Abstract
Background:
Clear cell ovarian carcinoma is rare and accounts for 1%-12% of ovarian epithelial carcinomas, depending on ethnicity. The prevalence of clear cell ovarian carcinoma in Asian, White, and Black women is 11.1%%, 4.8%, and 3.1%, respectively. Magnetic resonance imaging (MRI) shows that clear cell ovarian carcinomas are typically unilocular cyst-solid (34.9%) or multilocular-solid (41.4%); only 23.7% are solid with papillary projections. MRI can detect clear cell ovarian carcinoma with a sensitivity and specificity of 90% and 87%, respectively. Notably, sometimes ovarian masses have a solid feature and should be differentiated from uterine masses. Clear cell ovarian carcinoma has a better prognosis compared to serous carcinoma when diagnosed at an early stage, but it has a poorer prognosis at an advanced stage. The absence of a residual tumor is a favorable prognostic factor in patients with advanced-stage clear cell ovarian carcinoma. Herein, we present a case in which clear cell ovarian carcinoma was misdiagnosed as uterine sarcoma because imaging showed a mass with a solid uterine-like and necrotic area. In the present case, cytoreductive surgery was performed to remove the entire tumor and its infiltration to the sigmoid colon and left ureter. Hence, the patient had a better prognosis.
Case report:
A 57-year-old Indonesian woman presented to our hospital (Dr. Soetomo General Hospital) with post-menopausal bleeding, a large solid pelvic mass, and abdominal discomfort. The patient was diagnosed with uterine sarcoma due to the solid feature observed during ultrasonography and MRI. During the surgery, the mass was observed to originate from the left ovary, and primary debulking surgery with a multidisciplinary team was performed with zero residual tumor tissue. The tumor was histopathologically confirmed as clear cell carcinoma.
Conclusion:
MRI of clear cell ovarian carcinoma can be misdiagnosed as uterine sarcoma due to its solid feature. Additionally, the enlarged mass distorts the anatomical landmarks. Surgery with no residual tumor improves the prognosis for advanced-stage clear cell ovarian carcinoma.
Background
There are 5 types of epithelial ovarian carcinomas. They are defined according to their histopathological characteristics, including low-grade serous, high-grade serous, endometrioid, mucinous, and clear cell ovarian carcinoma. 1 Clear cell carcinoma is the rarest type and accounts for up to 12% of all ovarian carcinomas. The incidence of clear cell carcinoma depends on geographic and ethnic factors and is highest among Asian women. The prevalence of clear cell ovarian carcinoma in Asian, White, and Black women is 11.1%, 4.8%, and 3.1%, respectively.1,2 Clear cell ovarian carcinoma is usually diagnosed at an early age, given its associations with endometriosis and abdominal enlargement in the at-risk younger group.3 -5 Magnetic resonance imaging (MRI) is used to help diagnose clear cell ovarian carcinoma, with a sensitivity and specificity of 90% and 87%, respectively. 1
Surgery is the mainstay of clear cell ovarian carcinoma therapy; chemotherapy can be given as adjuvant therapy.4,6 Residual tumor tissue is an independent prognostic factor in patients with stage III-IV clear cell ovarian cancer.1 -3,5,6 Cytoreductive surgery without residual tumor improves the prognosis of advanced-stage clear cell ovarian carcinoma. Herein, we present the case of an older adult patient with advanced-staged clear cell ovarian carcinoma misdiagnosed as uterine sarcoma. However, surgery was performed with no residual gross tumor, and adjuvant chemotherapy was administered using a platinum-based agent, which prolonged the overall survival.
Case Report
A 57-year-old Indonesian woman presented to our outpatient clinic in 2018 with a 2-month history of post-menopausal bleeding, abdominal discomfort, and leg edema. Physical examination revealed an enlarged uterus. Abdominal ultrasonography was performed, and uterine myoma was suspected. Curettage was performed, and the patient was diagnosed with chronic cervicitis.
A pelvic MRI performed on July 16, 2018, revealed a solid mass measuring 5 × 4 × 4.9 cm originating from the posterior uterine body with a high suspicion of uterine sarcoma because of the heterogenous density and irregular border. Ultrasound Doppler revealed a thrombus in the left femoral vein. A thrombolytic agent was administered, and a total hysterectomy with bilateral salphingo-oophorectomy was suggested; however, the patient refused further treatment.
On October 15, 2021, the patient again presented to our outpatient clinic with an abdominal mass but without vaginal bleeding; the leg edema had resolved. Pelvic MRI results still indicated uterine sarcoma. The mass from the uterus had enlarged to 12.6 × 16.5 × 17.7 cm in size and was observed to invade more than half of the myometrial thickness on MRI. Surgery was planned, but the waiting list was long due to the coronavirus pandemic in our country until August 22, 2022.
Again, the patient presented to the emergency unit with chief complaints of vomiting and fever. Preoperative blood test results were normal; however, the cancer antigen 125 (CA 125) level was not examined. The pelvic MRI was repeated, and uterine sarcoma was suspected again (8.5 × 16.11 × 19.9 cm). The mass originated from the posterior uterine corpus, infiltrating the descending colon and causing fistulation. The mass also infiltrated the left ureter (Figure 1).

(A) MRI T2 coronal plane fat saturation; solid mass with large necrotic area originating from the left ovary (yellow arrow), fistulation between the mass and the sigmoid (red arrow), uterine (blue arrow). (B) T2 coronal plane fat saturation; dilated right ureter (yellow arrow), dilated left ureter (red arrow). (C) T2 coronal plane fat saturation; dilated right ureter (yellow arrow), infiltration mass to the left ureter (red arrow). (D) T2 axial plane; right ovary (yellow arrow), dilated right ureter (red arrow), uterine (blue arrow), the mass (green arrow), and dilated left ureter with infiltration (orange arrow).
Finally, a multidisciplinary team consisting of a gastrointestinal surgeon and urologist with experience in the diagnosis of uterine sarcoma performed the surgery. After the abdominal cavity was opened, there were large adhesions between the mass, omentum, and sigmoid colon; there were no ascites in the abdomen. The adhesions were released in collaboration with the digestive surgeon. Intraoperatively, the mass was identified to originate from the left ovary instead of the uterus. The tumor was 20 × 30 cm and had infiltrated the sigmoid colon and left ureter. The lymphatic node from pelvic to para-aortic was explored, and no enlarged lymph node was found; therefore, pelvic lymphadenectomy was not performed. The peritoneum, spleen, diaphragm, and hepatic regions were clear, with no tumor implantation. The uterus and right ovary were normal. Hence, cytoreductive surgery (total abdominal hysterectomy with bilateral salphingo-oophorectomy) was performed with no residual tumor. The gastrointestinal surgeon performed sigmoid resection and Hartman’s procedure, and the urologist performed resection of the infiltrated ureter and end-to-end anastomosis using a ureter stent. The patient was diagnosed with ovarian carcinoma after the surgery based on the gross pathology result (Figure 2).

(A) Large adhesions between the mass, omentum, and peritoneal tissue after opening the abdominal cavity. (B) The mass originates from the left ovary with a necrotic area (yellow arrow); the uterus is normal (blue arrow). (C) Fistulation between the mass from the left ovary and sigmoid (yellow arrow). (D) The mass (yellow arrow) and left ureter (blue arrow) after separation (E). Normal uterus. (F). Specimen after surgery, right adnexa (blue arrow), uterine (yellow arrow), and mass from the left ovary (red arrow).
The histopathology result was clear cell ovarian carcinoma of the left ovary with capsule infiltration. This histopathological diagnosis was made due to the presentation of uniformly atypical nuclei hobnail cells with papillary, tubular, and solid architectural patterns. However, immunohistochemistry was not performed to identify the origin since the macroscopic tumor originated from the left ovary, not the uterus. Although no tumor was found in the right ovary, uterus, or cervix, the tumor had infiltrated the muscularis propria of the sigmoid colon (Figure 3). The patient was diagnosed with stage IVB clear cell ovarian carcinoma (T1C2NxM1b) because the tumor infiltrated the sigmoid colon, and initiation of palliative platinum-based chemotherapy and taxane was planned. Carboplatin was used as a platinum agent in combination with paclitaxel for 6 cycles of chemotherapy. Intravenous paclitaxel 175 mg/m2 was administered for 3 hours, and intravenous carboplatin (with an area under curve 6) was administered for 30 minutes. Postoperatively, the CA 125, hemoglobin, and procalcitonin levels were 17.6 U/mL, 7 g/dL, and 10.3 ng/mL, respectively. The patient received a blood transfusion due to the excessive bleeding and antibiotics to prevent sepsis. She was hospitalized for 15 days before discharge, and the wound was healed. Presently, the patient has already received a second dose of chemotherapy and is doing well with a stoma on the abdominal wall.
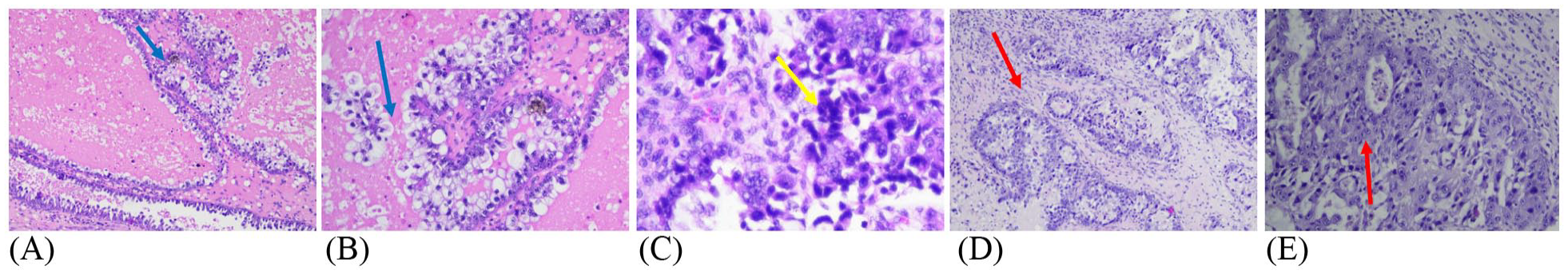
(A) Histopathological features (hematoxylin and eosin stain) with clear cytoplasm (yellow arrow). (B) Papillary tumor pattern with tubulocystic and solid appearance, anaplastic cells with prominent, oval nucleoli, pleomorphic, coarse chromatin, and clear cytoplasm suggesting clear cell carcinoma of the left ovary (blue arrow). (C) Hobnail cells (yellow arrow) (D and E) Metastatic clear cell carcinoma within the muscularis propria of the sigmoid (red arrow).
Discussion
Clear cell ovarian carcinoma was initially defined by the World Health Organization (WHO) in 1973 as having a histopathological profile characterized by clear cells growing in solid/tubular or glandular patterns as well as hobnail cells. In 2014, the WHO revised the ovarian carcinoma subtypes into 5, including (i) high-grade serous, (ii) low-grade serous, (iii) endometrioid, (iv) mucinous, and (v) clear cell carcinomas. Solid, pseudoendometrioid, and transitional cell carcinomas can be classified as high-grade serous carcinoma. Clear cell carcinoma is distinct from other ovarian cancer subtypes due to its lower response rate to chemotherapy. It accounts for 1%-12% of all epithelial ovarian carcinomas, and its incidence in Taiwan and Japan is high (13%-25%). 2
Usually, clear cell carcinoma is diagnosed in younger patients and at an early stage. Takano et al reported that the mean age for developing clear cell ovarian carcinoma is 52.4 years and is mostly diagnosed at an early stage (36% stage 1C); only 16 out of 254 cases were diagnosed as advanced stage/stage IV (6%). 4
Some gene alterations have been studied in clear cell ovarian carcinoma. AT-rich interaction domain 1A (ARDIA) and phosphatidylinositol-4,5-biphosphate-3-kinase catalytic subunit A (PIK3CA) mutations are responsible for up to 50% of clear cell carcinomas. Breast cancer 1 (BRCA1) and 2 (BRCA2) and p53 mutations are present in lower frequency in clear cell carcinoma, unlike high-grade serous carcinoma; BRCA mutations have been identified in 6% (1% germline and 5% somatic mutations) and p53 mutations in approximately 8.5%-21.6%. This distinctive molecular pathway plays a role in intrinsic chemoresistance.1,3,6
The most common risk factor for developing clear cell carcinoma is endometriosis. Zhao et al reported that clear cell carcinomas with endometriosis have a relatively good prognosis compared to that without endometriosis and are associated with (i) the absence of ascites (20% vs 52.9%), (ii) younger age (49.9 ± 6.4 years vs 5.9 ± 8.5 years), (iii) smaller tumor diameter (9.9 ± 3.7 cm vs 12.3 ± 5 cm), (iv) lower incidence of bilateral involvement (3.6% vs 14.3%), and (v) lymph node metastasis (1.8% vs 16.9%).1,7 In our case, no evidence of endometriosis was found intraoperatively. The patient was diagnosed with clear cell ovarian carcinoma at 57 years old, the tumor was unilateral, and the size was 20 × 30 cm. However, no ascites or enlarged lymph nodes were observed in this case.
Clear cell ovarian carcinoma presents with clinical features similar to other types of ovarian cancer, such as abdominal mass/distention (38.5%), abdominal or pelvic pain (33.1%), menstrual change (7.7%), and incidental findings during examination (20.7%). 5 The pathognomonic symptoms of clear cell ovarian carcinoma include paraneoplastic syndromes such as thromboembolic events, cerebral infarction, and cardiac arrhythmias. 1 In 2018, the patient presented with a complaint of leg edema. This was confirmed with Doppler ultrasound as being due to a thrombus in the left femoral vein, which is consistent with paraneoplastic syndrome. Clear cell ovarian carcinoma is usually diagnosed early; however, the patient presented in 2018 without a chief complaint of an abdominal mass. MRI showed that the mass had grown from 5 × 4 × 4.9 cm in 2018 to >30 cm in 2022. The patient’s chief complaints of vomiting and fever on presentation to the emergency unit were complications from the fistulation between the colon and the mass.
MRI can detect clear cell ovarian carcinoma with a sensitivity and specificity of 90% and 87%, respectively. 1 Clear cell carcinoma usually presents as large unilocular cysts with protrusions and an oval shape (57%). 8 The other form of clear cell carcinoma is a solid adnexal mass with a large necrotic or cystic area (43%).9 -11 Mostly clear cell ovarian carcinomas feature a large, cystic, unilateral adnexal mass; only 23% manifest as a solid mass with heterogeneous contrast enhancement in the necrotic area. Uterine sarcomas also feature a solid mass in the necrotic area but with intense contrast enhancement. 11 MRI has a higher sensitivity and specificity than ultrasound or computed tomography with an accurate mass characterization. 11 In this case, however, the patient was misdiagnosed with uterine sarcoma because the large solid mass (20 × 30 cm) detected on MRI distorted the landmark. Thus, the mass was assumed to have originated from the uterus. This confirms that clear cell ovarian carcinoma also may have solid features.
Pathology results can confirm a clear cell carcinoma diagnosis because the tumor has cystic, papillary, tubular/glandular, and solid architectural patterns with focal necrosis. Furthermore, the cells have a clear cytoplasm and uniformly atypical nuclei hobnail cells, and their nuclei are round to irregular and hyperchromatic. In comparison, high-grade serous carcinomas have solid, papillary, and glandular architecture. However, their large nuclei show a high mitotic activity (>12 mitoses per 10 high-power fields), and the nucleolus is prominent and eosinophilic. Immunohistochemistry with napsin A, Wilms Tumor-1 (WT-1), and estrogen receptor (ER) can also differentiate clear cell carcinoma from high-grade serous carcinoma. Clear cell carcinomas show a positive result for napsin A and a negative result for WT-1 and ER.
Endometrioid and clear cell carcinomas have both been associated with endometriosis, and endometrioid carcinoma has a better prognosis. The most common morphologic pattern is confluent (back to back) glands and rare stromal invasion; however, sometimes destructive stromal invasion is observed. Squamous metaplasia (morules or keratin pearls), cytoplasmic mucin, clear cell changes, and cilia and sex cord-like elements are observed when endometriosis or adenofibroma may be present in the background.1,3 In our case, endometriosis was not observed in the pathology results.
The CA 125 level in clear cell ovarian carcinoma is not as high as that in serous carcinoma; it is usually <200 U/mL, that is, within the normal limit.1,3,5,6 CA 125 level is elevated in 78.9%-87% of clear cell ovarian carcinoma cases and is associated with endometriosis, which is, in turn, considered a major risk factor for clear cell ovarian carcinoma.1,12 Our patient’s postoperative CA 125 level was normal (17.6 U/mL), although the preoperative CA 125 level was not examined because of the misdiagnosis. In our patient, no evidence of endometriosis was found intraoperatively and in the histopathology result.
Clear cell ovarian carcinoma has a better prognosis compared to serous carcinoma at an early stage but a poorer prognosis at an advanced stage.13,14 When diagnosed at stage III, the 5-year survival rate for clear cell carcinoma is lower than that of serous carcinoma (22.3% vs 47.3%). When diagnosed at stage IV, the 5-year survival rate for clear cell versus serous carcinoma is 0% versus 24.4%. However, if surgery can be performed without gross residual tumor, clear cell ovarian carcinoma would have a better 5-year survival rate than serous carcinoma (82.3% vs 75.2%). 2 The gold standard for advanced-stage clear cell ovarian carcinoma therapy is primary cytoreductive surgery followed by chemotherapy using a platinum-based agent. Since the frequency of BRCA mutation in clear cell carcinoma is low, maintenance therapy with a poly SDP-ribose polymerase inhibitor is not recommended. 1 In this case, the patient was diagnosed with clear cell ovarian carcinoma stage IVB, with no gross residual tumor, and chemotherapy had already been initiated using carboplatin and paclitaxel. In Indonesia, patients are mostly diagnosed with advanced-stage ovarian cancer (39.53%). 15 BRCA mutation was not examined in this patient due to our country’s lack of the required facility.
Conclusion
Clear cell ovarian carcinoma is a rare histopathological subtype of epithelial ovarian cancer that is typically diagnosed at an early stage. At an advanced stage, clear cell ovarian carcinoma has a poorer prognosis, but surgery without residual tumor improves the prognosis. MRI can diagnose clear cell ovarian carcinoma with a sensitivity of 90%. While most clear cell ovarian carcinomas are unilocular cysts, it is important to know that only 23% have a solid feature. In the future, the tumor origin should be differentiated more accurately when it has a solid feature to avoid a misdiagnosis. In this case, cytoreductive surgery was performed to remove the gross tumor, including its infiltration to the sigmoid colon and left ureter. Hence, the patient had a better prognosis despite a diagnosis of advanced-stage clear cell carcinoma.
Footnotes
Acknowledgements
We wish to acknowledge Editage (
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Brahmana Askandar Tjokroprawiro was taking care of the patient, collecting data, editing, formatting dan submitting the manuscript. Yoan Alexandria Angelina was making the draft of the manuscript and editing.
