Abstract
Background:
It is known that renal artery aneurysms may lead to hydronephrosis, but utter shrinking of the renal parenchyma due to a giant renal artery aneurysm has not yet been reported. This report is of an 88-year-old woman with resistant hypertension, hydronephrosis, and renal atrophy due to a giant saccular aneurysm of the left renal artery.
Case Report:
The patient presented with 2 weeks of worsening low back pain on the left side and resistant hypertension. The discovery of a left flank mass on physical examination, lead to the ultrasound detection of a para-aortic mass and a cyst-like partially septate structure replacing the left kidney. Thereafter, a contrast-enhanced computed tomography scan revealed a giant saccular aneurysm of the left renal artery causing severe hydronephrosis with severe parenchymal thinning. The extreme parenchymal atrophy of the left kidney made it an unlikely culprit of resistant hypertension, therefore the interventional radiologist considered that an endovascular attempt to re-establish the patency of the left renal artery would have scarcely produced any benefit. Given the advanced age of the patient, the vascular surgeon considered that risk of a nephrectomy outweighed the benefit, in agreement with the patient’s unwillingness to accept an invasive intervention. Therefore, she was discharged with blood pressure lowering and pain relief medication and was thereafter lost to follow-up.
Conclusion:
To our knowledge, this is the most severe kidney parenchymal shrinking in association with a renal artery aneurysm reported in the literature.
Background
A renal artery aneurysm (RAA) represents abnormal dilatation of a segment of the renal artery to more than twice the normal diameter. 1 This involves weakening of the vessel wall secondary to degenerative changes. 2 Several conditions can be related to aneurysmal development such as atherosclerosis, vasculitis, fibromuscular dysplasia, trauma or congenital defects.2,3 Most RAAs are asymptomatic and are discovered incidentally, but some of them can lead to complications prompting further investigation with the double aim of establishing whether a therapeutic intervention should be considered and of choosing among the various options. 3 We have found in literature case reports of hydronephrosis due to RAA compression: a 120 mm right RAA and a 50 mm left RAA with pelvic dilatation reported by Ozkan et al, 2 a 102 mm RAA associated with hydronephrosis described by Yang et al, 4 and a 90 mm RAA which led to hydronephrosis and renovascular hypertension reported by Bernhardt et al. 5 Cases of smaller RAAs associated with hydronephrosis were reported in literature.6,7 This report is of an 88-year-old woman with resistant hypertension, hydronephrosis, and renal atrophy due to a giant saccular aneurysm of the left renal artery.
Case Report
An 88-years-old Caucasian female with a history of arterial hypertension, osteoporosis and degenerative lumbar disk disease, on daily treatment with Indapamide 2.5 mg qd, Enalapril 10 mg bid, Isosorbide-mononitrate 40 mg qd and Alphacalcidol 0.25 μg qd presented with low back pain on the left side, which started 6 months previously and had grown worse during the 2 weeks preceding the presentation. The pain radiated down the left lower limb leading to difficulty in assuming an upright posture and in walking. The pain was partially relieved by analgesics. Every 6 months, she had a course of physiotherapy for her lumbar spine disease. She denied tobacco smoking or alcohol abuse. Physical examination revealed pale skin and mucosae, high blood pressure (190/80 mmHg), a regular heart rhythm at a rate of 90 bpm, a grade 2/6 systolic murmur at the right upper sternal border, a grade 2/6 apical systolic murmur, and bilateral carotid bruits (possibly radiation of the aortic murmur), normal breath sounds with no rales, a soft abdomen, no tenderness, but a palpable non-tender mass in the left flank and no edema. Blood tests revealed severe microcytic hypochromic anemia (hemoglobin 5.9 g/dL, mean corpuscular volume 66.4 fL, mean corpuscular hemoglobin 19.5 pg, mean corpuscular hemoglobin concentration 29.5 g/dL), low serum iron (12 µg/dL), a total iron binding capacity of 221 µg/dL (transferrin saturation 5.4%), low-normal ferritin (36.6 ng/mL), a slightly increased serum creatinine (108.76 µmol/L = 1.23 mg/dL; estimated glomerular filtration rate 39 mL/min/1.73 m²), N-terminal pro B-type natriuretic peptide (NT-proBNP) of 10 545 pg/mL suggesting volume overload. All other blood tests, including those reflecting inflammation, were within normal range.
No imaging studies had been performed prior to her current presentation, except for whole body bone scintigraphy. Consequently, abdominal ultrasound was performed and revealed a 100 mm mass in the left flank immediately adjacent to the calcified aortic wall (Figure 1A) and no obvious left kidney—instead, immediately adjacent to the mass, there was a cyst-like oblong structure of about 90 mm in length and 50 mm in width, with several septum-like structures projecting inwardly over a distance of about 1 cm from the wall of the cyst (Figure 1B). Some color doppler signal was evident inside the 100 mm-mass, with unclear significance. A contrast-enhanced thoracic and abdominal computed tomography (CT) showed a normal right kidney, but for a left lower polar cortical cyst of 27 mm, and a giant (transaxial diameters of 109 × 102 mm, cranio-caudal diameter of 94 mm), saccular, almost completely thrombosed aneurysm of the left renal artery with discrete parietal calcifications, a 13 × 9 mm nodular calcification at the superior-posterior pole (Figure 2A), and no active contrast media extravasation in the retroperitoneum. The giant aneurysm compressed the left kidney pelvis causing severe hydronephrosis with extreme thinning of the renal parenchyma (to a thickness of 2 mm) and of renal Bertin columns which appeared as thin septa-like structures (Figure 2B and C). On abdominal ultrasound, severe shrinking of kidney parenchyma was associated with the reduction to a minimum of the blood flow to the doomed kidney and power doppler detected no blood flow to the residual rim of kidney tissue. CT angiography failed to improve visualization as the aneurysm was almost completely filled with thrombotic material leaving but scanty room for contrast penetration. Consequently, the outpouching that holds the place for the aneurysm on CT angiography appears quite distant from the aorta (Figure 3), although the wall of the thrombosed aneurysm was in contact with the aortic wall, as demonstrated by both ultrasound (Figure 1A) and CT (Figure 2D). CT scan also revealed degenerative changes in the spine with diffuse bone demineralization, reduced vertebral body height of the second lumbar vertebra and body compression fracture of the third lumbar vertebra.

Left renal artery aneurysm on abdominal ultrasound (with the transducer positioned in the left flank) (A) It is showed that the heterogeneous mass (open arrows) is tangent to the calcified aortic wall (white arrowheads). Contributing to the heterogeneity of the mass are both areas of increased echogenicity (thick white arrow) reflecting a higher degree of fibrous organization and echo-free areas representing persistent blood flow (white star). The slim white arrow points to the lumen of the aorta (B) It is showed a mass with heterogeneous structure (open arrows) and, adjacent to it, a cystic mass (slim white arrows) with several septum-like structures projecting inwardly (white thick short arrows). White arrowheads point to the boundary between the 2 structures. There is a calcification throwing an acoustic shadow inside the mass (thick white arrow).
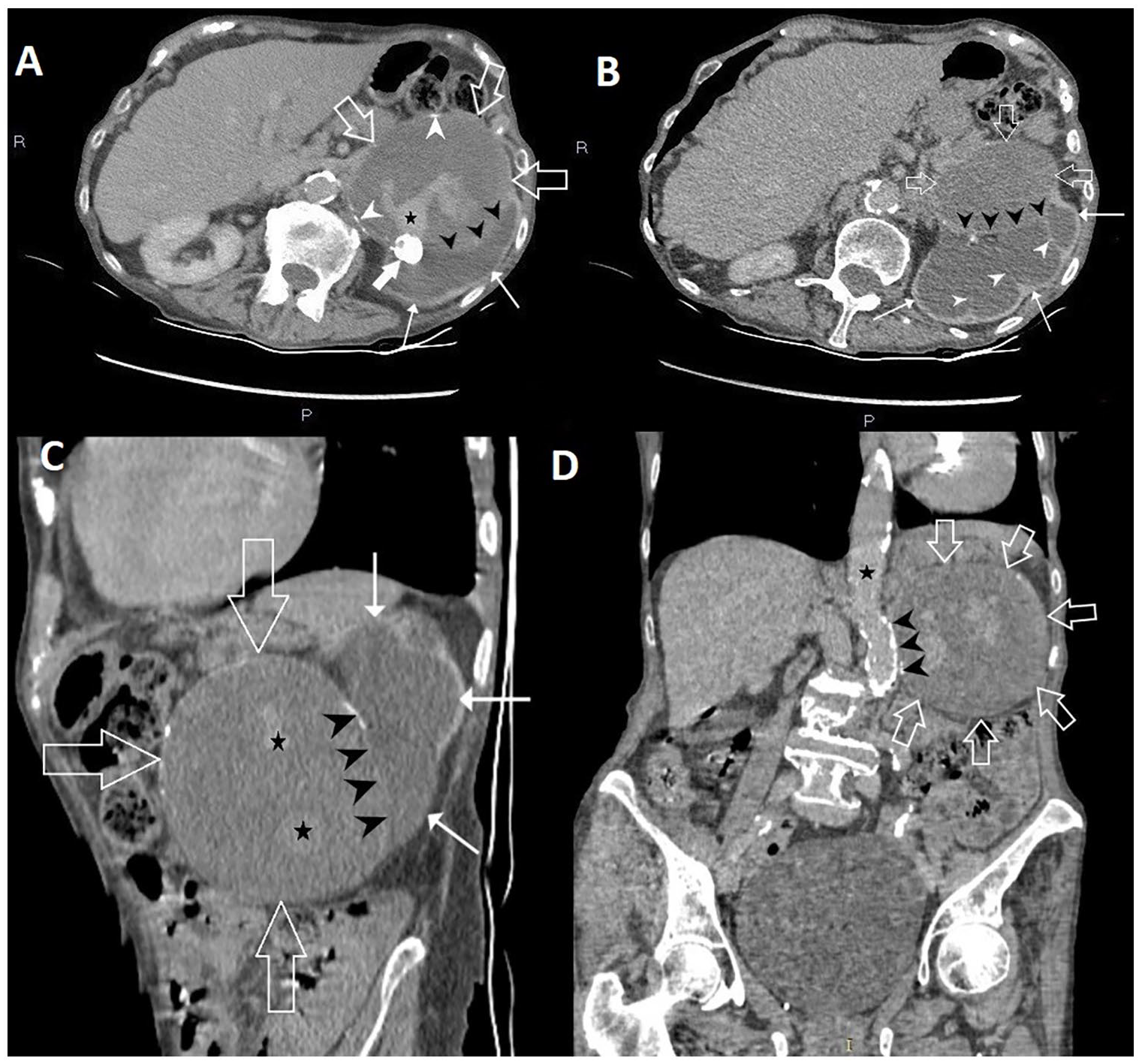
Giant left renal artery aneurysm on contrast-enhanced computed tomography—axial section through upper abdomen (A and B), sagittal reconstruction (C) and coronal reconstruction (D) A. It is showed a giant saccular almost completely thrombosed aneurysm of the left renal artery (open arrows) with discrete parietal calcifications (white arrowheads). There is a nodular calcification at the superior posterior pole of the aneurysm (white thick arrow). Persistent blood flow inside the aneurysm is marked by a black star. Black arrowheads point to the boundary between the giant aneurysm and hydronephrotic left kidney (slim white arrows) B. It is showed the thrombosed aneurysm of the left renal artery (open white arrows), the severely hydronephrotic left kidney with extreme parenchymal atrophy (slim long white arrows) in which the atrophied renal (Bertin) columns are still visible (white arrowhead). The border between the aneurysm and the hydronephrotic left kidney is indicated by black arrowheads C. The relationship between the thrombosed aneurysm (open arrows) and the hydronephrotic kidney (slim white arrows) is clearly visible. Black arrowheads point to the border between the 2 structures. Black stars indicate areas of blood flow (D) The relationship between the thrombosed aneurysm (open arrows) and the aorta (black star) is clearly visible. Black arrowheads point to the border between the 2 structures, demonstrating that the thrombosed aneurysm is in close contact with the calcified aortic wall.

A portion of the aneurysm seen on computed tomography angiography. Only a small portion of the aneurysm is visualized (thin white arrow) (as most of the volume of the aneurysm is occupied by thrombotic material)—consequently there appears to be a large distance separating the “aneurysm” from the aorta (although computed tomography and ultrasound clearly demonstrated that the aneurysm borders medially on the aorta—see Figures 1A and 2D). Some diffusely delineated “vascular” trajectories are visible in the intervening space (arrowheads). Left kidney (thick white arrows) is visible behind the aneurysm.
Next, the patient was evaluated by an interventional radiologist and a vascular surgeon. The former decided that the patient had no indication for endovascular treatment, taking into account the size of the aneurysm and the advanced stage of kidney parenchymal disease, while the latter considered that the patient was at a major surgical risk if subjected to nephrectomy. The patient agreed with this choice, declaring that she would not have accepted any kind of intervention. Her only wish was to be given some medication to alleviate her pain.
During her hospital stay, adequate blood pressure control was achieved with difficulty, requiring an angiotensin-converting-enzyme inhibitor (ACE-I) (Perindopril 10 mg), a calcium channel blocker (Amlodipine 10 mg), an imidazoline receptor agonist (Moxonidine 0.4 mg), a beta blocker (Bisoprolol 10 mg), and a thiazide-like diuretic (Indapamide 2.5 mg). A decrease in potassium level, from a normal value of 3.6 mmol/L at admission, to 3.16-3.3-3.2-3.3-3.74-2.7-3.05 mmol/L in blood samples taken at 1 to 2 days intervals, was noticed for which the only explanation seemed to be Indapamide administration, which made us to suspect that at home the patient took Indapamide intermittently if at all. Consequently, a potassium sparing diuretic (Spironolactone 25 mg) was added. The decision to associate the 2 diuretics was also supported by the increased NT-proBNP level suggesting volume overload and, consequently, the necessity for a more aggressive volume-controlling medication. Serum potassium level at discharge was almost normal (3.47 mmol/L), corroborating the usefulness of Spironolactone as an adjunct to the thiazide-like diuretic. Given the severity of her anemia the patient was transfused with 1 unit of packed red blood cells. An iron deficiency state was suspected based on her low transferrin saturation, although her ferritin was above the 30 μg/L threshold. 8 Coupled with the low red blood cell indices, this made an iron deficiency anemia very probable, prompting the administration of a challenge dose of iron sucrose (iron(III)-hydroxide sucrose complex).
At discharge, the patient was instructed to continue the blood pressure lowering medication given during her hospital stay and to check creatinine, sodium, and potassium level. A follow-up visit after 1 month was arranged but the patient failed to comply, and she was lost to follow-up. We were unable to reach her by phone.
Discussion
The information used in this section was searched in PubMed and ScienceDirect using as retrieval formula “renal artery” AND aneurysm AND hydronephrosis, which yielded 44 articles on PubMed, all relevant to our subject, but 1260 on ScienceDirect, the vast majority of which had no bearing on our subject. Therefore, we limited our references to those found on PubMed.
Above and beyond guidelines recommendations, the clinical judgment of the practitioner should appraise each case in all its particular features, including risk factors, expected survival, general biological status, the patient’s wishes and, the “do not harm” imperative, in order to reach the optimal decision. It is difficult to decide how the guidelines should be applied in extreme cases, and our case was a confluence of extremes: the age of the patient, the dimensions of the RAA, the severity of hydronephrosis, the thinning of renal parenchyma. There are similar cases in the literature, but none with such severe damage to renal parenchyma.4,5,9,10 Moreover, hydronephrosis caused by RAA is rare—a survey of cases reported between 1968 and 2001, performed by Miyagawa et al, identified only 16 cases of RAA-induced hydronephrosis. 6
The incidence of RAA is estimated to between 0.01% and 1%. 2 In a study conducted on 862 patients using contrast-enhanced abdominal CT scanning, the prevalence of RAA was 0.7%. Nonetheless, much higher estimates (9.7%) were provided by autopsy studies.11,12 The size of RAA varies. Those larger than 50 mm, are considered giant.13,14 We could not find the prevalence of giant RAA. Only several cases have been reported in the literature.13,14 There are cases of thrombosed RAAs reported in the literature,15 -19 but we couldn’t find any estimates of the prevalence/incidence of RAA thrombosis; however, distal parenchymal embolization presumably originating in the RAA was noticed in 8% to 11% of RAA patients. 20 Parietal calcifications are present in up to 40% of RAAs, but we could find no article reporting calcification(s) inside the RAA. 21 In a study conducted on 760 patients with a total of 865 RAAs, 56% of RAAs were calcified (nothing is said about the location of the calcifications) and a mural thrombus was present in 11%. 22 In most cases, RAA are asymptomatic and are discovered incidentally. 3 Occasionally, they become symptomatic as they increase in size leading to abdominal pain, renovascular hypertension, hematuria, renal artery thrombosis, arteriovenous fistula, and, the most dreaded complication, rupture.6,13 Compression of the pelvicalyceal system or ureter may result in hydronephrosis. 6
In the present case, the patient had low back pain on the left side, but this was most likely due to degenerative lumbar disk disease because the pain radiated down the left lower limb leading to functional disability. The difficulty in achieving adequate blood pressure control raised the suspicion of secondary hypertension.
Regarding the hypokalemia the patient developed during her hospital stay, it should be noticed that serum potassium level was normal at admission, therefore something that happened during her hospital stay must have been responsible for her hypokalemia. High diuresis could hardly be invoked as an explanation, as urine output was normal and no attempt was made to increase it by intravenous fluids. The only potassium-lowering pharmacological agent the patient received was Indapamide. We could not afford giving up its blood pressure lowering effect as we were facing a case of resistant hypertension and it is known that at least one diuretic agent is mandatory in the management of refractory hypertension with signs of volume overload 23 (which was indicated in our case by the high NT-proBNP level). The association of a potassium-wasting (such as Indapamide) with a potassium-sparing diuretic (such as Spironolactone) is highly recommended. 24 Spironolactone treatment in patients with impaired kidney function may result in hyperkalemia, but mineralocorticoid receptor antagonists are not only admitted as long as serum potassium level is <5 mmol/L, serum creatinine level is <2.5 mg/dL (221 μmol/L), and estimated glomerular filtration rate is >30 mL/min/1.73 m2, 25 but also recommended in cases of resistant hypertension given their ability to reduce cardiovascular morbidity and mortality and to slow down the progression both of kidney function decline and of cardiac and vascular remodeling.26 -29 In our patient, the creatinine level remained stable, and the patient was discharged with a serum creatinine of 1.23 mg/dL, slightly lower than the serum creatinine at admission (1.49 mg/dL). Given the risk of hyperkalemia (due to Spironolactone), as well as of hyponatremia (besides hypokalemia, due to Indapamide) the patient was instructed to periodically check her serum potassium, creatinine, and sodium levels.
In RAA, renovascular hypertension may be the consequence of altered geometry of the renal artery resulting in diminished kidney perfusion, 30 of renal artery stenosis associated to the RAA, 13 or of activation of the renin-angiotensin system induced by microembolism originating in the thrombus inside the aneurysm. 31 Nevertheless, the extreme degree of parenchymal thinning in our case, with presumable utter destruction of functional parenchyma (including renin secreting cells) argues against the left kidney as the culprit for the high blood pressure. The differential renal venous renin activity assessment might have been useful for settling this issue, but the high rate of both false-positives and false-negatives makes this approach of limited value. 32 The presumed atrophy of kidney parenchyma was probably the consequence not only of severe hydronephrosis, but also of reduced blood flow to the kidney due to aneurysm thrombosis. It is an established fact, supported by both animal experiments 33 and reported series of clinical cases,34,35 that hydronephrosis may induce secondary hypertension due to the imbalance between the vasoconstrictors (sympathetic tone, renin, thromboxane A2, prostaglandin F2α) and vasodilators (such as vasodilatory neutral lipids). 35 However, in the clinical cases series nothing is said about the severity of parenchymal shrinking in the affected kidney, therefore the conclusions of these studies cannot be extrapolated to our case.
Regarding the causes of the RAA, atherosclerosis seems the most probable in this case. 2 Atherosclerotic lesions have been demonstrated in this patient involving thoracic aortic, and supra-aortic and coronary arteries. Hence, it is conceivable that such lesions might have affected the left renal artery too, contributing to the development of the aneurysm, one further argument being the calcifications in the wall of the aneurysm. Ascending aortic ectasia demonstrated by CT was also presumably the consequence of systemic atherosclerosis. Other causes of RAA are less likely. Vasculitis was an improbable cause as the patient showed no clinical or laboratory sign of systemic inflammation and physical examination revealed no signs of vasculitis.2,36 -39 Fibromuscular dysplasia was an unlikely diagnosis given the advanced age of the patient. 40 Traumatic and iatrogenic causes such as biopsy or nephrostomy were also very improbable, as there was no trauma and no kidney puncture in the patient’s history. 13
Management of RAA depends on the size, anatomic localization, type/shape (saccular or fusiform), and complexity of the aneurysm, and on the existence, nature, and severity of the symptoms and comorbidities2,13,41 and includes open surgery (either resolving the aneurysm by plication, ligation or resection, with preservation of arterial continuity13,42,43 or nephrectomy13,44) or an endovascular procedure (stent-grafting or embolization 12 ).
An endovascular approach was deemed inappropriate in our patient because it was highly unlikely that the patient would have derived any benefit therefrom. The endovascular treatment has 2 main objectives: the occlusion/exclusion of the aneurysm and the reestablishment of the vascular patency in order to salvage the kidney.12,45 In the case of our patient, the aneurysm was already almost occluded by thrombosis and there was practically no left kidney parenchyma left to be salvaged. The risk of rupture was relatively low in this patient, as the aneurysm was almost completely thrombosed. 46
The cases most similar to ours that we could find in literature are summarized in Table 1.
Case reports of renal artery aneurysm causing hydronephrosis similar to ours..

Abbreviations: publ., publication; Ref., Reference; yrs., years.
Not only objective factors (age, general health state, expected survival), but also subjective ones (mainly the patient’s choice and desire) need to be considered when making decision in a case of RAA, or in every other medical condition for that matter. The advanced age of our patient put her at a high surgical risk for a nephrectomy, outweighing the doubtful benefit in terms of hypertension control. Therefore, the patient was managed conservatively with medication that efficiently lowered blood pressure and controlled pain.
Conclusion
What is distinctive in the present case is the extreme hydronephrosis leading to utter parenchymal thinning of the affected kidney due to the compression exerted by a large RAA—to our knowledge, this is the most severe kidney parenchymal shrinking in association with a RAA reported in the literature. The presence of a calcification inside the thrombosed aneurysm is another feature distinguishing our case from the other reported cases of RAA.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Maria Iuliana Ghenu, Francisc Iohann Bach, Maria Mirabela Manea, Dorin Ionescu and Dorin Dragoş made a significant contribution to the work reported. All authors have equally contributed to the conception of this paper and should be considered first authors.
