Abstract
Cancer-associated microangiopathic hemolytic anemia (CA-MAHA) is a rare paraneoplastic syndrome. The most effective approach to treating CA-MAHA is to address the underlying malignancy. Documented cases of CA-MAHA are limited to fewer than 50 patients in the literature. Herein, we present a 51-year-old female patient who developed CA-MAHA as a complication of gastric adenocarcinoma. Despite receiving neoadjuvant and adjuvant chemotherapy for gastric cancer, the patient experienced disease progression with metastatic lesions in the liver, pancreas, and other sites. This report highlights the challenges in diagnosing and distinguishing CA-MAHA from other similar conditions such as disseminated intravascular coagulation (DIC), hemolytic uremic syndrome (HUS), thrombotic thrombocytopenic purpura (TTP), and rheumatological paraneoplastic syndromes. Additionally, it concludes that CA-MAHA is associated with a poor prognosis and limited clinical benefit from treatment, emphasizing the need for early diagnosis and effective management strategies.
Introduction
Gastric cancer ranks as the fifth most common cancer 1 and the third leading cause of cancer-related deaths globally. 2 It is characterized by symptoms such as unexplained weight loss, abdominal pain, nausea, vomiting, and loss of appetite. 3
However, metastatic gastric cancer can also manifest through paraneoplastic syndromes, which are rare conditions caused by the presence of a hidden malignancy. These syndromes can affect multiple organ systems, including the neurological, dermatological, gastrointestinal, endocrine, hematologic, and cardiovascular systems. 4
Among them, cancer-associated microangiopathic hemolytic anemia (CA-MAHA) is considered a rare paraneoplastic syndrome that can occur as a result of malignancy; it is usually indicative of a poor prognosis. To date, there have been fewer than 50 documented cases of gastric cancer accompanied by CA-MAHA. 5
In the case of CA-MAHA, the most effective therapeutic approach is to treat the underlying malignancy. 6 In our case, we reported a 51-year-old female diagnosed with MAHA secondary to metastatic gastric adenocarcinoma. This case highlights the importance of early diagnosis to avoid potentially ineffective treatments and to exclude other similar differential diagnoses.
Case Presentation
A 51-year-old female patient with no significant past medical or surgical history, a non-smoker, and a strong family history of cancer, was in her usual state of health until 2 years ago when she was incidentally discovered to have gastric adenocarcinoma through laboratory tests and imaging while preparing for gastric sleeve surgery.
She received 4 cycles of neoadjuvant chemotherapy with the FLOT protocol (5-Fluorouracil 2600 mg/m2, leucovorin 200 mg/m2, oxaliplatin 85 mg/m2, and docetaxel 50 mg/m2 intravenously every 14 days). Following this, she underwent a gastrectomy, followed by an additional 4 cycles of adjuvant chemotherapy using the same protocol. The patient was then kept on regular follow-up.
One year later, she began experiencing lower back pain, which was initially managed with painkillers. However, her pain gradually worsened, prompting a CT scan. The scan revealed metastatic liver lesions, an infiltrative pancreatic mass, a left adrenal gland mass, and multiple enlarged lymph nodes (Figure 1).
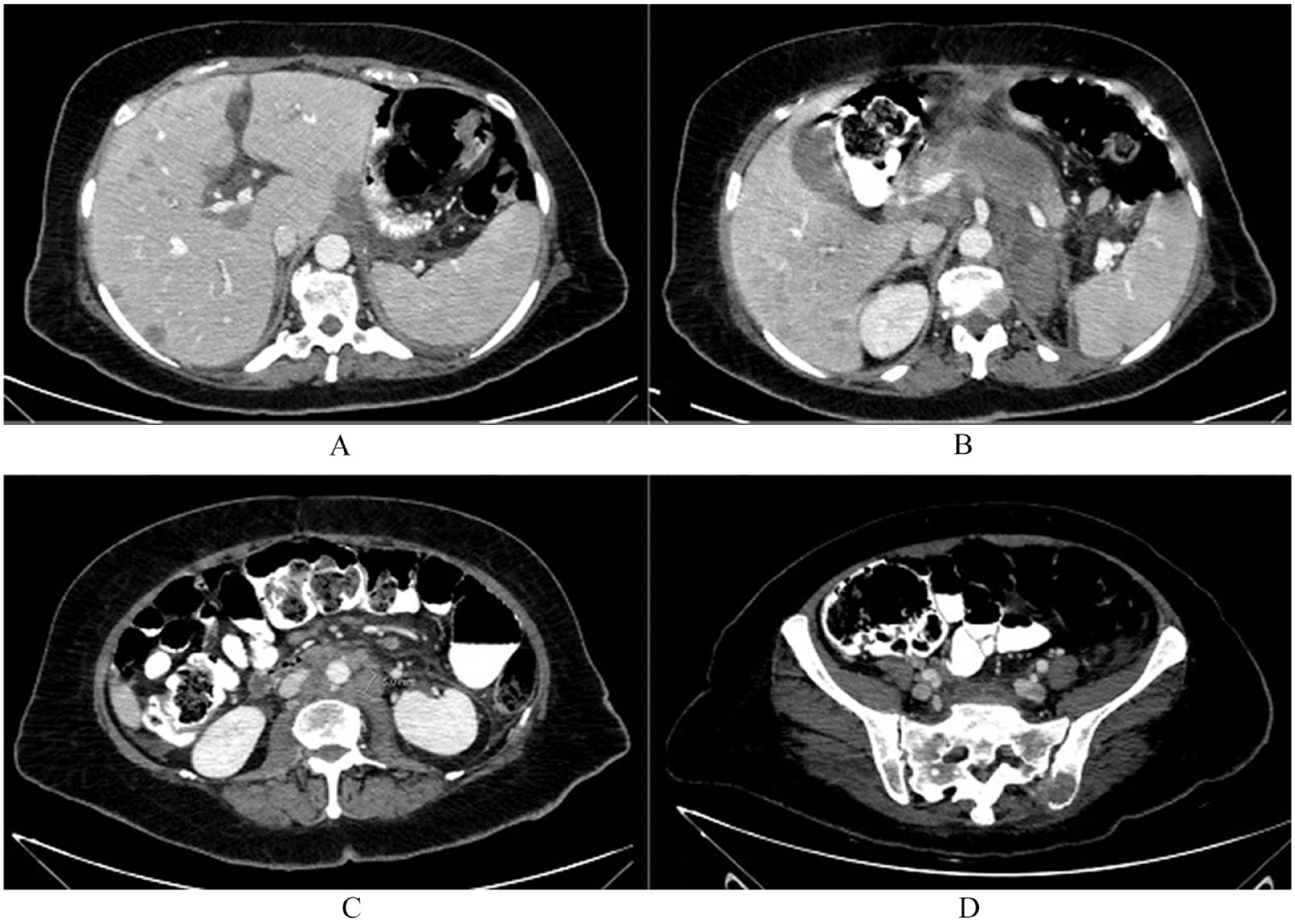
Multi-level selected axial images of the patient’s abdomen CT with IV and oral contrast showing widespread metastatic disease with multiple hypo-attenuating metastatic liver lesions are noted (A) In addition to left adrenal and pancreatic lesions (B). Multiple enlarged pathological para-aortic lymph nodes are noted (C) with a left iliac bone lytic lesion showing soft tissue component and cortical bone erosions (D).
The patient was referred for advanced oncology care. On admission, she appeared ill, with normal vital signs. She had an Eastern Cooperative Oncology Group-Performance Status (ECOG-PS) of 2. Her cardiopulmonary exam was unremarkable, and she exhibited clean, well-healed surgical scars on her abdomen. Her abdomen was soft to palpation with a palpable liver. She complained of lower back pain, with no radiation to the lower limbs. CBC and basic metabolic panel revealed no abnormalities.
After receiving 20 Grays of radiotherapy to the fourth lumbar vertebrae (5 Grays in 4 fractions) for pain control and palliative radiotherapy, the patient switched to the care of medical oncology for a new line of chemotherapy. Approximately a month later, significant findings were observed in her laboratory results as illustrated in Table 1. Both direct and indirect Coombs’s tests were negative, while the peripheral smear showed red blood cell fragments (schistocytes) scattered among normal red blood cells. Her PLASMIC score was calculated as a 6-point score (one point for her platelet count, one point for undetectable haptoglobin, one point for no history of stem cell or solid organ transplant, one point for MCV <90, one point for INR level, and one point for serum creatinine), which is considered high. 7
The laboratory results of complete blood count, basic metabolic panel, urinalysis, and coagulation studies of the patient on admission.

The hematology team was consulted, confirming that the patient’s condition involves MAHA and thrombocytopenia as a paraneoplastic manifestation associated with gastric cancer. They emphasized the importance of treating the underlying cause, gastric cancer.
The patient received 2 units of packed red blood cells and 6 units of platelets to address her anemia and thrombocytopenia, respectively.
A few days later, the patient exhibited decreased consciousness and profuse sweating without chest pain, nausea, vomiting, shortness of breath, or accompanying fever. Following consultation with the hematology team, the patient was urgently referred for plasmapheresis and chemotherapy because MAHA had been identified as a paraneoplastic syndrome expected to improve with chemotherapy. Unfortunately, the patient had a cardiopulmonary arrest while en route to the hospital and died before receiving chemotherapy or plasmapheresis.
Discussion
CA-MAHA is a type of Coombs-negative thrombotic microangiopathy commonly observed in solid tumors. It is characterized by red blood cell fragmentation, typically evidenced by schistocytes on peripheral blood smear examination. Laboratory findings often include elevated lactate dehydrogenase (LDH) levels, decreased or absent haptoglobin, low platelet count, and increased total bilirubin levels. These diagnostic indicators collectively contribute to identifying CA-MAHA in patients with underlying malignancies.8,9
According to theories reported in the literature, the pathogenesis of CA-MAHA suggests that tumor emboli cause microvascular obstruction, leading to red blood cell fragmentation and platelet consumption.6,10
Nevertheless, several cases in the literature demonstrate potential causes of CA-MAHA, such as fibrinoid necrosis in the bone marrow.11,12 Other studies suggest that MAHA can occur due to tumor-derived factors, procoagulants, and specific anti-cancer drugs (chemotherapy-induced MAHA). However, the precise pathogenesis remains unclear. 1
In evaluating overlapping clinical manifestations with diseases like thrombotic thrombocytopenic purpura (TTP), disseminated intravascular coagulation (DIC), and hemolytic uremic syndrome (HUS), the absence of neurological involvement does not align with a TTP diagnosis. The patient’s normal serum creatinine levels make HUS unlikely, while normal fibrinogen levels and the lack of PT and PTT prolongation rule out DIC. Additionally, respiratory symptoms and bone pain are more frequently associated with CA-MAHA compared to cases without MAHA as a paraneoplastic feature, as reported in the literature.9,13 This is consistent with our patient, who experienced bone pain that did not respond to painkillers.
According to the literature, other rheumatological paraneoplastic syndromes can be considered in the differential diagnosis of CA-MAHA. Systemic lupus erythematosus (SLE) can lead to a variety of hematological abnormalities, including autoimmune hemolytic anemia (AIHA). AIHA can mimic MAHA due to the presence of anemia, schistocytes on the peripheral blood smear, and thrombocytopenia.14-16 However, in our case, the patient did not present with malar rash, oral ulcers, arthritis, or renal involvement. Another important factor is that the Coombs test was negative; in SLE-induced AIHA, it would be positive.
These differentials highlight the complexity of diagnosing anemia in the context of metastatic gastric cancer and the importance of careful evaluation to distinguish between primary hematologic disorders and paraneoplastic syndromes.
Along with other published cases, patients with metastatic gastric cancer to the bone complicated by MAHA generally have a poor prognosis and often die shortly after being diagnosed with CA-MAHA. Therefore, a bone marrow biopsy is an important diagnostic tool to investigate the cancer.17,18 This is consistent with our study, in which the patient died nearly 1 year after being diagnosed with CA-MAHA which had metastasized to the bone.
Robert Lam et al and his colleagues, in their systematic review and case-control study on CA-MAHA, found that despite treatment, the overall prognosis remains poor. They observed higher mortality rates and worse survival compared to patients with metastatic gastric cancer without MAHA. 19 Consequently, it is important to investigate CA-MAHA in cases where metastatic gastric cancer worsens and the patient does not respond to treatment as expected.
There is no definitive treatment for CA-MAHA. Steroids, Rituximab, immunoglobulin, and plasmapheresis have shown effectiveness in typical cases. Platelet and red blood cell transfusions are also necessary. However, chemotherapy and effective tumor control are potentially the most impactful treatments for CA-MAHA.17,18 However, the patient experienced a cardiopulmonary arrest while being transported to the hospital and died before receiving chemotherapy or plasmapheresis.
Conclusion
In conclusion, this case emphasizes the critical need to recognize and address CA-MAHA in advanced gastric cancer. Despite challenges in differential diagnosis, early detection is vital. Prompt intervention, including chemotherapy and supportive care, is essential for improving outcomes. However, CA-MAHA presents a poor prognosis, with limited clinical benefits from cancer-directed and MAHA-specific treatments. Further research is necessary to optimize management strategies and understand its impact on survival in metastatic gastric cancer.
Footnotes
Acknowledgements
The authors thank Polytechnic Medical Students’ Research Association (PMRA) for their invaluable input and support throughout the research process.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data used to support the findings of this study are included in the article.
Patient Consent Statement
Written informed consent was obtained from the patient for the publication of this case report and accompanying images.
