Abstract
Lambl’s excrescence (LE), a rare thin linear fibrous thread of collagen and elastic fibrous tissue originating at closure margins of heart valve leaflets is considered a rare cause of thromboembolism, causing ischemic stroke, transient ischemic attack (TIA), acute coronary syndrome, or peripheral thromboembolism. The gold standard for diagnosing LE is transesophageal echocardiography (TEE). Due to the rarity of the disease and lack of significant research, no guidelines exist about the management of LE. Twenty-two papers about LE were reviewed, focusing on the management aspect aiming to assist in guideline publication. Articles were retrieved using PubMed database using “Lambl’s excrescences” as a keyword. All free full text papers up to 2023 were retrieved. In this report, we present the first publication of a case of a TIA secondary to LE in the United Arab Emirates. A 53-year-old male presented with transient attacks of left monocular blindness with complete recovery 2 days later. Extensive stroke work-up was negative, except for TEE, which showed an echo-dense linear mobile structure attached to the aortic surface of the aortic valve closure margin, suggesting LE. As no guidelines exist on the management of LE, the patient was started on Clopidogrel 75 mg daily and remained asymptomatic for 2 years. After reviewing the literature, we suggest the use of Aspirin for patients with ⩾1 embolic event attributed to LE while not on antiplatelets or anticoagulants, as it is the most used and recommended treatment option and is effective in preventing recurrent thromboembolic events.
Background
According to the Centers for Disease Control and Prevention (CDC), ischemic pathologies contribute to 87% of strokes in the United States, of which 14%-30% are found to be from a cardiac source, while 24%-28% are cryptogenic in origin.1,2 Lambl’s Excrescences (LEs) were first reported in 1856 by the physician Vilém Dušan Lambl. 3 They are rare single or multiple thin linear mobile fibrous threads originating at valve leaflets closure margins usually at the Arantius nodules, measuring ⩽2 mm in thickness and ⩾3 mm in length without gender predominance.3 -5 LEs are rarely seen on other heart structures such as papillary muscles, chordae tendineae, or atrial septum. 6 It was found that thromboembolic risks are independent of excrescences sizes. 1 A giant LE result when multiple adjacent excrescences join and form a complex. 6 Excrescences tend to occur in healthy people with increasing incidence with aging, where prevalence peaks between the age of 61 and 70, and then drops due to increased valve calcification, resulting in excrescences being masked under echocardiography.3,6 Wear and tear endothelial injury contributes to excrescences development that’s influenced by high pressure at the valve leaflets closure lines resulting in fibrin deposition and endothelial overgrowth, which explains the reason behind the left heart valves being significantly more affected than the right valves. 3 Excrescences are made of collagen and elastic fibrous tissues, surrounded by fibrin and mucopolysaccharide matrix covered by a single endocardial layer.3,5 Transesophageal echocardiography (TEE) is the gold standard tool for diagnosing LEs, having higher sensitivity (68%) and specificity (85%) than transthoracic echocardiography (TTE), where TTE missed 50% (7 out of 14) cases.3,7 Cardiac CT scan can be an alternative option to TEE when TEE is unavailable. 6 However, the only measure to reach a definitive diagnosis is histological evaluation of the lesion. 6 Due to disease rarity and the lack of statistically significant researches, there are no guidelines about LE management. 3 Management is primarily based on previous published case reports. In this report, we present the first publication of a case of a transient ischemic attack (TIA) secondary to LE in the United Arab Emirates. Additionally, 22 articles about LE were reviewed, focusing on the management aspect aiming to assist in guideline publication.
Case Report
A 53-year-old male patient with a past medical history of hypertension and dyslipidemia presented with transient attacks of left monocular blindness on 19-Feb-2021 with complete recovery 2 days later. The patient denied any other symptoms. The patient was on Atorvastatin and Candesartan for dyslipidemia and hypertension, respectively. Dyslipidemia was appropriately controlled while the blood pressure was not. The patient is a tobacco smoker. Physical examination showed an oriented, alert, and conscious patient, with a blood pressure of 158/93 mmHg. Other vitals were stable, with unremarkable cardiac and neurological examinations.
The patient underwent extensive stroke workup to identify the underlying cause. ECG showed sinus rhythm. TTE revealed normal chambers sizes, mildly impaired left ventricular diastolic function with an ejection fraction of 60%-65%. Carotid doppler ultrasonography and cerebral CT angiography with contrast were negative for stenosis, atherosclerotic changes, and occlusions. Brain magnetic resonance imaging showed chronic ischemic periventricular changes without signs of acute infarction. Laboratory results of coagulation profile, inflammatory markers, and blood culture were negative, which excluded hypercoagulable states and infective endocarditis vegetations, respectively.
TEE with bubble study showed an echo-dense linear mobile structure attached to the aortic surface on the aortic valve closure margin, suggesting LE, without evidence of a patent foramen ovale, left atrial appendage thrombus, or filling defects (Figure 1). Multiple TEE views from different angles excluded imaging artifacts. Fluorescein angiography showed a filling defect of the cilioretinal artery. The patient was started on Clopidogrel 75 mg daily and has been asymptomatic for 2 years.
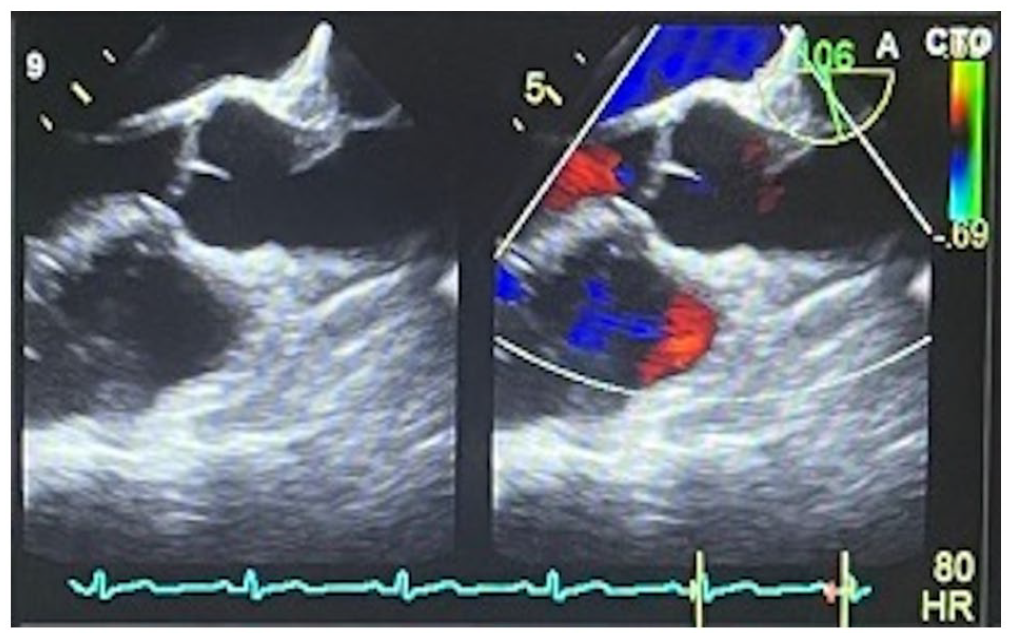
TEE showing mobile echo-dense linear structure attached to the aortic surface on the aortic valve closure line.
Methods
PubMed database was used to retrieve and review articles using “Lambl’s excrescences” as a keyword. All free full text papers up to 2023 were retrieved, which resulted in 33 studies. Out of the 33 studies, 11 were excluded. Four of them described giant LEs, which have different clinical presentations, anatomic locations, and management approaches. Three studies were excluded as they described cardiac papillary fibroelastomas (PFEs), 2 studies described cardiac tumors, while 1 study described LEs in fish, and 1 in dolphins. A total of 22 articles in English language about LEs were reviewed with an emphasis on management approaches.
Results and Discussion
LEs differential diagnoses include imaging artifacts, PFEs, vegetations, thrombus, and redundant valve leaflet, where cardiac CT scan aids in differentiating between each. 7 Valve vegetations appear irregular with different echogenicity from heart valves and have tendency to occur at any valvular area. 6 PFEs are often single, pedunculated, large, bulky with finger-like projections, and can originate from any endocardial surface away from valvular closure margins with higher embolization risk compared to LEs.3,8 3D TEE helps in differentiating LE from PFE. 9 Histological evaluation is the confirmatory tool in differentiating a LE from a PFE, where a single endocardial layer covers a LE compared to multiple layers in a PFE. 8
LE is a rare disease with increasing incidence and prevalence attributed to advanced equipment and increased stroke evaluation using echocardiography.6,10 LEs were found in 5.5% of 1559 patients evaluated by TEE and in 0.94% of patients between 61 and 70 years old who underwent TTE and TEE. 11 LE was present in 22% of patients who underwent TEE following a stroke. 7 Stroke patients with LE were younger (58.1 ± 15.4 years) compared to stroke cases without LE (68.6 ± 14.9 years), and had fewer stroke risk factors. 12
While most LEs are asymptomatic and found incidentally on echocardiography or autopsy, they are considered as a rare cause of thromboembolism, commonly from the aortic valve, either through fragmentation or microthrombi formation on top of the lesions, leading to ischemic stroke, TIA, acute coronary syndrome, or peripheral thromboembolism (retinal, renal, popliteal).1 -10,12 -17 A study involving 224 patients with recent stroke and 85 controls found LE present in 74 patients (33%) compared to only 6 patients (7.1%) from the control group. 18 It was concluded that LE is strongly associated with recent ischemic strokes in a multivariate analysis (odds ratio: 6.93, 90% confidence interval: 3.18-15.12; P-value < .001). 18 An autopsy was performed on a patient with stroke, metastatic colon cancer, Trousseau syndrome, and LE, which found similar histological findings of the right middle cerebral artery embolus and aortic valve LE lesion, which suggests LE as the cause of stroke under the influence of hypercoagulability state from Trousseau syndrome. 19 A higher LE prevalence was found in patients with embolic events (10.6% of 597 patients) compared to patients without embolic events (2.3% of 962 patients). 20 There is a clinical association between LEs and thromboembolic events, but no direct causal relationship is established to date. 6
One of the causes of the lack of identification of a causal association between LEs and embolic events is that these patients had other accompanying cardiovascular risk factors. 12 On the contrary side, several studies opposed the link between LE and thromboembolism, where they reported similar incidences of stroke and TIA among patients with and without LE, and supported the idea that no relation exists between LEs and strokes/TIAs.8,12,20,21 The opposing outcomes can be due to a small sample size, low stroke/TIA events, absent histological confirmation, misclassifying echocardiographic findings, short-term follow-up, and the wide confidence intervals, which means less statistical power in identifying small relations between LEs and cryptogenic strokes.8,12 No increased risk of stroke/TIA was noticed in 28 patients with LE and aortic valve stenosis when compared to 48 patients with aortic valve stenosis without LE undergoing transcatheter aortic valve implantation (TAVI). 22 However, the presence of LE along with a single embolic risk factor such as native valve calcification implements the use of cerebral protection devices due to a high embolic risk. 22
Cerebrovascular accidents (CVA) should be investigated thoroughly using carotid duplex ultrasonography, TEE, and hypercoagulability testing to determine the underlying cause, as detecting, and managing LE prevents recurrent attacks, life-threatening complications, and reduces morbidity.6,15 LE should be considered in cryptogenic strokes and TEE should be performed even with a normal TTE findings.3,10 TEE is recommended after 6-months and 1-year post-discharge to re-evaluate excrescence size. 14
We aim to study the management aspect of LE as no guidelines about management are published. Management approaches depended on 3 options, which are asymptomatic patients, patients with ⩾1 embolic event who aren’t on antiplatelets or anticoagulants, and patients with ⩾1 embolic event while on antiplatelets or anticoagulants. Management approaches were either monitoring, single antiplatelet therapy (Aspirin or Clopidogrel), dual antiplatelet therapy (DAPT; Aspirin with Clopidogrel), Warfarin, new oral anticoagulants (NOACs), Aspirin with Warfarin, or surgical intervention. Management plans from 22 studies were reviewed (Table 1).
Management approaches by 30 authors, including our case.

Abbreviations: DAPT, dual antiplatelet therapy; NOACs, novel oral anticoagulants; TEE, transesophageal echocardiography.
Four out of 5 authors (80%) decided to observe and monitor asymptomatic patients, while 1 author (20%) prescribed Aspirin, justified by the patient being a high-risk patient, having myocarditis and heart failure. Different approaches were implemented on patients with ⩾1 thromboembolic event who aren’t on antiplatelets or anticoagulants. Out of 32 management plans including our case, 10 authors (31%) used Aspirin. DAPT was used in 8 patients (25%), while Warfarin was used in 7 patients (22%). NOACs and surgical intervention were the least used options, where 2 (6.3%) and 4 (12.5%) patients received them respectively. Our patient was managed with Clopidogrel (3.1%). The most used management option for patients with ⩾1 thromboembolic event while on antiplatelets or anticoagulants was surgical intervention, where 6 out of 10 patients (60%) received it, followed by 2 patients (20%) who received Aspirin with Warfarin, and 2 patients (20%) who received DAPT. Management approaches are summarized in Figure 2.

Management options recommended and implemented by 30 authors including our case.
Data about management is lacking as there is no data in the literature comparing antiplatelets to anticoagulants in LE patients with one embolic event. 3 There is no consensus in the literature comparing the efficacy and mortality of medical and surgical approaches. 6 CVA recurrence rate is estimated at around 30% in patients with LE after the second thromboembolic attack, leading to the consideration of the surgical approach. 1 Patients not adherent to medical therapy should be offered surgical excision and valve replacement after a CVA or a TIA. 3 Management should be individualized for each patient, considering antiplatelets, anticoagulants, and surgical risks and benefits. 3 Warfarin helps in preventing secondary strokes by preventing microthrombi formation, not from lesion fragmentation. 10 A study claimed that LE can be a transient lesion and disappear over time. 16 Additionally, this study recommended that a TEE be performed just prior to undergoing surgical intervention, as the lesion disappeared on TEE during anesthesia on the day of the surgery, which resulted in the surgery being cancelled. 16 Another TEE was performed 3 weeks later, which did not show any lesion. 16
Results of various treatment approaches for cases with ⩾1 embolic attack while not on antiplatelets/anticoagulants were analyzed. Seven out of 10 patients who received Aspirin were followed-up, and the recurrence rate for an embolic attack during a period range of 2 to 36 months with a mean of 13.6 months was 0. Only 2 out of 8 patients were followed-up in the DAPT category, with no recurrence of embolic events for 6 and 36 months, respectively. Clopidogrel was used for our patient with no recurrence of embolic events during a follow-up period of 2 years. Two out of 7 patients who received Warfarin were followed-up. One of them had a single recurrent embolic event within 2 weeks, while the other patient remained asymptomatic for a follow-up period of 3 years. One of the 2 patients who received NOACs was followed-up and had no recurrent embolic event for a period of 3 months. Three out of 4 patients on whom surgery was performed, 1 was followed-up with no recurrent embolic event for 3 months. No follow-up was performed for patients who were asymptomatic. For the 10 patients who had ⩾1 embolic event while on antiplatelets/anticoagulants, only 2 were followed-up. One of them received surgery, while the other patient received DAPT. No recurrent embolic event was noted for 1 month and 1 year, respectively.
As seen by reviewing and analyzing the data, close follow-up is the recommended plan for asymptomatic patients. Aspirin is the most used and recommended treatment option for patients with ⩾1 embolic event while not on antiplatelets or anticoagulants. Additionally, Aspirin showed an effectiveness rate of 100% in this group of patients, as no recurrence of thromboembolic events was noted. A proper conclusion about other treatment options for this group cannot be formulated as the follow-up data were limited compared to Aspirin group. For patients with ⩾1 embolic event while on antiplatelets or anticoagulants, the most frequent treatment suggested was surgical intervention. Follow-up in that group is limited which hinders a significant conclusion.
Conclusion
LEs were attributed to be the cause of cryptogenic strokes/TIAs and should be suspected and evaluated for by TEE in these cases. Different management strategies of LE were found upon reviewing the literature. For LE patients with ⩾1 embolic attack not on antiplatelets or anticoagulants, Aspirin should be used as it was effective in preventing recurrence of thromboembolic events and was the most frequently used and recommended treatment option as suggested by the data. Management should be individualized to each patient, considering other comorbidities while evaluating the risks and benefits of the treatment given (medications vs surgical excision). Well-designed studies with long-term follow-ups are needed to confirm or reject causality between LEs and thromboembolic events and to establish management guidelines.
Footnotes
Author Contributions
MA wrote the first draft of the manuscript and contributed to the literature search and review. OA performed initial data collection and contributed to manuscript writing. FA contributed to critical review of the manuscript and guided all editing. All authors have reviewed and agreed on the final copy of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent
Consent has been obtained for the writing of this paper.
