Abstract
Dent’s disease is a rare genetic kidney disorder characterized by proximal tubular dysfunction, nephrocalcinosis, recurrent nephrolithiasis, and chronic kidney disease. Hypercalcemia is a rare finding in this disease. In this report, we present a case of possible Dent’s disease in a young adult male with hypercalcemia and chronic kidney disease. The diagnosis was evoked based on the presence of low-molecular-weight proteinuria, kidney stones, and renal failure. This case underscores the significance of considering Dent’s disease as a potential diagnosis, even in patients with chronic renal disease who exhibit hypercalcemia. It also emphasizes the importance of regular monitoring and management of patients with this condition to prevent further complications.
Introduction
Dent’s disease is a rare X-linked recessive inherited proximal tubulopathy that mainly affects young males. 1 Low-molecular-weight proteinuria (LMWP) is the most consistent feature, occurring in 99% of affected patients. Hypercalciuria is a common abnormality that can lead to recurrent nephrolithiasis and progressive renal failure. 1 Hypercalcemia is typically not observed in Dent’s disease, and its presence for some reason could obscure the initial presentation and pose a diagnostic challenge. In this report, we present a case of nephrolithiasis due to Dent’s disease in a young patient with hypercalcemia but without hypercalciuria.
Case Report
A 31-year-old man from a consanguineous marriage was referred for investigation of recurrent nephrolithiasis. His family history noted urolithiasis in his father. No history of self-medication or dairy consumption was found. Although the patient developed urolithiasis at the age of 20 while ingesting parsley infusions, he continues to believe in the plant’s potential health benefits for the kidneys and its ability to effectively treat kidney stones. Since that, he has had multiple recurrences, notably on the right kidney. He underwent bilateral extracorporeal lithotripsy with the placement of JJ catheters and bilateral surgery. On physical examination at admission, the patient’s height was 196 cm, weight was 74 kg, and body mass index was 19.2. His blood pressure was 100/60 mmHg. Urine dipstick showed no proteinuria, hematuria, or glycosuria. Additionally, there were no other extrarenal manifestations.
An analysis of a right coral-shaped stone removed by surgery at the age of 30 revealed calcium-dependent lithiasis with Weddellite (50%), Carbapatite (40%), Anhydrous Ammonium Acid Urate (5%), Carbonated Amorphous Calcium Phosphate (1%), and Whelwellite (1%).
For both the blood and urine collections, standard protocols were followed. For the blood collection, it was done in the morning after an overnight fast of 8 to 12 hours, with the patient instructed to avoid excessive physical activity or eating a large meal before the blood draw. For the urine collection, the patient was given a 24-hour urine collection container and instructed to collect all urine produced during this period. The patient was also instructed to store the urine in a cool place or in a refrigerator during the collection period and avoid excessive fluid intake or dehydration during the collection period to ensure accurate test results. He had all the tests performed as an outpatient in the nephrology department, and he was not suffering from renal colic at the time of the tests. Urinalysis revealed normal calciuria and phosphaturia, as well as a LMWP at 3 g/24 hours. Chemistry panel showed serum albumin level of 53 g/L. Blood tests revealed hypercalcemia (2.77 mmol/L) and a serum creatinine of 200 µmol/L, with an estimated glomerular filtration rate (eGFR) of 36.4 mL/min/1.73 m2, as calculated by the MDRD equation, the patient had stage 3B chronic kidney disease and a parathyroid hormone level of 19.1 pg/mL. The patient has had several negative urine culture tests.
The patient’s autoimmune profile, including anti-nuclear antibodies, C3, and C4 complements, was normal. The immuno-electrophoretic aspect of the serum was normal with absence of free light chains in the concentrated urine. The rest of the biological data are mentioned in Tables 1 and 2. Chest radiography showed the usual cardiomediastinal silhouette without any signs of adenopathy. According to the renal ultrasound, the right kidney is small while the left kidney is compensating by being enlarged (Right kidney: 90 cm; Left kidney: 11.5 cm). Additionally, the ultrasound showed medullary nephrocalcinosis, which means there are calcium deposits in the kidney tissue, specifically in the pyramids, which appear to be excessively bright on the imaging. The patient also underwent a vesicoureteric reflux evaluation, which did not reveal any reflux.
Biological data of our patient.
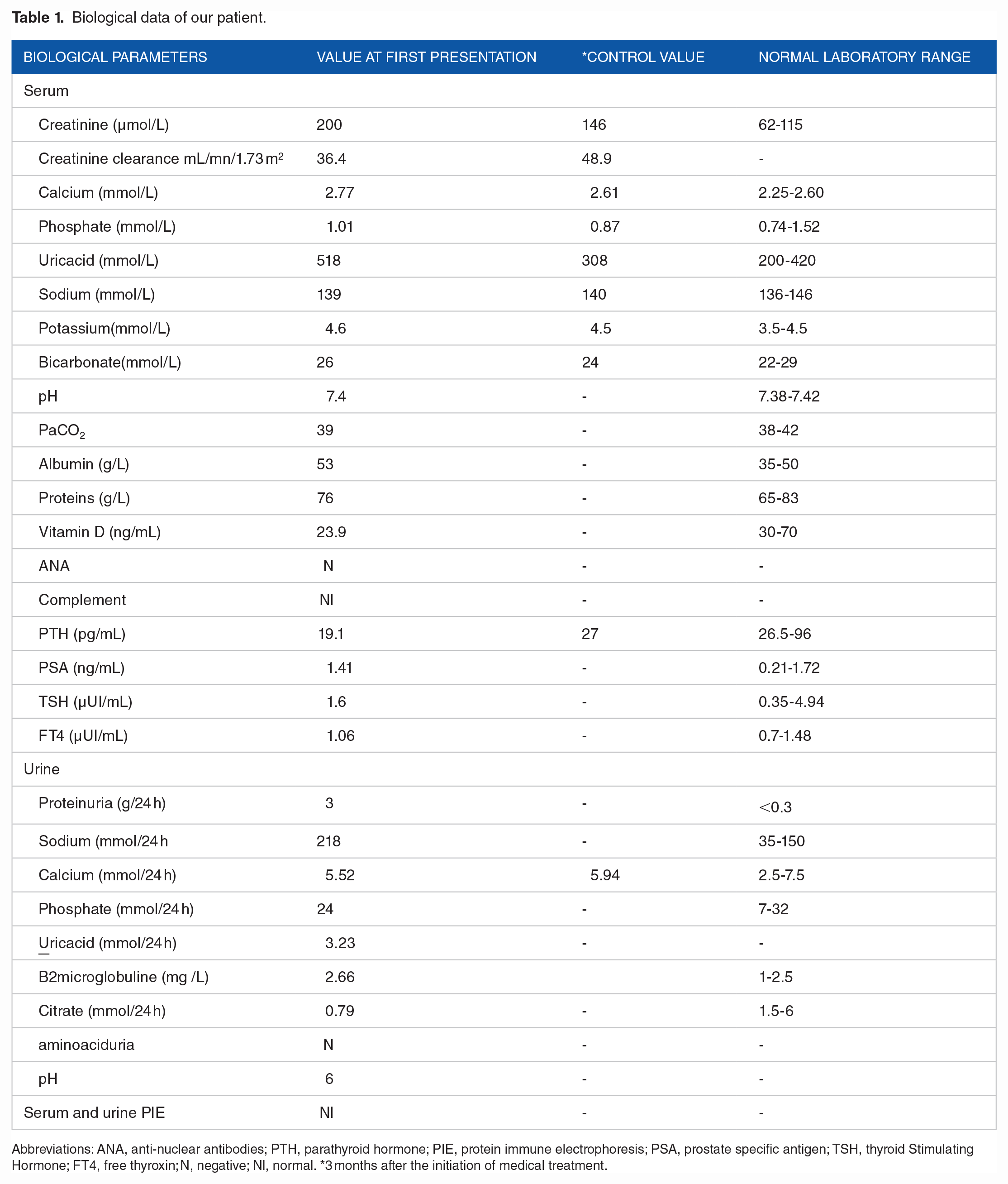
Abbreviations: ANA, anti-nuclear antibodies; PTH, parathyroid hormone; PIE, protein immune electrophoresis; PSA, prostate specific antigen; TSH, thyroid Stimulating Hormone; FT4, free thyroxin; N, negative; Nl, normal. *3 months after the initiation of medical treatment.
Twenty-four-hour urine analysis of our patient at admission.

Nutritional intake assessment:
Salt: intake of 12.8 g/day (recommended intake: 6-8 g/day).
Protein: intake of 69 g/day, which corresponds to 0.9 g/kg/day (recommended intake: 0.8-1 g/kg/day).
Treatment included dietary hygiene rules, overdiuresis, a low-sodium and oxalate diet with only 1 g of calcium intake, and allopurinol for his hyperuricemia. Three months after the initiation of medical treatment, serum calcium, creatinine and uric acid were trending toward normal values (Table 1).
Discussion
Our patient presented with calcium nephrolithiasis, as the stone analysis showed that the major component was Weddellite (50%). The rate of carbapatite greater than 15%, as well as its association with anhydrous ammonium acid urate and amorphous phosphate, indicates the presence of a urinary tract urease infection. The presence of whewellite is likely due to parsley infusions ingestion, which is a source of oxalate.
A large number of metabolic dysregulations have been reported to cause calcium stones. 2 When assessing the etiology of urolithiasis, the presence or absence of hypercalcemia is used to guide us toward the cause. If hypercalcemia is detected, the first diagnosis that comes to mind is primary hyperparathyroidism. However, if this diagnosis is ruled out, clinicians should also investigate sarcoidosis, especially in a young patient like the one in our case. Other potential causes to consider include vitamin D excess, malignancies, or hyperthyroidism. We carefully evaluated and ruled out these potential diagnoses, including systemic disorders that could cause hypercalcemia (Table 3). It is worth noting that despite having hypercalcemia without hypercalciuria, our patient did have a vitamin D deficiency, which is an important consideration when evaluating hypercalcemia. Additionally, the negative immunological assessment suggests that our patient’s condition was not caused by an autoimmune disorder.
Clinical and laboratory features supporting and contradicting the diagnosis of dent’s disease, and differential diagnoses.

We considered renal tubular acidosis to be an unlikely explanation for the patient’s condition, given that their serum levels of HCO3 were normal, and their aminoaciduria was negative. Furthermore, the patient’s urine pH was normal, and the observed hypocitraturia could be explained by the infection lithiasis or by Dent’s disease, as it has been reported that Dent’s disease patients with reduced renal function may present with hypocitraturia. 3 The patient’s clinical presentation and the absence of specific metabolic abnormalities also excluded Bartter syndrome as a possible cause.
Dent’s disease generally presents with significant hypercalciuria, mostly of the LMWP type, along with nephrocalcinosis, nephrolithiasis, and chronic kidney disease.3-5 We present the case of a young man who exhibited proteinuria in the nephrotic range. The presence of LMWP in our patient suggests proximal tubular dysfunction, and Dent’s disease was considered the most likely diagnosis. In fact, there is a form of Dent’s disease that occurs in only 15% of patients and is characterized by progressive renal failure, and the signs of Fanconi syndrome, which consist of aminoaciduria, glucosuria, and renal tubular acidosis. 8 One or more of these signs may be absent, which implies partial Fanconi syndrome, as is probably the case with our patient, in whom no aminoaciduria was found. Our patient had pre-existing chronic kidney failure with an eGFR of 36.4 mL/min/1.73 m2, which could explain the mild hypercalcemia without hypercalciuria. This may be linked to the dysregulation of renal calcium handling that can occur in various medical conditions, such as medullary sponge kidney or inherited renal tubulopathies.6,7
Dent’s disease refers to a heterogeneous group of X-linked disorders caused by mutations in either the CLCN5 (Dent disease1) or OCRL1 (Dent disease2) genes located on chromosomexp11.22 and xq25, respectively.7-11 Despite the fact that consanguinity has been reported in a few studies as a risk factor for Dent’s disease12,13 and genetic testing being the preferred method for confirming the diagnosis, it was not possible in our case due to limited availability of molecular genetic tests in our country. Additionally, genetic testing can be expensive and may not be covered by insurance, further limiting our ability to confirm the diagnosis through this method.
As there are no standardized treatment protocols or guidelines for individuals with Dent’s disease, treatment is mainly supportive and focuses on managing hypercalciuria and preventing nephrolithiasis.14,15 The rarity of the disease means that there have been no treatment trials conducted on a large group of patients.8-12 End-stage renal disease occurs in 30-80% of affected males between the third and fifth decades of life.2-5 In our patient’s case, the chronic renal failure partially improved with medical treatment and dietary adjustments. It’s important to note that there is significant intra-familial variability for prognostic severity, and so regular follow-up and monitoring will be necessary to manage and treat the patient’s condition.
Conclusion
The unusual presentation of hypercalcemia in our patient prompted us to consider several potential causes before arriving at a diagnosis of Dent’s disease. The presence of LMWP was a key factor in leading to this conclusion. This case highlights the importance of approaching calcium and phosphate metabolism disturbances with caution in the presence of renal failure. It is important to continue monitoring and managing the patient’s condition and to remain vigilant for potential complications, given the significant intra-familial variability for prognostic severity associated with Dent’s disease.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
M.H wrote and designed the manuscript. H.K conceived the presented case report and was in charge of the patient. K.B provided the patient’s biochemical analysis and interpreted his urinary lithiasis assessment. F.BH and E.A supervised the article. All authors discussed the results and contributed to the final manuscript.
