Abstract
Background:
Fixed drug eruption (FDE) is a drug hypersensitivity reaction caused by antibiotics and non-steroidal anti-inflammatory drugs primarily. Antondine injection is a drug used to reduce fever, mainly composed of aminopyrine, antipyrine and barbiturate, and its adverse reactions include anaphylactic shock, epidermolysis, rash, agranulocytosis, etc. FDE caused by Antondine injection is an uncommon phenomenon, and the drug eruption caused by these ingredients has been reported in the current case report. However, the fatal severe fixed drug eruption caused by it has not been reported. Here, we report a case of severe PDE after treatment with Antondine, highlighting it’s necessary to avoid these medications with this ingredient when using antipyretic medications.
Introduction
Fixed drug eruption is a unique type of adverse drug reaction, characterized by a rash on a fixed part of the body each time, when a specific drug is taken.1-2 There may be overlying vesicles and/or bullosa in the lesion, and when they cover 10% or more of the body’s surface area or more than 4 anatomical sites, this rash is called a generalized bullous fixed drug eruption (GBFDE).3-4 Non-steroidal anti-inflammatory drugs and antibiotics are the most common causes of FDE.1,5 Antondine injection, also known as compound aminobarbital injection, is a drug mainly used for emergency fever reduction in acute high fever, doctors should be aware of the possible skin adverse reactions, such as rash, urticaria, epidermolysis, and so on, and avoid to use this drug as much as possible.
Case presentation
A 61-year-old male with fever and body temperature of 38.5℃ received an injection of Antondine into the hip muscle at the clinic. Subsequently, swelling and pain occurred at the injection site, accompanied by loss of appetite, weakness of limbs, abdominal pain, swelling pain under xiphoid process and periumbilical cord, accompanied by yellow hydrolytic stool 4-5 times per day, and the patient was hospitalized in a local hospital. During the hospitalization, the patient still had fever and body temperature was up to 40℃. The patient developed back lesions (10 days after injection of drugs), and his condition did not improve after symptomatic treatment such as anti-infection and anti-diarrhea, and he was admitted to our hospital for treatment, the chest and abdominal CT indicated double lung infection and incomplete intestinal obstruction. He had suffered from hepatic hemangioma, hepatic cyst and gastric ulcer previously (occasionally abdominal distension, abdominal pain, no black stool, bloody stool).
The patient was admitted to our department for monitoring and treatment due to fever, significantly increased breathing and altered consciousness, considering sepsis. Physical examination: clear mind, listless spirit, T: 38.2℃, R: 30 times/min, P: 95 times/min, BP: 101/67 mmHg, 4 skin lesions of different sizes were found on the back (Figure 1). The abdominal physical examination: light tenderness under xiphoid process and around umbilicus, no rebound pain and muscle tension. The APACHII score was 16, and the mortality risk coefficient was 53.59%.

Four skin lesions of different sizes were found on the back.
After admission, the biopsy of back lesions and buttock mass was completed. The biopsy of buttock mass showed acute inflammatory cell infiltration of subcutaneous fat, which was consistent with panniculitis. A biopsy of the lesion in the back revealed diffuse infiltration of acute inflammatory cells (Figure 2). At the same time, the NGS of blood was negative. Relevant departments were invited for consultation, based on the medical history, physical signs and auxiliary examination, the back lesions were considered to be fixed drug eruption caused by Antondine injection. In terms of treatment, high-flow oxygen inhalation, anti-infection, acid inhibition and stomach protection, immunoglobulin and hormone impact therapy, and maintenance of stable organ function were given. After the above treatment, the patient’s condition did not improve, the degree of back lesions expanded, septic shock symptoms appeared, the dynamic APACHII score was 20, and the mortality risk coefficient was 68.35%. Changes of ICU clinical treatment indexes in our hospital (Table 1). The patient was discharged from the hospital due to his own reasons and died on the first day after discharge after follow-up.

Pathological section.
Laboratory tests during our ICU.
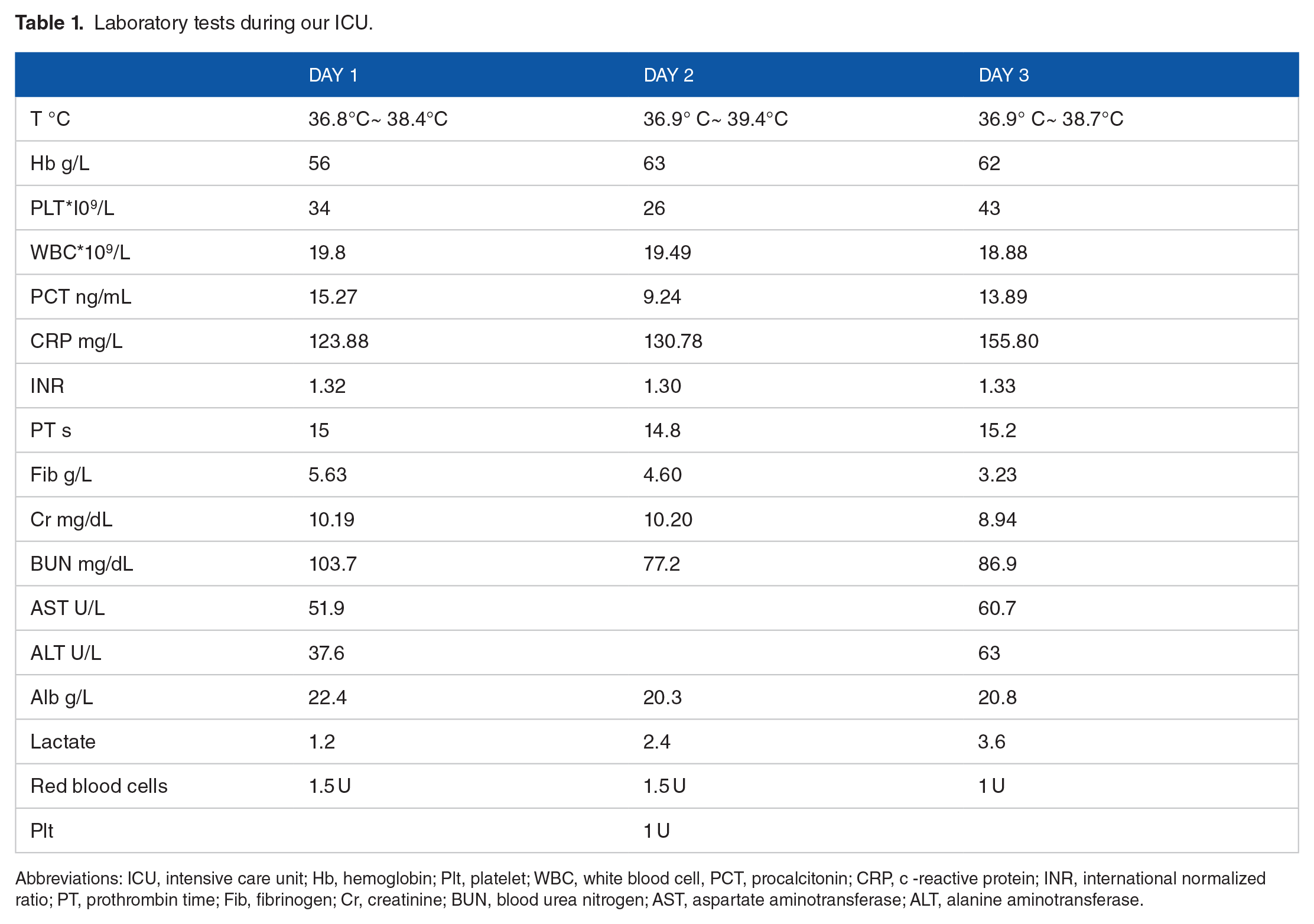
Abbreviations: ICU, intensive care unit; Hb, hemoglobin; Plt, platelet; WBC, white blood cell, PCT, procalcitonin; CRP, c -reactive protein; INR, international normalized ratio; PT, prothrombin time; Fib, fibrinogen; Cr, creatinine; BUN, blood urea nitrogen; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
Disscussion
A Fixed drug eruption (FDE) is a unique cutaneous adverse drug reaction that typically takes the form of a solitary, well-defined, erythema to purple, round to oval patch with a dark center that may appear as a blister, vesicle, and/or bullosa that is prone to rupture, leaving erosion or shallow ulcers.1 -3,6 The pathogenesis is still unclear, but current studies suggest that type IV hypersensitivity reaction to certain drugs or their metabolites is mainly mediated by CD8+, in which antibiotics and non-steroidal anti-inflammatory drugs are the main drugs.1,4-5,7 FDE can be confirmed by oral stimulation or patch tests, and oral challenges are the gold standard for diagnosis, but now they are often prohibited due to the risk of causing widespread FDE or GBFDE.2,8-9 It is usually distinguished from generalized bullous fixed drug eruption (GBFDE), Steven-Johnson syndrome, or toxic epidermal necrolysis.8,10
FDE can appear on any part of the body, and the most common areas include the lips, genitals, perianal area, hands, and feet.11-12 In this case report, the patient developed mass in the buttock after intramuscular injection of Antondine, accompanied by general fatigue, poor appetite, abdominal pain and other symptoms. At this time, He maybe have had adverse drug reactions. On the 10th day after injection, the patient gradually developed back lesions, and his condition did not improve after anti-infection treatment in other hospitals, then he was transferred to our department for supervision and treatment. Blood examination during hospitalization indicated severe anemia, thrombocytopenia and abnormal liver function (Table 1), combined with pulmonary infection and incomplete intestinal obstruction. After comprehensive patient history, physical signs and auxiliary examination, fixed drug eruption was considered after multidisciplinary consultation, and immunoglobulin and hormone shock therapy were added on the basis of anti-infection and organ function maintenance. The lesions increased further, septic shock appeared, and finally the patient was discharged due to his own factors. During follow-up, the patient died on the first day after discharge.
Antondine injection is a clinically used drug for emergency fever reduction, mainly composed of aminopyrine, antipyrine and barbiturate, it has been reported that these ingredients can cause drug eruption at present.13-14 An extensive review of the literature, we found that: the Antondine injection can cause serious drug side effects, such as anaphylactic shock, epidermolysis, rash, urticaria, agranulocytosis, purpura, etc.13-14 The World Health Organization clearly prohibits the drug for children, while some countries around the world have suspended its use, and China’s Ministry of Health also announced the elimination of this drug in 1982. However, it is still used in some county or township health centers.
At present, the main treatment for FDE is to remove the causative drug, but sometimes it is difficult to identify the causative drug, in this case it is necessary to provide a list of cross-reactive drugs as much as possible.1-7 The mainly treatment to relieve itching or pain with topical/systemic or oral corticosteroids and/or antihistamines, while in severe cases, intensive care should be considered.4,7,9 -11
Unfortunately, although the cases we reported received intensive care and related treatment in our hospital, they could not be successfully treated because they could not be effectively identified and treated at an early stage.
Conclusions
Through this case, we emphasize that the must be aware of the potentially serious side effects of this class of drugs. It is recommended that avoiding to use these such drugs is a necessary measure to prevent recurrence, and strengthen the supervision and early intervention of drug side effects.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chongqing Medical Key Discipline Development Project (No.zdxk202102).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors contributions
HT and ZW: review of the article, correspondence. YY: writing of the article, data collection. P-FL and QC: data collection. X-NW and L-YM: image processing. QM: contribution to the pathology part of the article. YY and P-FL: treat this patient. Ethical Review The ethics committee of Chongqing Army Specialty Medical Center has passed the ethical review.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The study has been approved by the Committee of Research Ethics of Army Medical Center of PLA, and informed consent was obtained from the relative of the patient.
Consent for publication
Written informed consent for publication of the clinical details were obtained from the relative of the patient.
