Abstract
A chordoma is a slow growing, locally invasive, low-grade tumor belonging to the sarcoma family. It mainly affects the sacrum and skull base. We present a case of thoracic chordoma initially presented with epidural hematoma (EDH), which is a rare clinical entity. We reported this case, and also performed a PRISMA-driven systematic review to summary the similar cases in the literature. This review includes the clinical characteristics and outcome of thoracic chordoma. Our case involves a 60-year-old male who, despite no history of trauma, presented with acute paraparesis. An epidural hematoma was identified at T6 level, leading to a surgical intervention involving T4-6 laminectomy and fixation. Six months subsequent to surgery, the patient experienced progressive lower limb weakness and spasticity. Computed tomography (CT) exhibited erosion of T6 and an associated aggressive mass. Magnetic resonance imaging (MRI) revealed a large heterogenous soft tissue mass arising from the vertebral body and right pedicle of D6, protruding in the epidural space and compressing the spinal cord focally at this level. The mass measured approximately 5 × 4 × 3.5 cm. Magnetic resonance myelography indicated a filling defect at T5–6 level, confirming the intraspinal location of the soft tissue lesion. Complete excision of the mass confirmed the diagnosis of thoracic chordoma. Postoperative follow-up demonstrated notable improvement in the lower limb spasticity and paraparesis, and the patient started adjuvant radiotherapy. This case underscores the importance of maintaining a high index of suspicion when evaluating presentations resembling EDH.
Introduction
Chordoma is a rare malignant tumor arising from embryonic notochord remnants. 1 It occurs mainly in the sacrum, base of the skull, particularly the clival region.2-4 Thoracic chordoma accounts for only 1% of all chordomas, it is characterized by slow growth, local invasion, and a high recurrence rate. 5 The clinical presentation of thoracic chordoma may include back pain, spinal deformity, neurological deficits, and respiratory symptoms. 3 The diagnosis of thoracic chordoma is based on radiological and histopathological findings and molecular markers. 5 The optimal treatment for thoracic chordoma is complete surgical resection with negative margins, which can improve survival and quality of life. 3 However, the surgical approach is challenging due to the complex anatomy and proximity to vital structures. 3 Adjuvant therapies such as radiation and chemotherapy have limited effectiveness and are mainly used for palliation or prevention of recurrence. Therefore, new therapeutic strategies based on targeted therapy and immunotherapy are being explored to improve the outcomes of patients with thoracic chordoma. 5 Here, we report a rare case of a thoracic chordoma, which was initially manifested with epidural hematoma (EDH) causing acute motor deficit. Moreover, we conducted a PRISMA-driven systematic review previous studies reporting thoracic chordomas.
Case Presentation
A 60-year-old man who was previously healthy presented with acute paraparesis. He reported no history of trauma or fever. Clinical examination showed bilateral lower limb weakness (2/5 Medical Research Council grade) without muscle atrophy and preserved deep tendon reflexes. Urgent CT and MRI showed an abnormal epidural density/intensity at the level of D6 (Figure 1A and B), requiring urgent surgical intervention to alleviate spinal cord compression. Intraoperatively, an epidural hematoma was recognized and evacuated. Short segment fusion at the levels of T5−T7 was performed (Figure 1C). Postoperatively, muscle power was restored, and the pain improved. Two months post-surgery, the fixation system was removed due to failure, likely due to technical factors. The patient was advised to wear a dorsolumbar support.

(A) Pre-operative selected axial CT image at the level of T6 “bone window” demonstrating a subtle poorly-defined abnormal density within the vertebral body, with multiple foci of intra-lesional calcification. (B) Pre-operative sagittal MRI spine, T2 fat-saturated image demonstrating high T2 signal abnormality within the vertebral body of T6. No vertebral body compression, collapse or destruction seen. There is another abnormality of focal intraspinal high T2 signal abnormality at the same level “the arrow,” which represents the EDH. Unfortunately, the T1-sequence “not shown here” was of low imaging quality due to patient-related motion artifact. The hematoma was confirmed intra-operatively. At this point, the Radiology Neurosurgery team interpreted the vertebral body lesion as a hemangioma, with a small extradural hematoma at the same level. Due to the acute presentation of the patient, the decision was made to take the patient to the OR to evacuate the extradural hematoma. (C) A postoperative sagittal CT image “bone window” showing a short-segment vertebral fixation at the levels of T5–T7.
Six months post-surgery, the patient showed bilateral lower limb weakness (3/5 Medical Research Council grade). Spinal MRI showed a heterogenous 5 × 4 × 3.5 cm soft tissue mass from the vertebral body and right pedicle of T6, protruding in the epidural space and compressing the spinal cord. Magnetic resonance myelography demonstrated significant compression of the cord (Figure 2).

(A) A sagittal T2W MRI spine of the same patient 6 months postoperative demonstrating significant progression of the signal abnormality in the vertebral body of T6. This is associated with a large heterogeneously high T2 soft tissue component bulging into the epidural spinal canal with extensive compression of the spinal cord and right neural foramen at the same level. There is also minimal caudal migration to the level of mid D7. (B) axial T2W MRI & (C) an axial CT at the T6 level showing the previously described signal abnormality, extending to the right pedicle and lamina with corresponding bony erosion well-demonstrated in the provided CT image. The MRI shows significant compression of the spinal cord, which is compressed and displaced to the left side, but with no intramedullary signal abnormality. (D) A coronal image of MR myelography showing a significant filling defect at the level of T6 and T7 confirming the intraspinal extension of the lesion and significant cord compression. (E) A postoperative sagittal CT image of the dorsal spine demonstrating interval longer fixation of the vertebrae at the levels of T4, T5 and T7, T8.
Under general anesthesia, decompression and mass excision were performed, followed by long-segment fixation via a posterior approach. The fixation consisted of segmental rod-screw constructs 2 levels above (T4, T5) and 2 levels below (T7, T8) (Figure 2E). Histopathological examination confirmed chordoma diagnosis (Figure 3). The patient was referred for radiotherapy and scheduled for regular follow-up. Complete resolution of his symptoms was reported, with no neurological deficits.
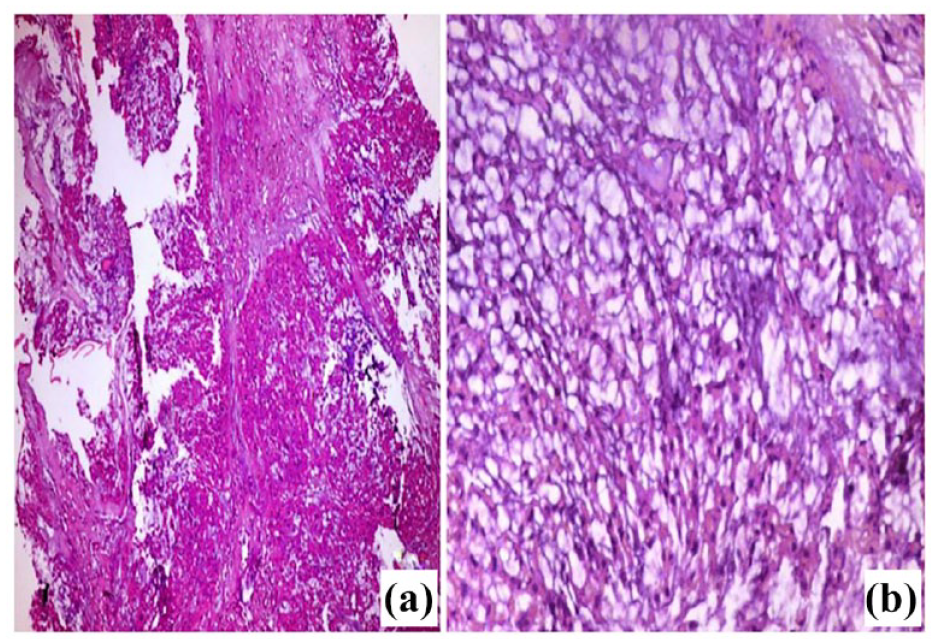
Light microscopic picture of chordoma (A) Tumor with multilobulated growth pattern of epithelioid cells. The lobules showed anastomosing nests or cords of acidophilic tumor cells. (B) Higher magnification of the tumor showing cords of acidophilic, vacuolated small and medium-sized cells with rounded nuclei, most of which had low-grade atypia. The tumor cells arranged in a basophilic fibromyxoid stroma, with no evidence of necrosis. H&E (A) 100x, (B) 400x.
Methods
PRISMA-driven systematic review for thoracic chordoma
Search strategy of systemic reviews
We conducted an extensive literature review was performed following the guidelines published for PRISMA. 6 An expansive computerized systemic review of published randomized controlled trials, cohort studies, and case reports, was performed by searching the following 3 databases: Scopus, PubMed, Science Direct. The key search terms included “chordoma,” “thoracic,” “spinal cord” and “tumor.” The search was limited to human studies and full text, written in English, published in English until May 22, 2022.
Screening and data extraction
The above-mentioned search strategy was completed by May 2022. Two independent investigators reviewed the relevance of all titles and abstracts identified from the different databases. The opinion of a third investigator was considered in articles with discrepancies. Full articles were further assessed when the abstracts met the inclusion criteria. The reviewed data were obtained and entered onto each reviewer’s ad hoc standardized data entry form. We compared the inclusion data for the year of publication, the number of patients studied, the sex and age of every patient, the initial presenting symptoms, the vertebral level of chordoma, the surgical approach deployed, any adjuvant radio or chemotherapy, the outcome of the patient, the follow-up period and the incidence of recurrence.
Assessment of methodological quality
The methodological quality of the included studies was assessed using the Newcastle–Ottawa Quality Assessment Scale, which scores from 1 (poor) to 9 (excellent). 7 Due to the lack of standardized descriptions for intermediate scores on this scale, we categorized studies with a total score of 7 or higher as high-quality.
Results
Our search yielded 160 citations. After reviewing the title and abstract and the entire text, 113 were discarded as they did not meet the proposed criteria. In total, 46 papers were identified for review, as illustrated in the PRISMA flow diagram (Figure 4). The clinical characteristics of the included articles and the current case report are summarized in Table 1 where 54 patients including the current case were described. The age ranged between 8 and 89, with a mean of 46.8 (20.5) years. Male patients slightly outnumbered female patients (=28; 51.9%, vs 26; 46.3%), and the sex of 1 patient was not mentioned. The most frequently reported tumor level was T3 (n = 18; 33.3%), followed by T2 (n = 15; 27.8%) and T4 (n = 14;25.9%). T8 and T9 were the least frequently reported levels in 2 patients (3.7% for each). Associated cervical involvement was reported in 3 patients, and in 1 patient, the sacral segments were involved. In 9 patients, the tumor was discovered incidentally on chest radiography. Sensory affection was the predominant complaint. Surgical resection was conducted in most studied patients (n = 50; 92.6%). However, in 1 patient, the tumor was too extensive for surgical treatment. Another patient was still under evaluation for surgery, and we could not identify if other 2 patients underwent surgical procedures.

PRISMA flow diagram for systematic review of thoracic chordoma.
The characteristics of included studies.

Abbreviations: LFU, lost to follow-up; NM, not mentioned; SOB, shortness of breathing.
Six patients underwent chemotherapy (11.1%), 35 patients did not (64.8%), and in 13 patients, the use of chemotherapy was not mentioned. Moreover, 21 patients underwent radiotherapy (38.9%), 22 patients (40.7%) did not, and in 11 patients, radiotherapy use was not mentioned. Complete cure with no residual symptoms or recurrence was reported in 30 patients (55.46%), and the identified follow-up period ranged between 2 days and 16 years. The majority of the studied patients (n = 34; 63.0%) were described as having favorable outcomes, while 11 patients (20.4%) were considered to have unfavorable outcomes. Recurrence was reported in 6 patients (10.9%), and residual regrowth was reported in another patient. Seven patients died, representing 13.0% of the studied patients; 421 patients survived (75.9%), while we could not identify the fate of 6 patients.
Discussion
A chordoma is uncommon malignant tumor of the axial skeleton that originate from embryonic remnants of the primitive notochord (earliest fetal axial skeleton, extending from the Rathke’s pouch to the tip of the coccyx): 50% in the sacral region, 35% in the skull base, and 15% in the vertebral bodies, lumbar spine, the thoracic spine.52,53 The exact etiology of chordoma is related to a genetic malformation of the notochord, which is the origin of the nucleus pulposus in humans. These genes are still being tested, but PTEN deficiency (mTOR) and the Brachyury gene are considered the most important. 54 Chordomas account for 20% of primary spinal tumors and 3% of all bone tumors and affect men and women in a ratio of 2:1. Although it occurs more frequently in the 40- to 60-year-old age group.52,53 The . Faheem et al. discussed a case of an 8-year-old boy with intramedullary chordoma without bone involvement. 21 Early favorable prognosis depends on how early we can detect the tumor and start its management. 55
Usually, the chordoma has a direct effect through nerve root compression, leading to chronic back pain or urinary and bowel disorders. 56 In this case, we discussed a 60-year-old male patient who presented with acute paraparesis due to cord compression by an epidural hematoma. Six months later, there was an increasing paraparesis due to the soft tissue component, which was later demonstrated as thoracic chordoma by histopathology examination. This paraparesis is not commonly associated with chordomas. However, suppose a chordoma tumor grows large enough or extends into the spinal canal. In that case, it can compress or impinge on the spinal cord or nerve roots, leading to neurological symptoms such as weakness, numbness, or paralysis in the legs. The severity of paraparesis or other neurological deficits associated with a chordoma can vary depending on the size and location of the tumor. 57
It was mentioned that the most common presenting complaint was chest or back pain, followed by anterior mediastinal compression syndrome, hoarseness, dysphagia, and cough. 5 One-third of reported patients were asymptomatic and detected during routine chest examinations 20 Many preliminary reports described several presentations for thoracic chordoma10,12,15 Among these reports, chordomas were initially misdiagnosed as different neoplasia, including hemangioma, benign neurinomas, 23 and adenocarcinomas. 13 Others reported chordomas metastasizing to lymph nodes, lungs, 18 bones, brain, and viscera. Nonetheless, this type of chordoma is more aggressive. 52
Though few authors proposed primary mechanisms for the association of hemorrhage with chordomas, the exact mechanism is not fully understood. Chordoma can cause dural vessel proliferation if they are in contact with each other. 58 It was hypothesized that rapid tumor growth may cause small friable blood vessels to rupture. Also, subsequent occlusion of small vessels can cause necrosis and hemorrhage in the tumor. 59 We assume a possibility for a chordoma to directly affect nearby blood vessels or disrupt the normal vascular supply to the spine, which could potentially lead to bleeding or vascular complications, but this would be a rare and secondary effect. All these causes may explain the presence of an initial epidural hematoma even before the diagnosis of chordoma in our case. In the present case, the hematoma demonstrated hyperintensity on T2-weighted sequences, indicating a subacute nature and reflecting the paramagnetic properties of hemoglobin breakdown products. The noticed high T2 signal within the vertebral body of T6 and the corresponding abnormal density on CT were thought to be related to a vertebral hemangioma, which is the most common primary tumor of the spine. 60 In response to the emergent presentation of the patient with acute paraparesis, a prompt decision was taken to proceed with surgical intervention. The objective of the surgery was the expeditious decompression of the spinal cord through hematoma evacuation. Intraoperatively, no masses were seen at that time.
Six months after surgery, the patient showed recurrence of the lower limb motor deficit. An urgent CT was requested, which demonstrated a lytic lesion within the vertebral body, right pedicle and lamina of T6 associated with a large soft tissue lesion expanding the spinal canal. The MRI scan confirmed the epidural extent of the lesion, and MR myelography demonstrated the significant compression of the cord which helped for presurgical evaluation. At this point, the preliminary diagnosis of vertebral hemangioma was refuted due to presence of the aggressive features and soft tissue component. However, these features did not confirm chordomas, as some chordomas may mimic other neoplasia. 13 So, a biopsy should be examined under the microscope to ensure the specific pattern of physaliferous bubbly cells within the myxoid stroma, which indicates a chordoma lesion. A tissue sample should also be examined for a high Brachyury gene value manifesting in high concentrations. 61
The first line of treatment for chordomas is surgical resection. Stacchiotti et al, 62 reported multiple variations in resection margins according to the chordoma consensus group approach, such as wide resection, marginal resection, and intralesional resection. Also, the posterior approach for spinal decompression was recommended in giant chordomas with neurological deficits. 19 The present case underwent short and long-segment fixation. The short segment fixation was conducted prior to the diagnosis of chordoma. Following an unsuccessful outcome, the fixation device was removed. Nevertheless, the long-segment fixation was successful, and the patient improved. McLain et al, acknowledged some multifactorial causes of short-segment instrumentation failure in thoracolumbar spines, including the limited ability to maintain sagittal correction in the face of axial instability and the undue bending moments generated at the screw hub. Once initial bending failure happens, additional collapse is likely, developing loss of lordosis, predisposing to a higher incidence of clinical failure and associated pain. 63 On the other hand, literature supports the use of long constructs in thoracic and thoracolumbar segments for greater force reduction and better stabilization. 64 The thoracic spine is comparatively immobile and extending the construct into these segments has little mechanical cost, while offering more extensive fixation. Contemporary instrumentation systems are versatile, allowing fixation with pedicle screws to permit the surgeon to directly instrument vertebrae and provide 3-column fixation in unstable injuries and to minimize the length of fusion. 65
Radiotherapy plays a vital role in the management of the tumor, even in disease control or after surgery to prevent a recurrence. A clinical trial conducted on 50 patients who underwent spinal chordoma resection to examine the effect of proton radiotherapy on recurrence rate and local control showed that a recurrence rate was less common for primary tumors (11%), and the local control was high. 66 Therefore, the ideal treatment protocol is resection of the tumor followed by proton irradiation.45,67 A 5-year survival after this protocol was estimated to be 50%–60%, and the median overall survival for the tumor was about 7 years. However, the recurrence rate, tumor metastasis, and tumor extent mainly determine the overall survival rate. 68 Conventional chemotherapy plays no role in the management of chordomas; however, a small study suggested that imatinib mesylate may have antitumor activity in patients with chordomas. 69 A later study showed that the combination of imatinib mesylate and metronidazole cyclophosphamide had a synergistic anti-angiogenic effect on pericytes and endothelial cells. 70 Royo Crespo et al 25 reported good response of thoracic chordoma to chemotherapy in an elderly male. Contradicting these studies, Pai, 37 reported unfavorable outcomes and long-term disability and neurological deficits in the form of urinary incontinence in 49-year inoperable male patient diagnosed with thoracic chordoma.
In advanced cases where surgery is not feasible, radiofrequency ablation may be considered as a palliative alternative. 71 In the current study, several key factors influenced the decision to initiate adjuvant therapy. Histopathological examination confirmed the diagnosis of chordoma and showed low-grade atypia, suggesting a risk of aggressive tumor behavior. Following meticulous intraoperative tumor resection and spinal cord decompression, the surgical team aimed to achieve complete tumor removal with negative surgical margins, which is associated with favorable long-term outcomes in chordoma cases. 72
Patient Perspective
It was a startling revelation for me to discover that I had a tumor in my spine and to undergo 2 surgeries within 1 year. Following the surgery, I can now carry out my daily tasks. I was worried when the doctor indicated that I might not fully regain my muscle power, but fortunately enough, I did.
Conclusion
Chordomas of the thoracic spine are rare neoplasms that exhibit diverse clinical presentations. Most cases are asymptomatic and discovered incidentally. Early management is associated with a better prognosis. So, early warning signs, including EDH should be considered critically. An EDH should not be exclusively attributed to trauma unless tumors, including chordoma are ruled out. We reported an atypical initial presentation of chordoma with EDH that was successfully treated by decompression and fixation.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to the study conceptualization, Methodology, Formal analysis, Writing, Reviewing and Editing the manuscript.
Ethical Consideration
The current case report was implemented following the CARE (CAse REport) guidelines, 73 following the declaration of Helsinki and its later amendment, 1964. An approval from the institutional review board of the Faculty of Medicine Hospitals, Zagazig University, was obtained before commencing this work.
Informed Consent
Informed consent was obtained from the patient, including his consent to publish radiological and histopathological images. However, we concealed the patient’s identity, and no data revealing his identity was published.
