Abstract
Non-obstructive azoospermia (NOA) is the most difficult form of male infertility to manage. It usually requires sperm retrieval from the testis, which is most challenging due to sperm rarity. Here, we describe the recovery of testicular sperms that had been missing and whose original retrieval results were negative. Salvage microsurgical testicular sperm extraction and sperm testing were performed on a 36-year-old male with NOA. Neither in the operation room nor after an inspection in the embryology laboratory were any sperm detected. The obtained tissue was advised to be frozen because the patient data and surgical microscopy predicted a favorable outcome, and the tissue processing was done in an inappropriate environment. About 1 month later, the specimen was thawed, crushed, and re-examined. Successful oocyte fertilization resulted from an effective detection of sperms and their direction to intra-cytoplasmic sperm injection. This is the first case report that, to the best of our knowledge, describes the stepwise laboratory processing of testicular tissue and its capacity to recover lost sperms in challenging NOA cases and under less-than-ideal working conditions.
Introduction
Non-obstructive azoospermia (NOA) is diagnosed in about 60% of infertile men with azoospermia. 1 Sperms have been demonstrated to be present in limited loci of active spermatogenesis throughout the testes of men with NOA. 2 Because of this restricted existence of sperms, microsurgical testicular sperm extraction (mTESE) is considered now the best surgical technique to retrieve rare sperms in NOA patients. 3 With just a single sperm retrieved during mTESE, this will enable couples to move forward with ICSI with the prospect of becoming parents, making such protocol the optimum current treatment of NOA. 4 Hence, the sperms from males with NOA may be valued as highly as gold.
The sperm retrieval rate (SRR) associated with mTESE may vary between 20% and 70.8%.5-18 This discrepancy in the SRR range is related to peculiar clinical and laboratory features of patients, the skills of the andrologists to perform the microsurgical technique, and the embryologist’s skills to accomplish a certain laboratory technique for tissue processing to isolate testicular sperms.7,19 Salvage mTESE provides a second chance when the testicular sperm extraction initial trial fails. However, attempting to salvage mTESE with a futile outcome after previously failing to extract testicular sperm can be emotionally draining. This will cause a couple to lose faith in ICSI therapy and their ability to have a child as a result.
In this case report study, we describe how we successfully rescued testicular sperms, via a staged processing of testicular tissue, after salvage mTESE in an infertile male with NOA when the embryology laboratory first indicated that there was no sperm.
Case Presentation
A 36-year-old man, who had been experiencing primary infertility for 13 years, came to the Andrology Clinic, Fertility Center X, Egypt. He received a NOA diagnosis at another reproductive clinic 4 years prior. The diagnosis was based on the absence of sperm in many subsequent examinations of semen, and the testicular tissue collected via the conventional TESE operation. The following histological analysis, which revealed Sertoli-only cell syndrome (SOS), further supported this diagnosis.
The patient denied using any recreational drugs and was not a smoker or an alcoholic. He stood 176 cm tall, weighed 76 kg, and had a 24.5 BMI. The patient’s prior medical history was irrelevant with no undescended testes, sexually transmitted illnesses, scrotal injuries, or infertility-related medications such as chemotherapeutics or anabolic steroids. The patient’s most recent treatment was antioxidant therapy 1 year ago. The results of the general physical examination were unremarkable. An examination of the scrotum revealed properly positioned testicles with typical consistency and no anomalies. The scrotal duplex scanning did not reveal any signs of varicocele or other scrotal diseases, and the right testis measured 12.5 cc and the left testis measured 12 cc. The hormonal assay detected low testosterone (1.87; the normal range, 2.49-8.36 ng/ml), but normal levels of other hormones including luteinizing hormone (3.14; normal range, 1.5-9.3 mIU/ml), follicle-stimulating hormone (FSH) (5.47; normal range, 1.4-18.1 mIU/ml) and prolactin (5.8; the normal range, 4.04-15.2 ng/ml). Using Multiplex Real Time PCR, no Y-chromosome microdeletions were found. The patient received counseling and had a salvage mTESE operation arranged.
Around 2:30 PM, the salvage mTESE surgery was carried out using the surgical microscope (X25) while under general anesthesia. The treatment was carried out in accordance with Schlegel’s original description, 5 which called for extending a coronal incision in the right testis from the antimesenteric region to the tunica albuginea. Small samples were obtained from opaque, relatively dilated tubules in the testicular parenchyma, which otherwise showed uniformly thin tubules throughout. These opaque dilated tubules are hallmarks of sperm-containing tubules,3,5-18 (the reader is kindly directed to the reference 5 for the highly illustrative images of these tubules). According to the scheme followed by the embryology laboratory in this fertility center (Figure 1), the samples were placed in Petri plates containing erythrocyte lysing buffer and HEPES-buffered Earle’s medium supplemented with 0.5% human serum albumin (Sigma, Cairo, Egypt) and moved to the embryology laboratory. Yet, no sperm could be identified intraoperatively while inspecting with an inverted microscope (X400) after forcefully crushing the extracted testicular tissue with a plastic syringe pistole. The left testis was, therefore, examined. It also displayed the same thin picture of its tubules. Nevertheless, a small number of opaque tubules that were thicker than the others could be seen. Similar to how the tubules of the right testis were handled, these opaque tubules were also collected. However, the embryologist also reported the same outcome about the lack of sperm. The crushed tissue afterward underwent laboratory processing, including centrifugation (1500 rpm). The pellets that were created were suspended in culture fluid and microscopically examined. Regrettably, there was no sperm to find during the cell suspension’s microscopic testing, which was conducted by 2 young technicians from 5:00 to 7:00 PM. The operating andrologist recommended keeping the processed tissue in liquid nitrogen to give the suspension inspection a second opportunity in the near future based on the patient data and the hopeful look of the obtained seminiferous tubules. The embryologist plunged the processed tissue in liquid nitrogen except for a part, which was sent for the histopathological examination (Figure 1). Because no sperms were available, the retrieved eggs of the wife were, therefore, subjected to vitrification.

Flow chart illustrating the manipulation of the collected testicular tissue after TESE/mTESE.
About 1 month later, the embryology laboratory got the histopathological report of the tissue sample. The report showed the existence of about 20% of the seminiferous tubules with full spermatogenesis with otherwise SOS. Early in the working day, two embryologists with greater expertise allowed the frozen suspension of processed testicular tissue to thaw, promptly hastened the suspension inspection and crushed any previously leftover large or rough tissue fragments. Fortunately, after an inverted microscopic inspection, 3 sperms could be recognized. The wife’s frozen eggs had thawed. The sperms were utilized right away for ICSI. A total of 3 eggs were successfully fertilized. Three embryos (day 3) were transferred. Two weeks later, the hCG test was, unluckily, negative.
Discussion
The present manuscript is a case report suggesting a staged approach for laboratory tissue processing following a salvage mTESE in a patient with NOA and SOS pathology. This staged approach was able to revert the initial embryology report of sperm lack in the tissue samples and detect rescuing the existent rare sperms.
The mTESE represented a breakthrough in the management of NOA. 5 This methodology has a high SRR, which is 1.5 times more than traditional TESE and 3 times greater than testicular sperm aspiration. 11 However, the SRR after mTESE seldom exceeds 70%.5-18This may largely come from the exceedingly rare and difficult-to-harvest testicular sperm present in males with NOA. 20
In the present report, the initial handling of testicular tissue expedited by the embryology laboratory was unable to identify any sperm in the patient’s samples. However, this laboratory treatment has a number of problems. First, by a pistole of a plastic syringe within a Petri dish, mechanical crushing was accomplished, resulting in rough shredding. According to several studies, fine mincing with pincettes is the most effective way to get small delicate tissue fragments, rupture the seminiferous tubules, and release the most testicular sperms. 21 It is known that rough tissue pieces are able to make a barrier prohibiting sperm transit out of the tissue suspension. 22 Second, in the course of digesting the tissue, no angiocatheter was used. The rupture of seminiferous tubules and extraction of sperm can be aided by passing the treated tissues through the angiocatheter, which may enhance sperm output by roughly 470% according to some reports. 7
Third, 1500 rpm was the centrifugation speed employed. This pace was lower than that of other employees who utilized 500 G 7 , equivalent to 5000 rpm (personal communication). It is generally known that improper centrifugation speed can lead to poor sperm recovery. 23 Fourth, centrifugation was carried out in test tubes without using a density gradient, which is a better way to push the responsible cells to the tube’s bottom. 22 In addition, if no density gradient was used, big chunks of rough tissue would fall toward the bottom along with the cells of interest. Fifth, the unsuccessful mechanical crushing attempt at sperm recovery was not followed by enzymatic treatment, which is widely recognized by researchers to increase the likelihood of sperm recovery. 7 Using enzymatic digestion in such a salvage instance, which had higher fibrosis after the prior testicular surgery, 24 may be very beneficial. Sixth, the processing of recovered testicular tissue was accelerated toward the end of the day in a busy fertility management center. It is quite probable that human mistake led to occasional sperm loss due to weariness. 25 Seventh, the embryologists in charge of handling this tissue sample have just recently started to process testicular tissue in the laboratory to extract sperm. In many publications, the experience of the embryologist is a crucial requirement for performing mTESE and effective sperm retrieval.3,7,19,21,22,25 Moreover, testicular sperms are typically immotile, occasional, 20 as in the case report presented here, and challenging to distinguish from adjacent cells, a task requiring expertise. Eighth, in this challenging salvage instance, the microscopic inspection time (2 hours) was not long enough to thoroughly examine the processed testicular tissue. The chance of successfully retrieving sperm is positively related to the amount of time spent looking for it. This sperm searching can take up to 12 to 14 hours.25,26 This short microscopic inspection, in this case report, might easily overlook rare sperm.25-27
On the other hand, several observations in our patient suggested that testicular sperm could be present. First, upon microscopic magnification, some tubules were opaque and dilated, which are characteristics of tubules containing sperm. 3 , 5 - 18 Second, the patient had SOS. According to several researchers, this kind of testicular histopathology has a higher possibility of sperm recovery than other types like maturation arrest. 28 In addition, tubular hyalinization, which is often linked to poor SRR, 29 was not existent in the patient’s pathology. Third, the patient’s FSH level (5.47 mIU/ml) was within the normal range. According to Yücel et al, 29 FSH is significantly lower in patients who successfully have salvage mTESE than it is in those who have unsuccessful salvage mTESE. FSH levels are generally not predictive of sperm retrieval for primary mTESE instances, as has been widely published in recent clinical articles (Table 1).9,10,12-15,17 In addition, recent comprehensive systemic reviews and meta-analyses 30 as well as summaries of the literature 31 on this subject reached the same conclusion. On the other hand, the lack of enough studies and data makes it difficult to draw firm conclusions on the predictive value of FSH level to forecast SRR in instances of salvage mTESE (Table 2).28,29,32-34 This is especially true when a patient, like the one described above, falls into the SOS category. Although SSR in the SOS categories was recorded in these investigations (Table 2), the corresponding FSH levels in these SOS categories were not. It is also noteworthy that in these articles presenting experience with salvage mTESE, precise tissue processing is not addressed.
Studies reporting the FSH levels in NOA and the associated SRR after primary mTESE.

Abbreviations: FSH, follicle-stimulating hormone; NOA, nonobstructive azoospermia; SRR, sperm retrieval rate; mTESE, microsurgical testicular sperm extraction.
Studies reporting the FSH levels in NOA and the associated SRR in all NOA groups and the SOS category after salvage Mtese.
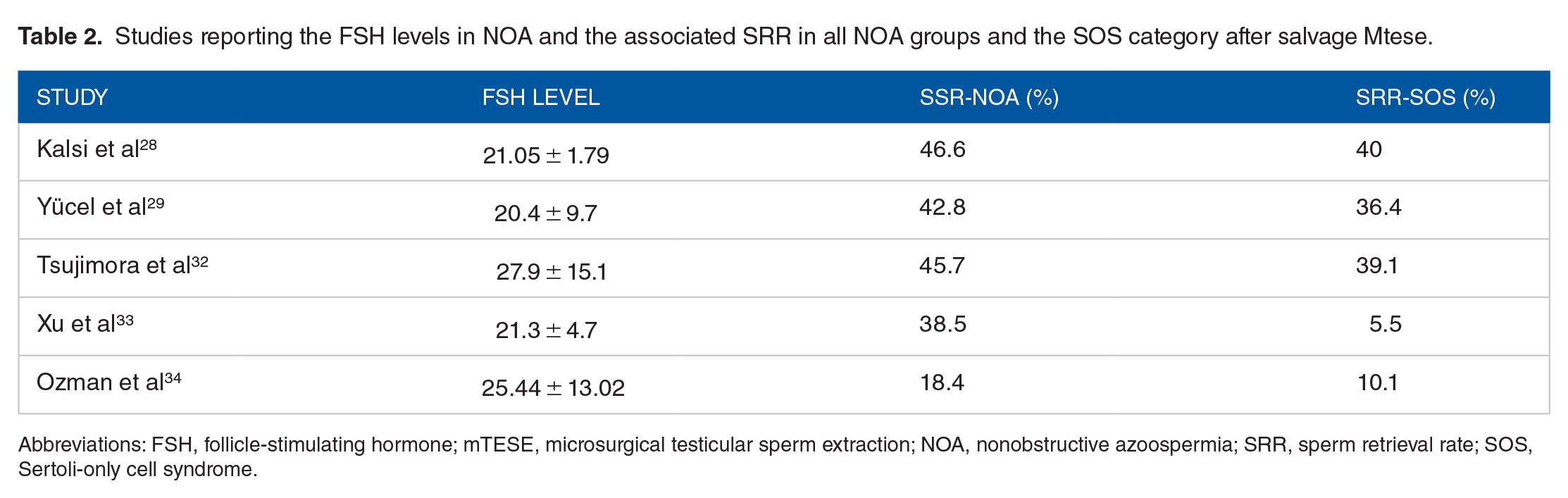
Abbreviations: FSH, follicle-stimulating hormone; mTESE, microsurgical testicular sperm extraction; NOA, nonobstructive azoospermia; SRR, sperm retrieval rate; SOS, Sertoli-only cell syndrome.
The retrieved testicular tissue in the current case report was preserved by freezing till the subsequent evaluation. The purpose of preserving the tissue was to optimize working conditions so that more seasoned embryologists might be accessible, who were not available in the initial session. Those experienced embryologists speeded up the process early on the day to prevent worker tiredness and the loss of rare sperms. In addition, the freezing method along with crushing the frozen tissue have similarly employed by other researchers, who hypothesized that the freezing-crushing procedure could increase the SRR. 6 This combined strategy was designed to crack open additional seminiferous tubules, increasing the likelihood of finding hidden sperm that have not been discharged with crushing alone.
The fact that the current study is a single case report with no strong conclusion on the efficacy of this staged laboratory processing to recover rare sperm may spark discussion. The rescue of these scarce sperm could be an incidental finding while doing a second stage of microscopic check-up. The freezing-crushing measure employed, however, can not be ignored in this situation. This measure depended on a well-controlled investigation, with no place for a second microscopic inspection. A statistically significant influence on sperm retrieval was seen as a result of the measure. 6
It is becoming more and more obvious that the processing of testicular tissue in the embryology laboratory after doing mTESE and the surgical procedure itself have a proportional impact on the net outcome of finding sperm. 19 This was the situation with the patient who was reported, where proper laboratory processing could transform a negative result in surgical sperm collection into a positive result. This again supports the findings of other studies. 7
With the establishment of a learning curve 35 and improvements in microsurgical sperm extraction, 5 andrologists can more effectively harvest seminiferous tubules that are more likely to contain sperms. However, laboratory tissue processing after mTESE continues to be difficult because the procedure is currently carried out manually, can take long hours of attentive examination, and is reliant on the examiner’s level of expertise, level of fatigue, and capacity to see sperm.25 -27 It is remarkable that there are now no standardized guidelines or learning curves for laboratory skills for tissue processing. 19
For improved sperm retrieval in patients with NOA, a number of innovations have recently been tested. These include magnetic-activated cell sorting, which uses superparamagnetic nanoparticles and columns to detach individual cells from diverse cell populations, microfluidics, which uses tiny gushes of fluid to isolate sperm, and fluorescence-activated cell sorting, in which fluorescent-marked cells are collected based on light emitted from a laser source.19,25 In addition, Lee et al devised a machine learning algorithm to identify rare sperm, with 95.8% sensitivity and 91% positive predictive value. 36 These new advances may make it possible to find a few rare sperms in patients with NOA who previously had non found.
Conclusion
The staged approach of handling testicular tissue after salvage mTESE may be a valid option, which successfully saves existing rare sperms. This will offer excellent potential for increasing the SRR and allow an infertile couple the opportunity to move on with ICSI and have a child. Andrologists should keep this integrated approach in mind while doing surgical sperm extraction and discovering promising testicular tissue. This is especially true in salvage instances under unfavorable working conditions and upon receiving early indications of sperm deficiency from the embryologist. Once more, there is an urgent need for standardized guidelines for testicular tissue processing in embryology laboratories, along with the establishment of a learning curve. New cell-sorting devices that can harvest a big number of sperm are still needed in the meanwhile.
Footnotes
Author Contributions
N.S. made contributions to the study’s conception and design, data analysis and interpretation, and the article’s final approval of its format. O.S.H. helped to develop the draft and gave final permission to its format.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The patient has consented to his case information for publication purpose.
