Abstract
Kwashiorkor is a serious nutritional disease. The 7-month-old male patient presented with severe metabolic acidosis and elevated liver enzymes. His condition was similar to fatty acid oxidation defect. He was taken to the hospital on the previous day with complaints of poor sucking and difficulty breathing. Treatment of upper respiratory tract infection was provided and then he returned to his home. He was cyanotic in bed after 12 hours. The patient, who was taken to the hospital unconscious and not fed for 12 hours, was not given any food orally in the first health center. Until laboratory tests are done, only iv electrolyte and a fluid containing dextrose were given. When the laboratory results were found to be significantly pathological, he was urgently referred to our hospital 4 hours after the admission. The content of the iv treatment applied at the time of referral was not clearly written. Low electrolytes, uric acid, liver enzymes, urea and creatinine elevation were detected at an outer center. Ketosis, lactic acidosis and dibasic aciduria were detected in urine organic acid analysis. He was diagnosed with Kwashiorkor and refeeding syndrome according to the clinical and laboratory findings. His complaints improved with appropriate treatment.
Introduction
Kwashiorkor is a life-threatening nutritional disease that causes multiple organ failure in patients who have been eating insufficient protein for a long time.1-4 Mistakes in the treatment of patients with severe malnutrition can lead to multiple organ failure. The malnourished patient's body tries to achieve metabolic balance in order to maintain the intracellular and extracellular balance. Phosphorus, magnesium, potassium are leak out of the cell if they are insufficiently taken with nutrients in order to maintain the necessary processes for the continuation of life. However, when high energy are given with re-feeding, insulin, an anabolic hormone, rises rapidly and quickly moves electrolytes to the intracellular space in order to maintain the balance. At this stage, magnesium, potassium, especially phosphorus, decreases rapidly in extracellular fluids in the treatment. In addition, water-soluble B group vitamins, especially thiamine, begin to show clinical signs of deficiency rapidly. The findings of thiamine deficiency cause disruption in the functions of mitochondrial enzymes, of which it is a cofactor. This clinical condition, known as refeeding syndrome, can be confounded with metabolic disorders due to the resulting secondary mitochondrial insufficiency.5-7 We decided to present this case report due to a patient who was referred to our department suggesting fatty acid oxidation defect due to severe metabolic acidosis and elevated liver enzymes.
Case Report
Outpatient treatment was given for upper respiratory tract infection in the hospital that the patient applied with complaints of bruising, inability to suck and breathing difficulty. The 7-month-old male patient was sent home 12 hours after the first hospital he applied to. Later, he was found in his bed in a perioral cyanotic state and was taken to the same center. Severe metabolic acidosis was observed and he was referred to our hospital with a preliminary diagnosis of metabolic disorder upon elevation of liver enzymes. Since it was presented as a rapidly deteriorating clinical picture, the patient was accepted with a preliminary diagnosis of fatty acid oxidation disorder or mitochondrial disease until his background information was learned.
We learned that his mother abandoned him when he was 1.5 months old and his grandmother and his uncle’s wife had fed him with low protein foods for 5.5 months. His movements slowed down and his legs became swollen in the last 3 months. His hair was sparse, dry and red. Infected wounds in his mouth and edema in his legs were present. The patient had a confused state but he was responding to painful stimuli. The abdominal examination revealed 2 cm hepatomegaly and a dimple in the sacral region as the pathological findings (see Figures 1 and 2).
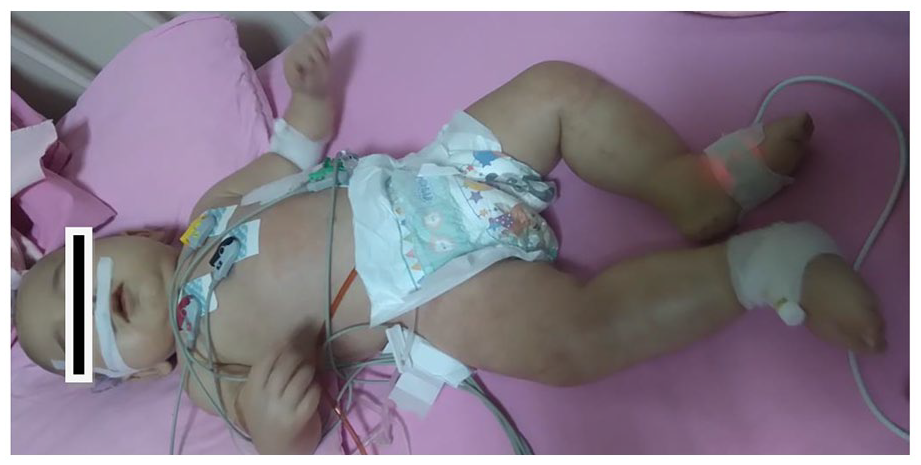
The appearance of patient’s edematous legs before treatment.
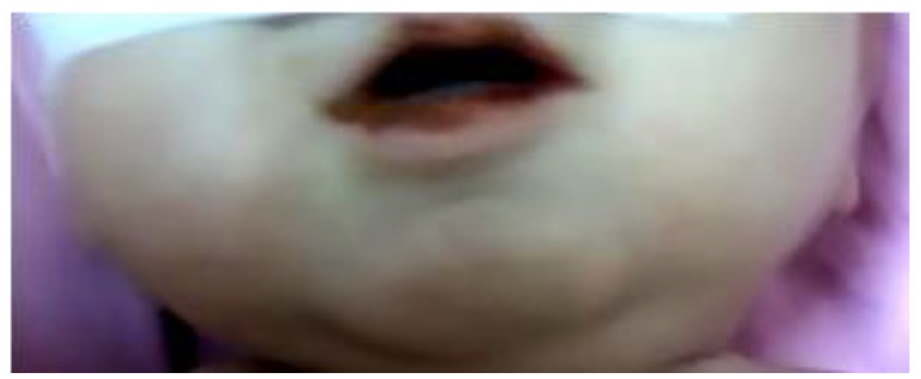
Patient’s oral findings before treatment.
Our patient was fed with 0.7 g/kg/day protein, 33 to 50 kcal/kg/day energy with excessive juicy food and irregularly used rice starch for over 5 months. On admission, the patient’s body measurements were determined to be 6 kg (<3 p) weight, 64 cm (<3 p) height, and 43 cm head circumference (3 p). Acutely mild, chronically moderate malnutrition (weight for height 89%, height by age: 88%, weight z score −2.4, height z score −3.17, and head circumference z score −0.4) was also observed. He had hypoalbuminemia consistent with his nutritional history and examination results.
The patient, who was taken to the hospital unconscious and not fed for 12 hours, was not given any food orally in the first health center. Until laboratory tests are done, only iv electrolyte and a fluid containing dextrose were given. When the laboratory results were found to be significantly pathological, he was urgently referred to our hospital 4 hours after the admission. The content of the iv treatment applied at the time of referral was not clearly written. Phosphorus, potassium, magnesium were low; uric acid, liver enzymes and creatine were high. Lactate in urinary organic acid analysis revealed: 1994 (33-185) mg/g creatine; pyruvate: 233 (5.1-22.6) mg/g creatine; 3OH butyric acid: 1175 (0.1-11.1) mg/g creatine; 2OHIVA: 54 (0-1.3) mg g creatine; 3OH IVA: 137 (10.4-67) mg/g creatine; Adipic acid: 1227 (<34.3) mg/g creatine; Suberic acid: 451 (<10.1) mg/g creatine; 4OH phenyl lactic acid: 1518 (> 3.1) mg/g creatine; 4 OH phenyl pyruvic acid: 188.5 mg/g creatine (<0.4).We observed that excretions were present at high values.
We thought that the patient had Kwashiorkor and refeeding syndrome, and we monitored the fluid he received and excreted. Cardiac monitoring was initiated. Enteral parenteral total fluid intake was restricted and electrolytes with low levels were replaced. Treatment was initiated with 10 mg/kg/day allopurinol for uric acid, low calories (40 kcal/kg/day) and fully hydrolyzed medical foods. Fully hydrolyzed medicinal food was given drop by drop with an enteral pump, starting at low concentration and increasing to full concentration in 2 weeks with electrolyte controls. Energy and fluid amount in nutrition has been gradually increased. Phosphorus, magnesium, potassium supplements were given intravenously according to daily controls. Thiamine was given parenterally at a dose of 100 mg/day for the first 10 days until the blood lactate level was within normal limits. The patient’s clinical and laboratory findings improved within 2 week (see Figure 3). Outpatient control was requested after 2 and 4 weeks following the discharge. Laboratory results are presented in Table 1. All pathological clinical findings of our patient had improved as seen in Figure 4 at the outpatient clinic follow-up 1 month later. Consent of the family was obtained for publication.

Almost completely improved post-treatment skin and neurological findings and the tone.

Appearance of the patient at the outpatient follow-up after 1 month.
Laboratory results of our patient before and after treatment.

Discussion
In 1970s, physicians were considering nutritional problems especially the clinical findings related with dilution errors in powdered infant formulas for the forefront of differential diagnosis.8-12 Over time, nutritional problems started to fall behind in priority ranking depending on the societies in which families live. Nutritional errors do not come to mind as the source of the problem thinking that the babies are breastfed before any supplementation with additional nutrients. The condition was similar in our patient. In addition, the time that the physician allocates decreases if the number of patients increases. Therefore, taking a complete medical history and performing a detailed physical examination can be skipped sometimes. According to the narrow differential diagnosis range regarding the symptoms, approaches of requesting an examination and arranging temporary treatment cause aggravation rather than solution to the problems.
In particular, water-soluble vitamin deficiency related findings were seen in the oral mucosa and skin. Fatty liver was seen on abdominal ultrasound and liver enzymes were high in blood tests as the signs of long-term low protein and high-carbohydrate diet. We initiated high-calorie intravenous fluid since we considered a metabolic disorder. As a result, the refeeding syndrome was triggered. The muscle enzymes increased when the electrolytes in extracellular fluids entered the cell by the aid of insulin and creatine phosphate reserve was used to compensate phosphorus deficiency. Tissue damage caused uric acid elevation. Thiamine deficiency among group B vitamins reminded secondary mitochondrial deficiency findings and lactic acidosis.5-7,13 During this period, when the metabolic balance turned upside down and our colleagues made an instant evaluation directing the patient to the metabolic diseases department with a pre-diagnosis of metabolic disorder due to severe metabolic acidosis lactate elevation, hepatomegaly high levels of muscle enzymes and uric acid were persevering.
It has been proposed that Kwashiorkor develops due to insufficient high quality protein consumption, and a great focus about its symptoms and physiopathology has been present in the literature for half a century. Researchers have searched for solutions to understand the underlying pathology by investigating the affected tissues via both the clinical researches performed on patients with the diagnosis and the experimentally created, similar disease settings. 14
In addition, iatrogenic Kwashiorkor developed in protein-restricted amino acid metabolism disorders have provided significant improvements in diagnosis and treatment. According to the first theories of researchers focusing the macromolecular level, edema could develop due to the lack of oncotic pressure, and fatty liver and skin findings could occur due to the vitamin, mineral and trace element deficiencies, especially the group B vitamins.1-7,14
More detailed studies on cellular level suggested that the condition could be explained in several different ways such as: molecular disorders due to deficiency of single carbon transfer because of methionine deficiency or cellular damage due to the increased oxidative stress in the cell arising from glutathione deficiency which depends on insufficient sulfur-containing amino acid consumption.4,13 It has also been shown that tissue and organ damages are inevitable if the dietary problem lasts longer.
Although researchers tried to explain the unexpected, unsuccessful results and the increased rate of life-threating situations by immune pathological mechanisms in patients with severe malnutrition especially on the beginning of treatment, studies and increasing information in the 1940s has shown that lost lives, mostly due to refeeding, are not in line with the body’s tolerance.1-7,10 Secondary mitochondrial insufficiency due to thiamine deficiency as in our patient might complicate the clinical picture by causing serious metabolic acidosis which may mimic metabolic diseases.
In addition, phosphorus deficiency causes an increase in creatine phosphokinase as a result of use of muscle creatine phosphate as a backup source. However, Kwashiorkor and refeeding syndromes can still be missed and current or rare diseases may be the first to come to mind despite the studies and the information accumulated for more than forty years.5-7
Metabolic disorders are less common than nutritional diseases. We do not underestimate the diagnosis of rare diseases but would like to note that physicians have started to miss the diagnosis of common diseases as they do not follow the classical medical diagnostic routine. If the preventive medicine system regains its former importance, more successful fight against malnutrition and related complications will be possible.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
