Abstract
Excessive water consumption is an extremely rare and potential asthma risk factor with very few cases reported in the literature. Common triggers of asthma include genetic factors, smoking, allergens, and viral respiratory infections. The adult patient with asthma reportedly drank too much water and was unable to get relief from his asthma while hospitalized. The patient’s asthma was better controlled with the use of diuretics and control of the patient’s fluid intake and output. This case explores asthma induced by excessive drinking of water.
Introduction
Asthma is one of the most common chronic respiratory diseases, affecting more than 300 million people worldwide. 1 The prevalence of asthma is the highest in Australia, followed by Sweden, and is higher in both high-income and low-income countries than in middle-income countries. 2 Asthma not only affects people’s health but also brings a large economic burden. 3 Genetic factors, 4 air pollution, 5 smoking, 6 psychological stress, 7 allergens, 8 and respiratory viral infections 9 were risk factors for inducing and exacerbating asthma. The study that excessive fluid administration can cause acute asthma exacerbation in children has been reported, 10 but excessive drinking water inducing asthma exacerbations in adults is uncommon in clinical practice. We mainly introduced an adult patient who experienced an acute asthma exacerbation induced by excessive drinking water during hospitalization and elucidated the relationship between excessive drinking water and asthma exacerbation.
Presentation of the Case
A 72-year-old Han ethnic male patient from China with increased chest tightness, wheezing, and shortness of breath was admitted to the hospital on March 10, 2023. The patient developed bronchial asthma 2 years ago without an obvious trigger. After treatment with inhaled budesonide formoterol inhalation powder nebulizer (II) (9 µg twice daily) and oral aminophylline tablets (0.1 g twice daily), his bronchial asthma was effectively controlled. In addition, the patient had a history of bronchitis and a chronic obstructive pulmonary disease for 2 years, hypertension for more than 21 years, chronic heart failure for more than 6 months, lacunar cerebral infarction for more than 3 years, coronary atherosclerotic heart disease for 8 years, and chronic hepatitis B for more than 20 years, for which he was prescribed standard myrtle oil enteric capsules (adult pack) (0.45 g orally twice daily), sacubitril sodium valsartan tablets (100 mg orally once daily), isosorbide mononitrate tablets (20 mg orally twice daily), and entecavir tablets (0.5 g orally once daily). The patient was admitted with an absolute lymphocyte value of 0.93 × 109/l (normal range: 1.10-3.20), an absolute neutrophil value of 2.70 × 109/l (normal range: 1.80-6.30), ultrasensitive C-reactive protein of 0.80 mg/l (normal range: 0.00-10.00), type B brain natriuretic peptide precursor (NT-pro BNP) <10.00 pg/ml (normal range: 0-125), and left ventricular ejection fraction of 67%. Chest CT showed (Figure 1) chronic bronchitis with emphysema and alveolar manifestations and small nodules in the upper lobe of the right lung. The patient was diagnosed with chest tightness, wheezing, and shortness of breath caused by bronchial asthma attacks. On top of the original treatments, the patient was treated with low-flow mask oxygenation (3 L/min) from 10 to 14 March, sodium methylprednisolone succinate for injection (40 mg iv drip once daily) from 11 to 14 March, and hydroxypropyl theophylline injection (4 ml iv drip once daily) from 12 to 14 March.
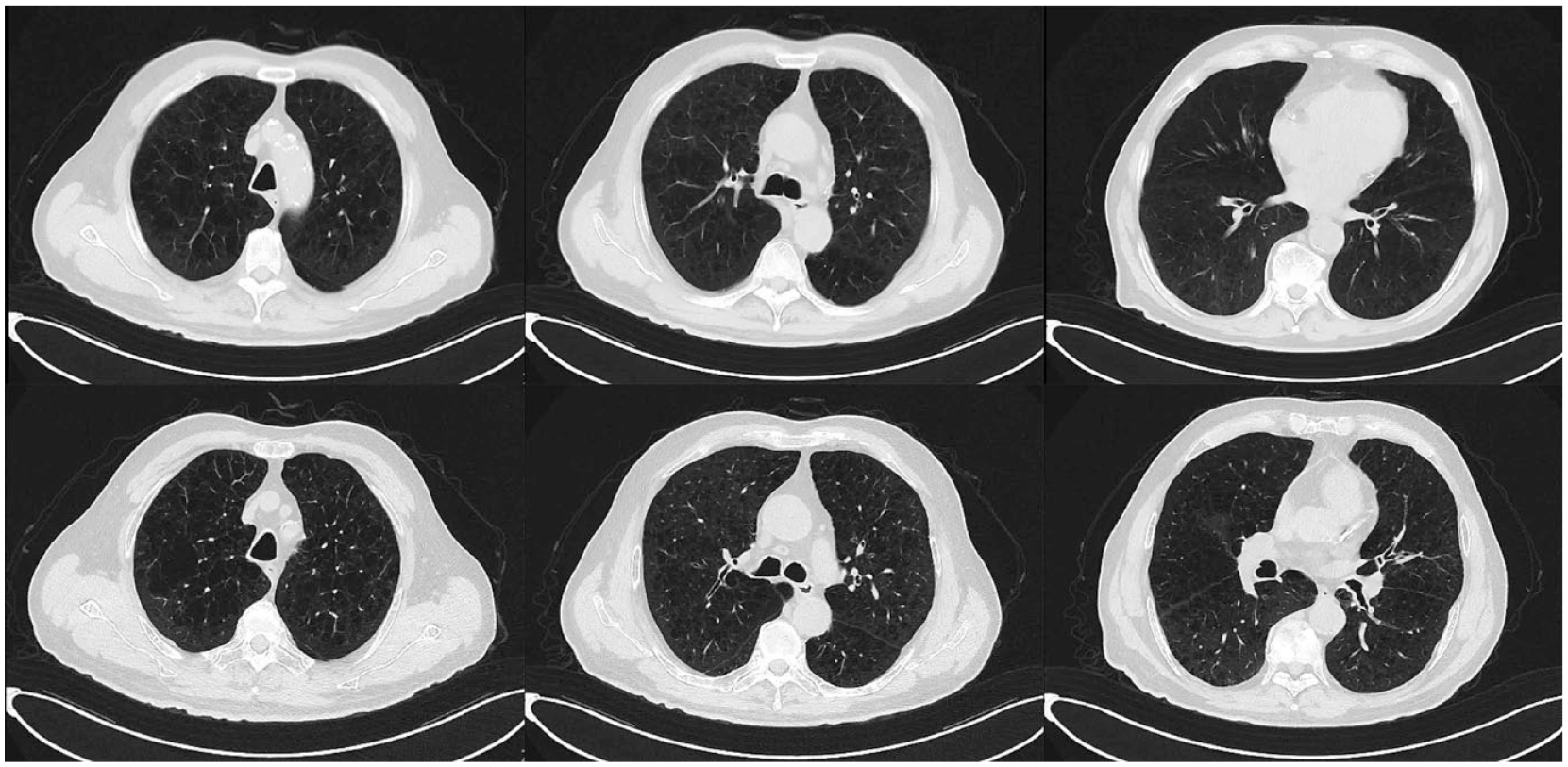
Chest CT: The image showed a symmetrical bony thorax, centered mediastinal trachea, increased and disordered texture of both lungs, uneven enhancement of transmission brightness of both lungs, multiple vesicular transparent areas in both lungs and part of the unclear boundary. A small nodular shadow (im13, 0.4 cm in length) was found in the apical segment of the right upper lobe. Bilateral pleural thickening was found, and bilateral pleural effusion was not found.
The patient had a slight reduction in chest tightness, wheezing, and shortness of breath during the first 5 days of admission, the number of asthma attacks was 1 time/day and the total duration of asthma attacks was approximately 10 minutes/day. However, the patient’s wheezing and shortness of breath suddenly worsened and he was unable to sleep flat on his back in the early hours of March 15. He was immediately treated with nebulized inhalation of budesonide suspension and salbutamol sulfate nebulized inhalation solution. There were wheezing and shortness of breath with a wheezing sound in the throat, and pitting edema in both lower limbs on the morning of March 15. The patient was asked in detail, and he recounted that he drank large amounts of water because of a dry mouth resulting from oxygen inhalation using a mask after admission. He had no history of esophageal reflux, diabetes, and diabetes insipidus and symptoms of acid regurgitation, retrosternal burning pain, and so on. In the 5 days before admission, the total daily fluid intake was 4100 ml, including about 3500 ml of drinking water and 600 ml of intravenous infusion, but the total daily fluid output was about 1000 ml, which was in a serious positive balance. However, the patient did not drink a lot of water at home, and the fluid intake and output were kept in balance. The symptoms of chest tightness, wheezing, and shortness of breath on March 15 were significantly more severe than at the time of admission, and the duration of an asthma attack was about 30 minutes. We rechecked the patient’s absolute lymphocyte value, absolute neutrophil value, ultrasensitive C-reactive protein, procalcitonin, and NT-pro BNP, which were 0.33 × 109/l, 6.06 × 109/l, 0.90 mg/l, <0.05 ng/ml, and 72.5 pg/ml. To reduce the volume load of the heart, we gave him an intravenous furosemide injection of 2 ml. The patient had 4 asthma attacks and the total duration of asthma attacks was about 90 minutes on March 15. Starting on March 16, we began to strictly control the patient’s water intake and record the patient’s daily intake and output (Figure 2). The patient was treated with furosemide injection (2 ml iv) on March 16, bumetanide injection (1 mg iv) on March 17, dexamethasone sodium phosphate injection (5 mg iv pumped once daily) from March 15 to March 20, furosemide injection (4 ml iv pumped once daily) from March 18 to March 20 and on March 25 and (6 ml iv pumped once daily) from March 21 to March 24, aminophylline injection (2 ml iv pumped once daily) from March 18 to March 22, salbutamol injection (0.4 mg iv pumped once daily) from March 16 to March 18, furosemide tablets (20 mg orally once every other day) since March 16, spironolactone (20 mg orally once daily) from March 16, and sodium methylprednisolone succinate for injection (80 mg iv drip once daily) from March 20 to March 23, (40 mg iv drip) on March 24, and (20mg iv drip once daily) since March 24. The patient’s symptoms of wheezing and shortness of breath were significantly relieved, and there was no wheezing sound in the throat during asthma attacks from March 17 to 19. The number of asthma attacks per day and the total duration of asthma attacks in the patient gradually decreased from March 18 (Figure 2). He also stopped gaining too much weight, and his use of diuretics and steroids was tapered (Figure 3). The patient’s absolute lymphocyte value, absolute neutrophil value, ultrasensitive C-reactive protein, and procalcitonin were 0.72 × 109/l, 10.98 × 109/l, 1.60 mg/l, and <0.05 ng/ml on March 20. The patient was treated with moxifloxacin hydrochloride sodium chloride injection (0.4 g iv drip once daily) from March 20. Thereafter, his asthma was gradually controlled and his fluid intake and output were in dynamic balance (Figure 4). He was discharged on 30 March.

Fluid intake, fluid output, number of asthma attacks, and total duration of asthma attacks. Fluid intake = water intake + intravenous fluid volume (×103 ml); Fluid output = urine output (×103 ml); Number of asthma attacks: the total number of asthma attacks in a day; Total duration of daily asthma attacks: total duration of daily asthma attacks (×10 minutes).
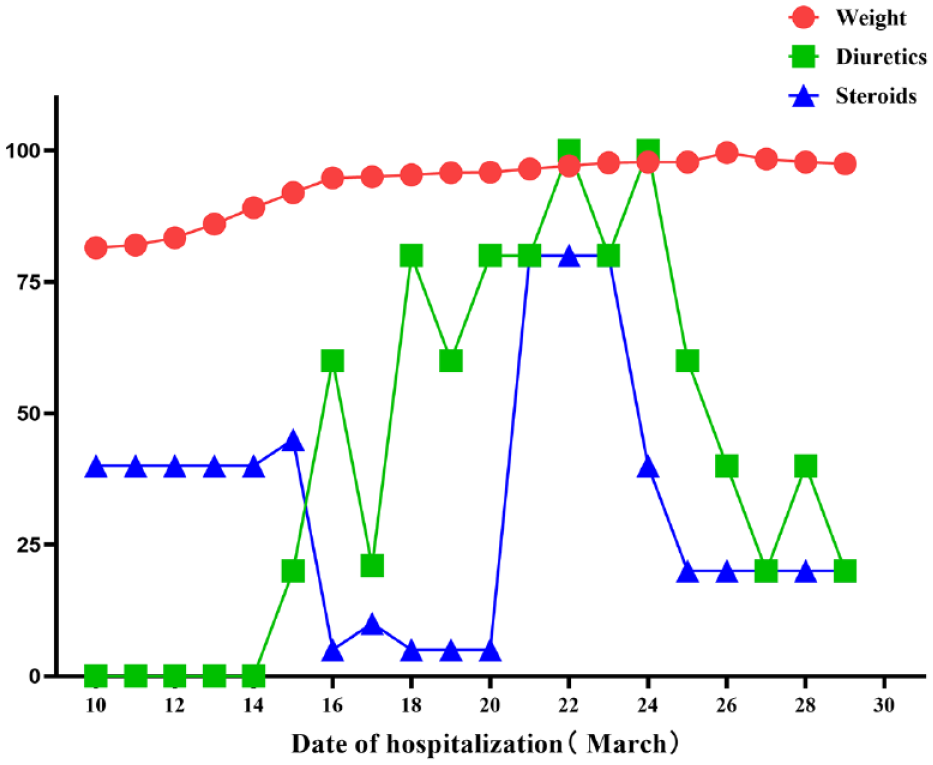
The patient’s weight and the dose of diuretics and steroids. Diuretics: furosemide injection (20 mg), bumetanide injection (1 mg), furosemide tablets (20 mg), and spironolactone (20 mg); Steroids: sodium methylprednisolone succinate for injection (40 mg) and dexamethasone sodium phosphate injection (5 mg).

Net fluid intake, number of asthma attacks, and total duration of asthma attacks. Net fluid intake = fluid intake − fluid output (×102 ml); Number of asthma attacks: the total number of asthma attacks a person has in a day; Total duration of daily asthma attacks: total duration of each asthma attack in a day (×10 minutes).
Discussion
Clinically, it appears to be rare for asthma to be induced by excessive drinking water. In this case, the cause of an asthma attack may be caused by a respiratory viral infection when the patient was first admitted to the hospital. On the sixth day of admission, the patient’s wheezing and shortness of breath worsened with a wheezing sound in the throat and pitting edema in both lower limbs, and the patient’s fluid intake and output were in severe positive equilibrium for the first 5 days of admission. More importantly, the patient’s asthma was better controlled with the addition of diuretics and control of the patient’s fluid intake and output on top of the original treatment. Therefore, we seriously suspected that the patient’s asthma exacerbation was induced by excessive drinking water.
The relationship between excessive drinking water and asthma exacerbation may be related to promoting the formation of pulmonary edema. If the intake of water in the body increases too much without an increase in excretion, this can put the body in a state of fluid overload, which may lead to pulmonary edema. 11 More importantly, the negative pleural pressure of asthma patients compared with normal people is increased more at the end of inspiration, 12 which causes an increase in pulmonary vascular volume and pulmonary capillary transmural pressure, 13 resulting in massive fluid infiltration from the pulmonary capillaries into the interstitial lung tissue, and the formation of pulmonary edema is also further promoted by the increase of pulmonary circulation blood volume.
Pulmonary edema can reflexively stimulate the vagus nerve, which causes bronchial constriction; it may also lead to the formation of edema of the tracheal mucosa, 14 both of which can cause airway hyperresponsiveness, thereby aggravating and inducing the attack of asthma. 14 Several clinical studies have demonstrated that fluid load can cause pulmonary edema, which induces asthma attacks and exacerbations. A retrospective study showed that an increase in the whole heart end-diastolic volume index in 42 critically ill patients promoted the occurrence of extravascular pulmonary water index >10 ml/kg. 15 A prospective study found that intrathoracic blood volume index and total end-diastolic volume index were both significantly and positively correlated with extravascular lung water index in 16 patients with septic shock. 16 A retrospective cohort study showed that fluid overload was significantly and positively correlated with a longer hospital stay, the duration of β-agonist therapy, and the duration of supplemental oxygen in 1175 children with acute asthma exacerbations; a prospective cohort study found that a fluid load of ⩾7% in 123 children with acute asthma exacerbation increased the risk of extravascular lung water. 10
Conclusion
Excessive drinking water may be a risk factor leading to asthma exacerbation. It can overload the body’s blood volume, which can cause the exacerbation of asthma by promoting the formation of pulmonary edema. In clinical practice, we should pay attention to the relationship between fluid load and asthma exacerbation to avoid extending the treatment time of asthma due to excessive drinking water, so that patients with asthma or those who will be affected by asthma can maintain a better quality of life.
Footnotes
Author Contributions
SL was responsible for drafting the article. SL, YQ, MZ, and RW were responsible for acquisition of data and analysis of interpretation of data. HJ, YL, FJ, and LX were responsible for conception and design of the manuscript. HJ was responsible for critically revising the article, final approval of the article, and funds collection.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a study on the Key R&D Program of Shandong Province, China (Major Scientific and Technological Innovation Project) (No. 2021SFGC0503).
Availability of Data and Materials
The data for this case report are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The requirement for ethics approval and consent for this case report was waived.
Consent
The patient in this case report signed written informed consent.
Consent for Publication
Written informed consent for publication of identifying images or other personal or clinical details was obtained from the patient.
