Abstract
Background:
Uterine fibroids, or Leiomyoma is a type of Smooth Muscle Tumors of the uterus (SMTs) and are common in the black race. Giant uterine fibroids, on the other hand, are uncommon and may occur during patient dissimulation. Dissimulation may occur because of a dread of surgery and hospitals visits, fear of surgical death, chronic intake of herbal concussion, and a religio-traditional strong belief system on instant healing following prayers, among others. Myths like belief of defecating the uterine fibroids, some herbs that can melt them away, and the belief that such illness may follow ancestral curses can fuel dissimulation. The surgical approach can be a source of challenge, careful case selection considering the size and number of tumors can be helpful.
Case report:
We present a 35-year-old nulligravida who presented to the clinic with a 14-year history of progressive abdominal swelling. Examination revealed a firm mass with a symphysio-fundal height of 55 cm. She subsequently had an open abdominal myomectomy with all the myoma nodules weighing 12.9 kg in total! Histology confirmed uterine fibroid.
Conclusion:
It is possible to offer open myomectomy in patients with giant uterine fibroid following careful patient selection with a consent for possible hysterectomy. Dissimulation can be minimized with repetitive counseling of patients. The choice of surgery depends on the size and number of uterine fibroids, but surgical approach does not necessarily influence fecundity.
Introduction
Uterine fibroids (UF), also known as uterine leiomyoma, are one of the most common types of uterine tumors. 1 Giant uterine leiomyomas are classified as weighing 11.4 kg or more.2,3 The largest uterine fibroid ever documented in the literature weighed 63.3 kg and was removed postmortem in 1888. 2 The largest uterine tumor ever removed from a patient who survived the treatment weighed 45.4 kg, making it the 34th largest tumor weighing more than 18.2kg removed since 1878. 3
UF is a critical global women’s health issue with significant physical, emotional, and socioeconomic consequences. Racial differences, age, race, hormonal variables, obesity, and lifestyle (diet, physical activity, and stress) are all risk factors for UF. 1 They are more common in Africans and people of the black race, and they appear later and in larger proportions. 4
Fibroids of enormous size are an uncommon benign tumor variation of normal uterine muscle. Patients with inertia or dissimulation may develop them, and their treatment is generally surgical. 5 Because of the broad and well-established use of ultrasonography in gynecological clinics, their diagnosis is usually not missed. 6 These tumors affect about half of all women over the age of 35, with a higher incidence during the reproductive period due to hormone-stimulated growth.3,6 Notably at the age of 50, 80% of African women and over 70% of Caucasian women have uterine fibroids. 7 It has many treatment approaches ranging from careful watchful waiting to medical, then to surgical procedures with hysterectomy serving as the only definitive treatment. 8 A veritable consideration that affects the choice of surgical approach is the size and number of uterine fibroids. 9 Important to note that the type of surgical procedure used does not influence the pregnancy rate. 9
Case Presentation
We present a 35-year-old nulligravida who came to the outpatient clinic of Havana Specialist Hospital, Surulere Nigeria, West Africa with 14-year history of progressive abdominal swelling. The abdominal swelling has grown over the past years to reach her epigastric region with associated shortness of breath especially on exertion. There was occasional diffuse abdominal pain with no change in her menstrual pattern. She has a 28-day regular menstrual cycle with 4 to 5 days of moderate flow. She admitted to intake of some herbal products with undisclosed names. She has no drug allergy. There was no constipation but had easy satiety. There was no progressive weight loss, foul smelling vaginal discharge or yellowness of the eyes. She is not a known hypertensive or diabetic mellitus patient. She has no history of surgery or blood transfusion in the past. The patient admitted being afraid of possible surgery and hence stayed away from hospital premises and consultation over a period of 14 years. She does not smoke cigarettes or tobacco in any form. She is aware of modern contraception but does not use any. She is married in a monogamous family setting. Physical examination revealed a firm abdominal mass with a symphysio-fundal height of 55 cm (Figure 1). The mass was non tender. There were epigastric and hypochondria fullness. Could not get below the mass but could get above it just about 2 cm short of the xiphisternum.

Before uterine incision, a 55 cm symphysio-fundal distance.
Investigations
Ultrasound scan—Revealed no free peritoneal fluid, normal bowel loops, no mass on the bowel nor any para-aortic lymphadenopathy seen. The liver was of normal size with a span of 133 mm having a smooth outline and normal parenchymal echogenicity. There was no focal lesion seen. The intra and extra hepatic ducts and vessels were normal. The kidneys were normal in size with normal cortico-medullary differentiation. There were no masses nor calculus seen. The urinary bladder wall was smooth and of normal size. There was no mass or calculus seen within it. The uterus was markedly enlarged at 330 × 290 × 267 mm with normal endometrial plate thickness measures 9.67 mm. There were submucous and intramural oval hypoechoic nodes less than 10 mm, some with echogenic rings, some with echo free areas located in the fundal, anterior, and posterior aspects with sizes ranging from 33 × 31 mm to 138 × 133 mm. The ovaries were visualized and normal in size and echotexture. There were no adnexal masses seen, the pouch of Douglas was clear.
She had Axial T1, axial T2, Coronal T1, coronal T2, MRCP 3D Magnetic Resonance Imaging (MRI) which revealed an enlarged uterus extending to the right sub hepatic and left hypochondriac regions with heterogenous myometrium due to multiple myomata with the following locations [fundal sub-serosal—26 × 170 mm; anterior sub serosal—151 × 148 mm; posterior intramural 118 × 84 mm; posterior intramural 45 × 38 mm; 49 × 25 mm; right lateral intramural—49 × 37 mm; anterior intramural—57 × 36 mm; 54 × 35 mm; anterior cervical—205 × 176 mm] . distorted endometrium. The ovaries were not visualized with the enlarged uterus demonstrating a mass effect on the kidneys, stomach, and urinary bladder. The liver had a normal span of 150 mm with normal homogenous signal intensity. There was no mass seen within the parenchyma. The spleen, gall bladder, pancreas, stomach, small and large bowel were normal. There was no area of peritoneal fat stranding, ascites or adenopathy noted. The colon showed good distension with no mural lesion or area of structuring or mucosal outpouching noted within it. The kidneys were normal in size, outline, position, and excretory function with no mass or pelvicalyceal dilatation in either kidney. The adrenal glands were normal bilaterally with no mass noted within it. The urinary bladder was normal. There were no adnexal mass(es) seen.
The tumor markers revealed CA 125 of 48.7/ml (0.0-35); alpha fetoprotein of 3.0 kU/l (<7.3); CEA (Carcino-embryonic antigen) of 4.8 µg/l (non-smokers—0-5.0 µg/l); Her full blood count, kidney function and liver function were essentially normal. She was assessed for HIV I and II and were negative.
She was counseled and informed consent was obtained for open myomectomy with possibility of an abdominal hysterectomy under General anesthesia with endotracheal intubation. The general surgeons and urologists were informed for a multidisciplinary approach intraoperatively.
Differential Diagnosis
We suspected a possibility of a leiomyosarcoma in this patient owing to its size. Patient had investigations which dismissed the possibility of leiomyosarcoma. Though serum LDH could not be assayed due to cost, an investigation would have aided in diagnosis of possible leiomyosarcoma. MRI findings did not typify possible leiomyosarcoma.
Treatment
The management plan for such a giant leiomyoma uterus was explained to the patient which is usually a hysterectomy. The patient needed uterine conservative surgery, which in this case was an open myomectomy as she expressed her desire to have children in the future. The fact that an open myomectomy can be attempted was explained to her with a low threshold for a hysterectomy. A written consent for these surgeries and anesthesia were obtained. She subsequently had open abdominal myomectomy under general anesthesia with endotracheal intubation using a midline incision with a supraumbilical extension up to the xiphisternal prominence.
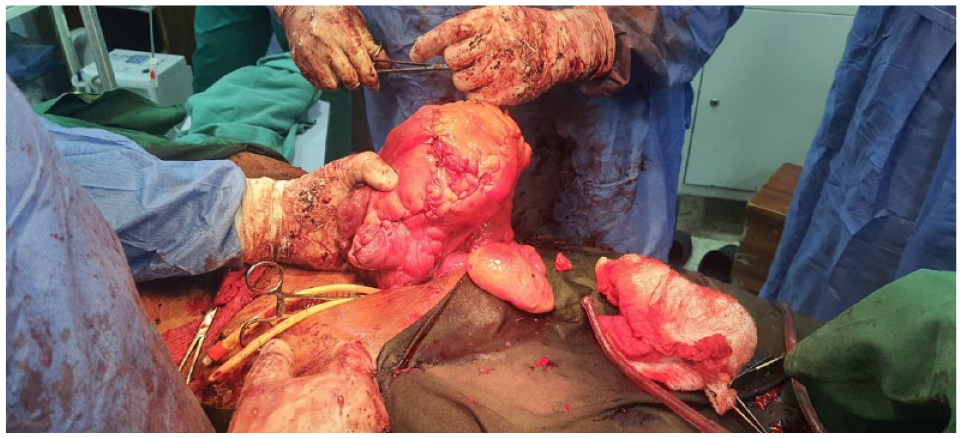
The intrabdominal findings were a hydra headed uterus with giant myoma nodules ranging from pedunculated variety with a sessile base, intramural types, sub serous and submucous types with all the myoma nodules weighing 12.9 kg with the largest weighing 5.7 kg and smallest 400 g (Figures 2 and 3). The hydrosalpinx of the left tube was seen while the right tube and right ovary were grossly normal. The left ovary appears enlarged. The gut and liver, omentum, stomach all appeared normal with no deposits seen.

Giant uterine fibroid showing application of large sized clamps for dissection of a part of the pedunculated fibroid with a sessile base.

The whole uterine fibroid mass showing weight (12.9 kg) on weighing scale.
The uterine mass was held up by 2 assistants following which a torniquet (size 22 F foleys catheter) was applied at the isthmic region of the uterus. The 3 very large pedunculated uterine fibroid nodules were removed first, and hemostasis secured.
An extensive hood incision was made through which all the other uterine fibroid nodules were enucleated, mainly of intra mural variety. The endometrium was explored and a 1 × 1 cm submucous fibroid was found and removed. Uterine reconstruction surgery was done meticulously with Vicryl #3/0 for the closure of breeched endometrium. All the dead spaces thus left after enucleating the uterine fibroids were obliterated with Vicryl 2. The serosa was closed with Vicryl 2/0 in a baseball fashion as proposed monofilamentous sutures were unavailable (Figure 4).
Post uterine reconstruction.
Surgicele was placed on some of the raw looking repaired serosa wound. The torniquet was released about 1 hour 20 minutes and hemostasis secured. After confirming that no contiguous structures were injured and the instruments and gauze were counted and noted to be complete, the peritoneal cavity was copiously lavaged with saline and a closed Penrose drain placed in the Pouch of Douglas and was left in situ. The abdominal cavity was closed in layers and sterile dressing applied on skin wound.
Outcome and Follow Up
She was monitored in the High Dependency Unit (HDU) post operatively and later stepped down to the ward following observation of stable clinical state. Her post operative packed cell volume was 20%, following which patient had a total of 5 units of packed cells transfused while on admission. The Penrose drain was removed on day 3 post surgery. She was very satisfied with the outcome of her surgery and was discharged on day 5 post surgery with a post transfusion packed cell volume of 27%. She is seen at the Gynaecologic clinic 2 weekspost surgery on follow up. She had no complaints; operative site was well apposed.
The histology result was discussed with her. Macroscopy: About 36-week size uterine fibroid. Specimen consists of multiple nodular masses weighing 12.9 kg. The largest measures 27 × 19 ×12 cm while the smallest measures 2 ×2 ×2 cm. The cut sections showed solid grayish white surfaces with whorled appearances and cystic degeneration. Microscopy: Interlacing bundles and fascicles of mature spindle shaped smooth muscle cells. No cellular atypia was seen (Figure 5). Diagnosis: Leiomyomata (Figures 6 and 7).

A month follow up showing healed midline scar with supra umbilical extension.
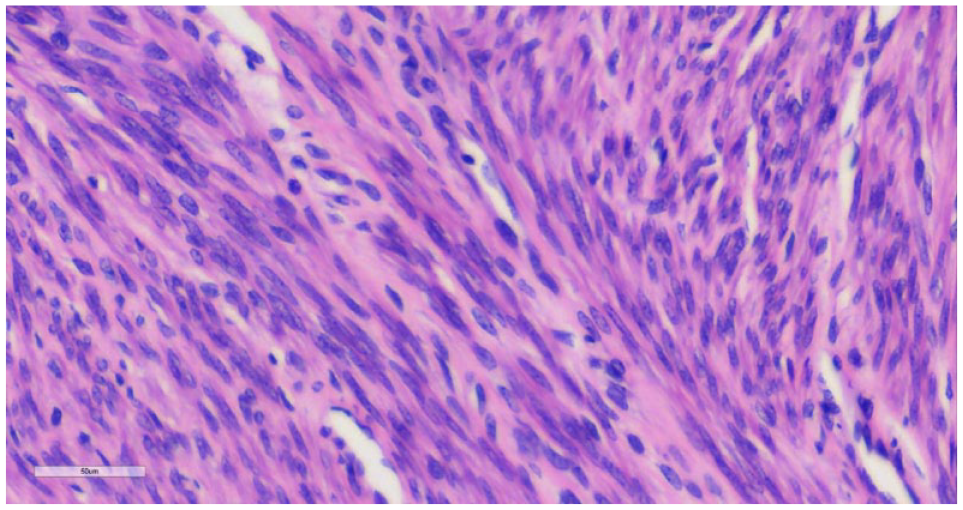
Micrograph showing interlacing fascicles of smooth muscle cells with interspersed blood vessels at x400 magnification.

Histopathology report of index patient.
She was seen 2 weeks afterwards, then a month after her surgery. Her skin wound has healed satisfactorily, and she was happy with the surgical outcome (See Figure 6). The patient was encouraged to come to the hospital after 3 months of surgery to further discuss her fertility options.
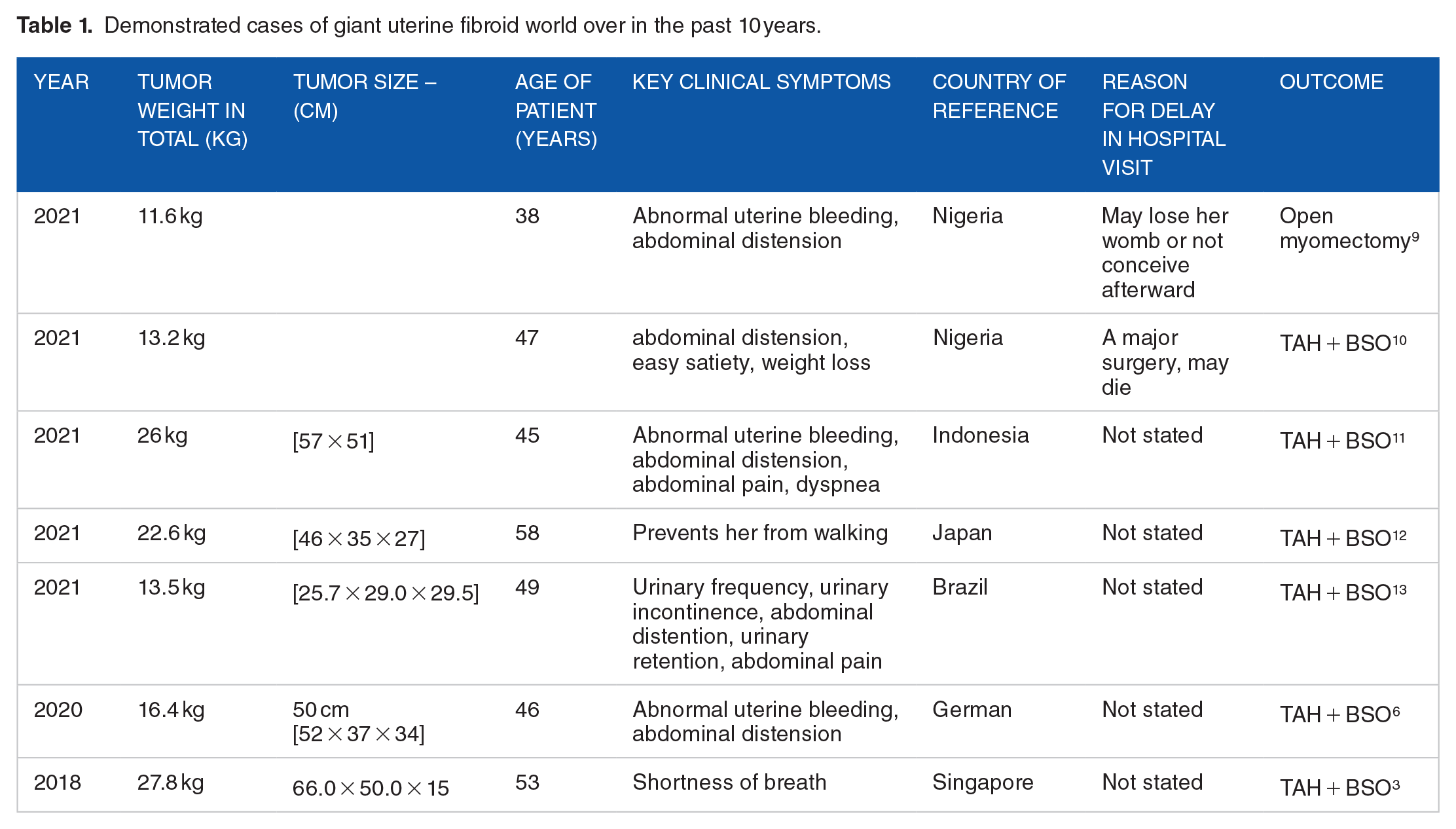
Our case merited a diagnosis of Giant uterine fibroid having surpassed 11.4 kg or more.2,3 We measured a symphysio-fundal height of 55 cm in the clinic and the mass weighed 12.9 kg intraoperatively. This uterine mass was less than the largest uterine fibroid mass ever reported in literature by Evans and Pratt, 2 but greater than 11.6 kg according to Ikechebelu et al report. 9 In 2021, a Pubmed search reported by Viva et al demonstrated a PubMed database recorded approximately 60 cases of giant uterine myomas world over in the past half a century. 6 Table 1 demonstrated cases of giant uterine fibroid world over in the past 10 years (Table 1).
Demonstrated cases of giant uterine fibroid world over in the past 10 years.
Due to the large size of the uterine fibroid, we entertained a possible diagnosis of Leiomyosarcoma. Nagai et al presented a Preoperative Sarcoma Score (PRESS) in 2014 to aid in the prediction of uterine sarcoma.2,14 At age over 49 years old, serum LDH above 279 U/L, positive endometrial cytological findings, and MRI findings showing high intra-tumoral signal intensity on T1 weighted images indicating intra-tumoral hemorrhage and coagulative necrosis, or a heterogeneous mass on T2 weighted images were all predictive factors in PRESS score. This patient’s MRI did not show area (s) of coagulative necrosis or hemorrhage and our patient was a 35-year-old nulligravid lady. Other tumor markers however were essentially normal.
This PRESS scoring system was updated in 2015 with a score of 4 points or higher indicating a higher risk of uterine cancer positive predicting sarcoma with a suggested accuracy of 93.7%, 92.3% positive prediction value and 94.0% negative predictive value.2,15 LDH assay is expensive in our environment, and we did not consider it expressly due to the MRI findings which were nonsuggestive of a possible leiomyosarcoma. Sarcoma occurs in 1%-2% of postmenopausal women and has a rapid doubling time of 4 weeks. 16 The leiomyoma in this pre-menopausal nulligravid lady had a slow but steady growth over a period of 14 years.
Uterine leiomyoma is the most prevalent type of pelvic tumor in women and estimated that by age 50, around 70% of women will have uterine leiomyoma. 2 They are benign monoclonal tumors that develop from myometrial smooth muscle cells. They usually manifest with atypical uterine hemorrhage, pressure symptoms, or reproductive abnormalities, such as infertility or obstetric difficulties. According to the International Federation of Gynaecology and Obstetrics (FIGO) classification, they are classified as sub-serosal, intramural, submucosal, or cervical myoma based on their position in the uterus. 17
It is interesting to know that leiomyosarcoma is one of the Uterine smooth muscle tumors (SMTs). SMTs are a group of neoplasms that originate from the smooth muscle cells in the wall of the uterus. 18 Since they promote the proliferation of uterine smooth muscle cells with more estrogen and progesterone receptors than normal uterine smooth muscle, estrogen and progesterone may be crucial in the formation of SMTs. The formation of SMTs is influenced by cytokines and local growth factors, like interleukins, vascular endothelial growth factor (VEGF), and transforming growth factor-beta (TGF-β) respectively.18-20
While phosphorylated proteins are implicated in tumorigenic signaling pathways, anti-apoptotic mechanisms, and cell survival in leiomyosarcomas, MED12 gene mutations and chromosomal abnormalities are frequently observed in leiomyomas. 21
Abnormal vaginal bleeding, pelvic pain, and a quickly expanding uterus are possible symptoms of leiomyosarcomas. Surgery is the normal course of treatment, while chemotherapy and radiation are other options. 22 Index patient had a steady growth of her tumor over a period of 14 years and had no menstrual changes rather than steady but slow growth of her abdominal girth. This was different from symptoms of the 38-year-old reported by Ikechebelu 9 who presented with abnormal uterine bleeding but had similar abdominal distension. Our patient was offered open myomectomy due to her desire for childbirth and intraoperative findings of mostly giant subserosa myoma variety with intact endometrium.
Certain distinguishing characteristics, such as cytological atypia, the mitotic index, and the existence of coagulative tumor cell necrosis (CTCN), serve as the basis for the histological distinction between leiomyoma and leiomyosarcomas. 18
Bell et al characterized 4 histological subtypes of troublesome SMTs. 22 Less than 10 MFs (Mitotic Figures)/10 HPFs (High Power Field), diffuse moderate-to-severe atypia, and “atypical leiomyoma with low risk of recurrence” (AL-LRR) are the characteristics of the first groups. As well as the lack of CTCN. Under the second group, “atypical leiomyoma with limited experience” (AL-LE), there is no CTCN, less than 10 MFs/10 HPFs, and localized or multifocal severe atypia. The third classification, known as “smooth muscle tumor with low malignant potential” (SMT-LMP), is distinguished by the presence of CTCN, less than 10 MFs/10 HPFs, and absent or minor atypia. Lastly, there is no indication of tumor cell necrosis or atypia in the fourth group, “mitotically active leiomyoma with limited experience” (MAL-LE), which shows more than or equal to 20 MFs/10 HPFs. The histology of index patient showed interlacing fascicles of smooth muscle cells with interspersed blood vessels and no atypia.
The World Health Organization (WHO) 18 recently launched a new category of SMTs termed “smooth muscle tumor of unknown malignant potential” (STUMP). STUMPS demonstrate some histological features that show possible malignant behavior, but do not meet all the Stanford diagnostic criteria for leiomyosarcoma. They therefore do not match the traditional categories of benign or malignant. Despite the major role that imaging techniques play in the diagnosis of SMTs, the diagnosis of STUMP can still be challenging due to a blur line between benign and malignant tumors imaging features. The optimal management of STUMP has polarized opinion till date.
It is important to let the patient know during her several clinics that management of giant leiomyoma uteri is a tough operation with the danger of major bleeding and the need for possible blood transfusion. If the uterine bleeding cannot be managed intraoperatively, an early recurse to total or subtotal hysterectomy may be indicated.
A postoperative hematoma is likely to form because of a potentially big and complex surgical incision site, difficulty in achieving watertight closure of the dead spaces left after fibroid enucleation. Injury to neighboring structures such as the fallopian tube, urinary bladder, small bowel, or ureters is conceivable. These were discussed with the patient.
It is important to discuss the likelihood of postoperative adhesion and paralytic ileus after surgery. The possibility of uterine rupture and the requirement for elective cesarean section were considered as postoperative reproductive outcomes. 16 A possible recurrence of the uterine fibroids was discussed as she is black, her uterine fibroids were multiple, and she is between 30 and 40 years of age; well known risk factors for recurrence. 23
This patient was adequately informed, educated and counseled about the diagnosis, treatment plan, possible intraoperative complications, surgical risks, and the benefits of her surgery. An informed written consent was obtained, and feedback addressed.
The surgical removal of giant leiomyomas has considerable morbidity and technical challenges. As a result, meticulous perioperative care is critical to achieving the greatest surgical outcome. Preoperative patient optimization is crucial, and meticulous examination of cardiorespiratory and renal function is advised. The patient should be assessed and treated for deep vein thrombosis (DVT). Our patient had compression stockings intra- and post-operatively and she had Enoxaparin sodium injections post operatively for DVT prophylaxis.
Any preoperative anemia should be treated considering the expected significant blood loss. 2 Patient had significant blood loss intraoperatively with a post operative packed cell volume of 20%. She was subsequently transfused with 2 units of blood preoperatively and an additional 3 units of blood post operatively and was later discharged with a PCV of 27%. Furthermore, she had potassium correction for hypokalemia of 2.5 mmol/l which was optimized to a potassium level of 3.5 mmol/l.
Patient had foley’s catheter application at the level of uterine isthmus to reduce significant blood loss. Due to differing perspectives and a paucity of documentation, the tourniquet technique is noteworthy but not always used. For example, it has been suggested that blocking the infundibulopelvic arteries could impair fertility, cause ischemia damage to the ovaries, impede ovarian circulation, and decrease ovarian reserve. 24 Incognito and group in their reported clinical experience has demonstrated significantly lowering blood loss following the Foley catheter use as a tourniquet during a laparotomic myomectomy. Their study advised routine use of Foley catheter tourniquet as it has the potential to greatly reduce the need for blood transfusions and improve patient outcomes and safety. 25
The patient should be positioned intraoperatively at 15° tilt away from the midline to allow proper breathing while attempting to reduce compression of the inferior vena cava. A multidisciplinary surgical strategy involving an experienced Gynecologists, Gynecology oncologists, General surgeons, Urologists, or Plastic surgeons is required for the surgery. Meticulous dissection and careful hemostasis are advised. 2 Our patient was operated on by 2 senior gynecologists, with invites made to both general surgeons and urologists in case their services were required. The patient’s hemodynamic condition must be closely monitored both intraoperatively and postoperatively, with quick replenishment of fluid losses. Our patient was transferred to a high dependency unit following surgery for close monitoring.
Let’s conclude by admitting that surgical removal of giant fibroids is technically difficult, challenging 10 and entails a high risk of morbidity and mortality due to the dangers of massive hemorrhage and possible postoperative complications. Our patient had an open myomectomy as against hysterectomy despite the giant nature of her uterine fibroids because she was nulligravid and most of the myoma nodules were not necessarily submucous. The approach of patients with giant uterine fibroids is usually surgical. However, the type of surgery will be influenced by the size and number. 9 Ikechebelu and group managed a 38-year-old lady with 11.6 kg uterine fibroids via open myomectomy. The uterine mass was close to the size of the index patient. 9 Surprisingly, Ezugwu et al had an open myomectomy on an even larger uterine fibroid mass of 16.8 kg. The lady who was followed up and eventually became pregnant. 10 Our case demonstrates the need for careful case selection, good preoperative assessment, optimization, and the importance of handling such case management by an experienced Gynecologist with a good multidisciplinary back up.
Conclusion
It is possible to offer open myomectomy in patients with giant uterine fibroid following careful patient selection with a consent for possible hysterectomy. Dissimulation can be minimized with repetitive counseling of patients. The choice of surgery depends on the size and number of uterine fibroids, but surgical approach does not necessarily influence fecundity. Some spiritual homes believe that uterine fibroids follow ancestral curses, and such patients must be exorcized. Such misconception must be discussed with community leaders, pastors, Imams, lord’s spiritual, and market leaders. These leaders must be educated on the pathophysiology of uterine fibroids to reduce dissimilation among patients. Awareness about different treatment options must serve as content of these discussions.
Footnotes
Acknowledgements
We thank all hospital staff who contributed to the management of the index patient. Special thanks to Dr Asukwo Edet, the chief anesthetist who anesthetized the index patient. Authors would like to appreciate especially the contribution and voluntary agreement of the patient.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
EI conceptualized, designed, wrote the original draft, the review & editing of the manuscript and was involved in the software, formal analysis, investigation, methodology, resources, data curation, project administration, supervision, validation, visualization and approved the version to be published. ICI, ROM, AOA, and CCA participated in the visualization, formal analysis, supervision, and project administration, critical revision of the manuscript while GUE, ABU, JII, and UUO were involved in writing the review & editing, formal analysis, investigation, resources, visualization, and supervision for important intellectual content and approved the version to be published. All authors approved the version to be published and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Data Availability Statement
Not applicable.
