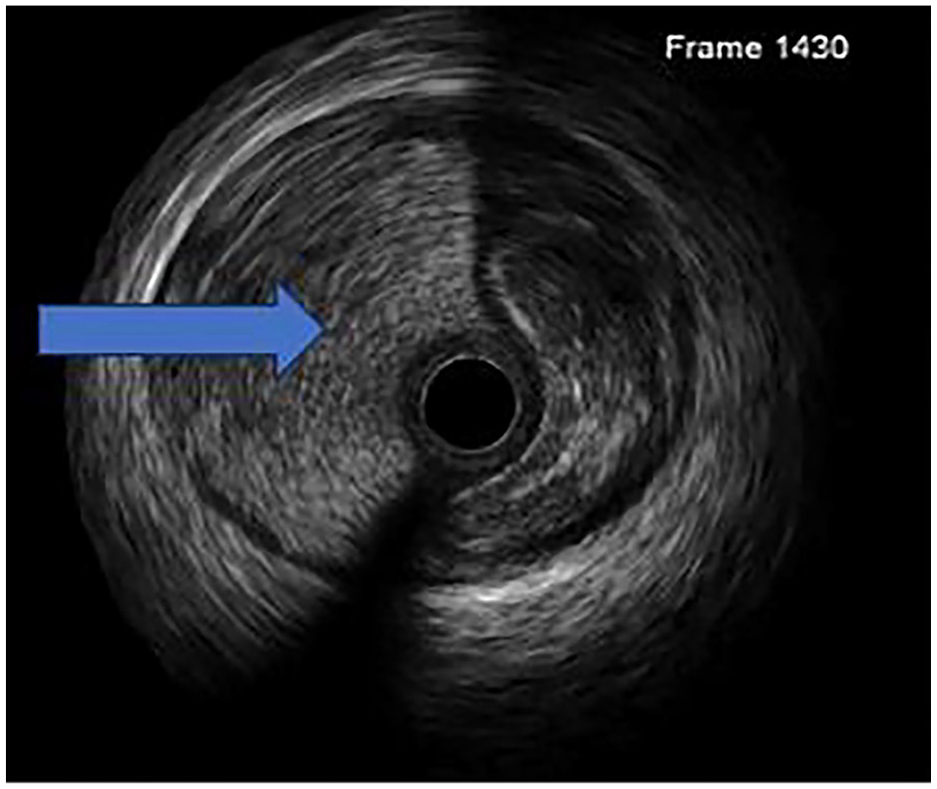
Patient 1—the arrow indicates a false lumen of the dissected artery in the intravascular ultrasound.
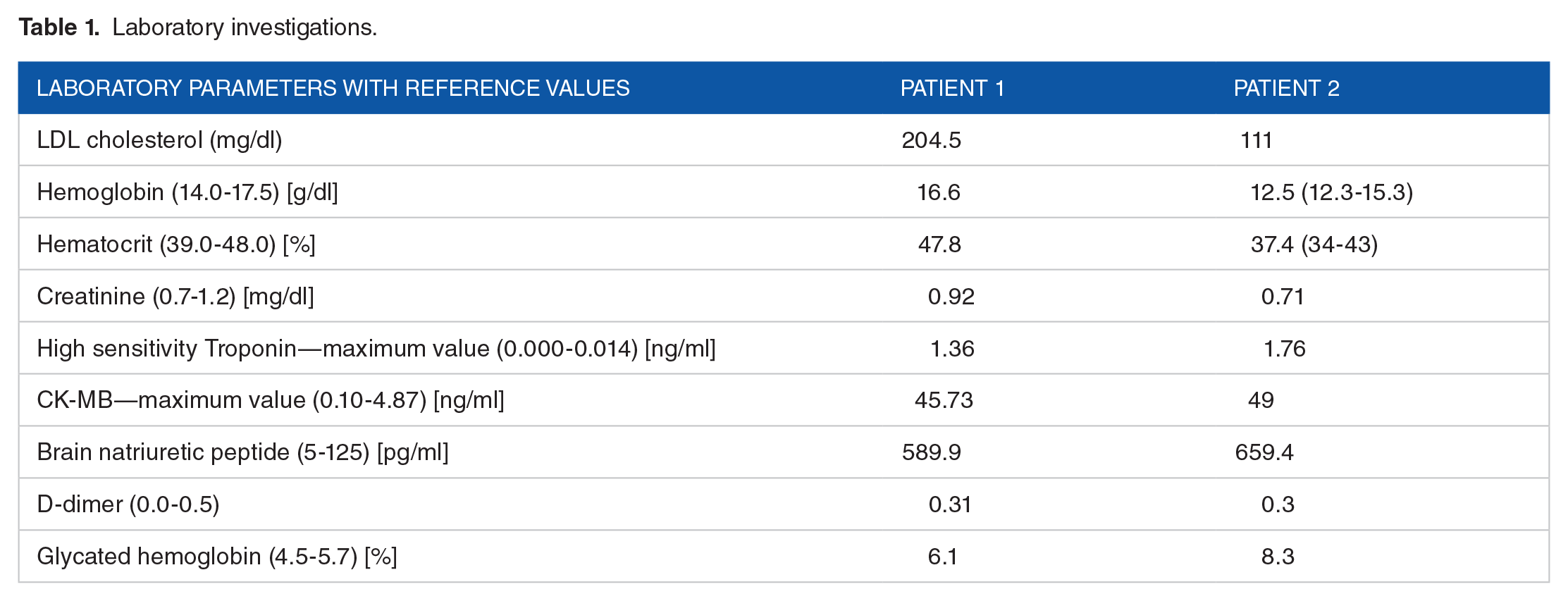
During the next days, the patient remained asymptomatic, and the chest pain did not recur. Echocardiography showed hypokinesis of the basal segment of the inferior wall, with a left ventricular ejection fraction of 50%. Laboratory results, including troponins, are detailed in Table 1. The patient was prescribed DAPT for 12 months. The medications she had previously been taking chronically, including BB and statin, were continued.
Laboratory investigations.
Currently, there is a 4-year follow-up in this patient without reinfarction or other cardiac events.
Case 2
A 32-year-old Caucasian male, previously untreated and an active cigarette smoker, presented to the hospital during the weekend with symptoms suggestive of an acute coronary event. The patient reported feeling generally healthy but experienced recurrent short-lasting episodes of chest pain over the preceding 24 hours, with a notable escalation in severity to constant pain for 1 hour prior to admission. Upon evaluation, he was diagnosed with STEMI affecting the inferior and lateral walls (Figure 7). The patient was hemodynamically stable (Killip 1). Prompt administration of DAPT (aspirin 300 mg and clopidogrel 600 mg) and intravenous heparin bolus (5.000 IU) were initiated. Then, urgent coronary angiography was performed, revealing a thrombus in the left main coronary artery, causing less than 50% stenosis (Figure 4). Since there was no flow impairment, no interventions such as stenting or thrombosuction were performed. The patient was qualified for initial medical therapy and heart team evaluation. He received an Integrilin bolus followed by intravenous infusion, alongside rivaroxaban administration, in the subsequent days. DAPT was continued. The patient’s chest pain resolved during the coronary angiography procedure. When the angiogram was analyzed by a local heart team, apart from the presence of a thrombus, dissection of the left main was noticed. Subsequent CCTA imaging confirmed the dissection of the left main artery protruding into the circumflex artery, accompanied by thrombus formation (Figure 8). SCAD type 3 was diagnosed.
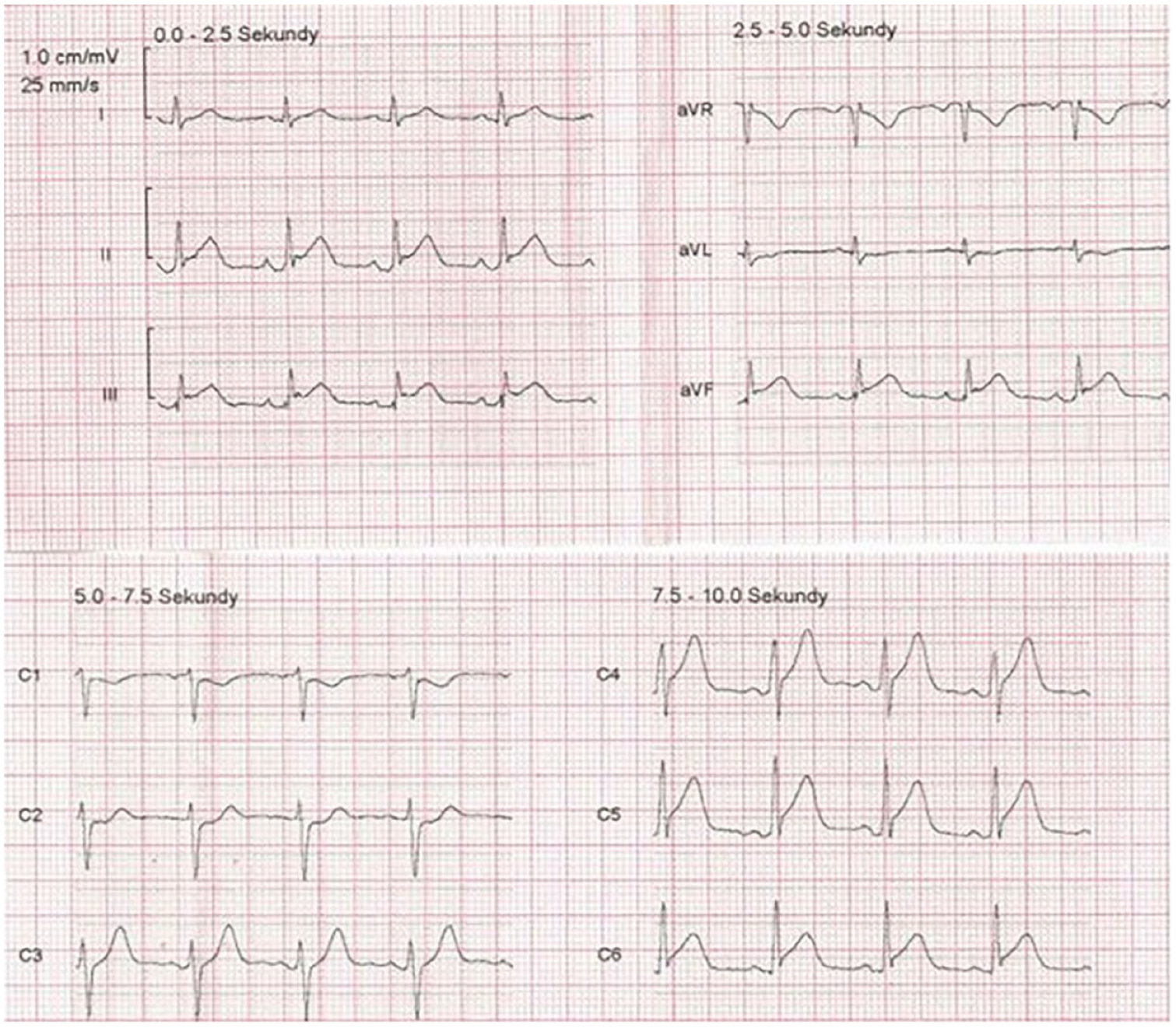
Patient 2—ECG with ST-elevation myocardial infarction of the inferior and lateral walls.
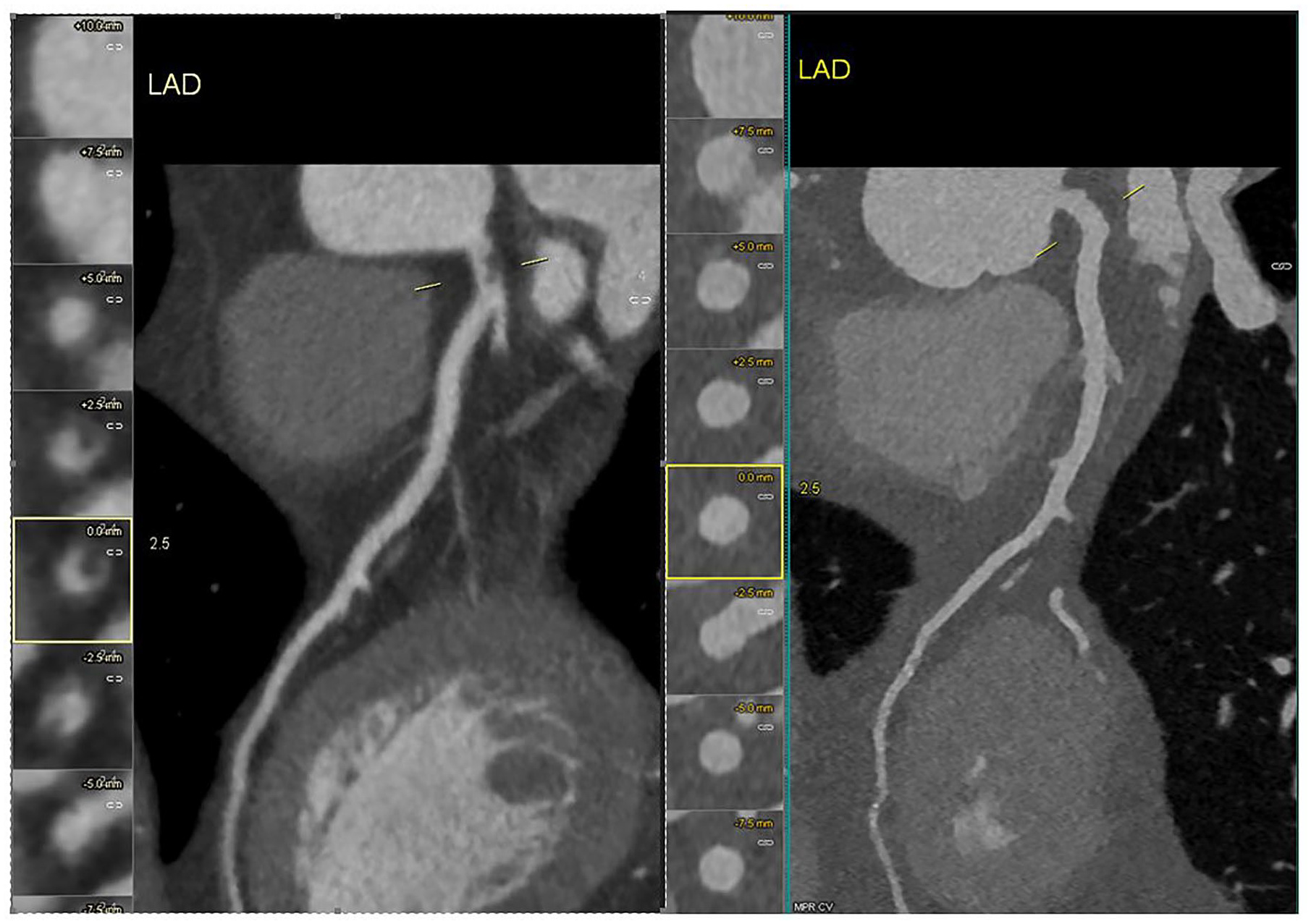
Patient 2—Coronary computed tomography angiography demonstrating dissection of the left main artery protruding into the circumflex artery, accompanied by thrombus formation (left picture). A follow-up scan revealed no pathological changes (right picture).
A detailed interview with the patient revealed a history of regular exercise at the gym, also on the day of admission, and denied any use of stimulants.
Throughout the observation period, the patient did not experience any recurrence of chest pain. The echocardiographic assessment revealed hypokinesis of the apical segment of the lateral wall, with an ejection fraction (EF) of 60%. Laboratory investigations, including troponin levels, are summarized in Table 1. Clinical assessment, including the corridor walk test before discharge home, yielded negative findings. After a week of hospitalization, the patient was discharged home. Due to the thrombus protruding into the lumen, despite the data from the DISCO trial, rivaroxaban 20 mg daily and DAPT were maintained post-discharge. A follow-up CCTA scan conducted 3 weeks after the onset of STEMI revealed no pathological changes (Figure 8). Rivaroxaban was discontinued, and DAPT was continued for 12 months. Additionally, the patient received a statin and a beta-blocker as part of their medication regimen. Due to an elevated HbcA1 low glucose diet and re-examination was recommended.
Currently, there is a 1-year follow-up in this patient without re-infarction or other cardiac events.
Discussion
The main message of this paper is that an individual approach and awareness of SCAD is necessary in the case of ACS without typical atherosclerotic lesions. The disease has different faces; depending on its type and the patient’s clinical manifestation, the diagnostics and treatment should be individualized. It should also be remembered that, despite common knowledge, the disease is not limited to young females.
In the case of STEMI, diagnostics starts with coronary angiography. In type 1 of SCAD, as in the first case, coronarography alone is sufficient to make a diagnosis. Due to ongoing angina, flow impairment, and ST-segment elevation, an invasive strategy was chosen. The intervention was IVUS-guided to identify and enter the true lumen and choose adequate stent size and length. When stent implantation is necessary, it is advisable to use the appropriate length (far before and after dissection) to prevent hematoma propagation or implantation of a short stent for coverage only at the gate of dissection. Finally, using intravascular imaging to show the true lumen and dimensions of the dissected vessel should be mandatory.8,12,15 All of these principles were met in the case of the first patient.
Additional imaging techniques may help establish the other types of SCAD. Even though coronary angiography in the second case was performed and analyzed by an experienced interventional cardiologist, the initial diagnosis was not SCAD but an isolated thrombus in the left main artery. This emphasizes the diagnosis difficulties, especially in type 3 SCAD, where it might be mistaken for an “ordinary” atherosclerotic lesion. To confirm the diagnosis of type 3, as in the second case, CCTA was performed. To shorten the time of that treatment to a minimum, control CCTA was performed 3 weeks after the onset of STEMI, and antithrombotic therapy was stopped after confirming the thrombus resolution. From today’s perspective, intravascular imaging should be performed to confirm the diagnosis during the initial procedure. As for the treatment, it seems that after the resolution of the thrombus, we could have discontinued not only the anticoagulant drug but also one of the antiplatelet drugs.
To our knowledge, no data on managing thrombotic lesions accompanying SCAD exists. In our opinion, in the case of dissection and thrombus, direct oral anticoagulants may be appropriate analogously to medical treatment of intraventricular thrombus, with CCTA monitoring the progress of therapy. However, randomized trials are needed in this area.
We conclude that different presentations of SCAD, not limited to Yip-Saw’s classification and clinical presentation but taking into consideration localization of the lesion and presence of intraluminal thrombus, require different treatment strategies. Figure 9 presents a proposal for the management of SCAD based on the current guidelines, a literature review, and our practice.

Proposal for the management of SCAD based on the current guidelines, a review of the literature, and our practice.
Footnotes
Acknowledgements
None
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization: MW and MZ; Methodology: IP, MD, MK; Validation: MW and MZ; Formal analysis: MW and MZ; Investigation: KK, KM; Resources: IP, MD, and MK; Data curation: IP, MD, and MK; Writing—original draft preparation KK, MW, KM, and IP; Writing—review and editing: MD, MK, and MZ; Visualization: KM and MZ; Supervision: MW and MZ. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
All data and materials generated or used during the study appear in the submitted article.
Ethics Approval
Not applicable
Informed Consent
Written informed consent was obtained from the patients for the anonymized information to be published in this article.
Use of Artificial Intelligence
AI was not used in the process of writing or editing of the text.
