Abstract
Cerebral vasculitis is a very rare extra-articular complication of rheumatoid arthritis (RA) that is often challenging to diagnose. Elevated titers of rheumatoid factor (RF), anti-cyclic citrullinated peptide antibodies (anti-CCP), and antinuclear antibodies (ANA) have been linked with severe complications. The absence of highly elevated titers of RF, anti-CCP, and ANA can complicate the diagnosis of RA-associated cerebral vasculitis. We report the case of a 59-year-old woman with long-standing arthritis maintained on rituximab and leflunomide who developed sudden headaches and altered level of consciousness. Laboratory work-up revealed normal lymphocyte count and mildly elevated total serum protein and anti-CCP with negative RF and ANA and no evidence for viral or bacterial infections. Cerebrospinal fluid analysis (CSF) showed slightly elevated anti-CCP with normal levels of CXCL-13 and interleukin 6 (IL-6). Brain magnetic resonance imaging (MRI) showed ill-defined lesion of high T2 signal. Using MR angiogram, MR perfusion, and MR spectroscopy, the diagnosis of rheumatoid cerebral vasculitis was confirmed. The patient was treated with intravenous methyl-prednisolone with fast complete improvement. We conclude that adequate immunosuppression in RA might not be able to prevent rare extra-articular manifestations such as rheumatoid cerebral vasculitis.
Introduction
Rheumatoid arthritis (RA) is a systemic inflammatory disease that can affect several extra-articular targets, including the central and peripheral nervous system. 1 Neurological manifestations are unusual and usually associated with the severity of the disease. 1 These include meningitis, vasculitis, and the development of CNS rheumatoid nodules. 2 Even in the presence of a quiescent disease, neurological manifestations, such as rheumatoid meningitis, can take place. 3
In this report, we describe the case of a patient with long standing RA well-maintained on rituximab and leflunomide who presented with non-specific neurological symptoms. Laboratory work-up was negative except for moderately elevated total serum protein and slightly positive anti-cyclic citrullinated peptide antibodies (anti-CCP). Work-up for systemic infectious and inflammatory processes was negative. Neuroimaging findings were consistent with cerebral vasculitis. A rapid response to pulse therapy with intravenous methyl-prednisolone was noted.
Case Presentation
A 59-year-old woman presented with worsening headaches and dizziness of 3 day duration. She also reported feeling imbalanced, gait disturbances, and short-term memory loss. The patient complained of episodes of palpitations and hyperventilation suggestive of panic attacks, along with worsening mood.
She was diagnosed with seropositive RA 7 years prior to presentation when she presented with pain and swelling of her bilateral metacarpophalangeal joints. At the time of diagnosis, she had negative rheumatoid factor (RF) with strongly positive anti-CCP. The immunoglobulin levels were not quantified.
At the time of presentation, her RA was in remission on rituximab (2 g per 6 months for 4 years; last dose was 2 months prior to presentation) and leflunomide 20 mg daily. She had suboptimal response to hydroxychloroquine, methotrexate as well and leflunomide as monotherapy. She also had hypothyroidism treated with levothyroxine. The patient was a non-smoker.
Physical examination showed no evidence of any synovitis in her peripheral joints. She was not febrile with regular apical pulse. Neurological examination revealed no focal deficits. A Montreal Cognitive assessment (MoCA) was performed and she scored 27/30 (within normal limit), losing points on visuospatial/executive function, attention, and language.
Investigations showed a hemoglobin of 12.1 g/dL, hematocrit of 37%, white blood cell of 5500/cu.mm with 63% neutrophils and 29% lymphocytes, and platelet count of 234 000/cu.mm. The erythrocyte sedimentation rate was slightly elevated at 27 mm/hour, while C-reactive protein was normal at 0.9 mg/L. The patient had normal liver and renal function, including urinalysis. Her total serum protein was slightly elevated at 88 g/L (Albumin 50 g/L and globulin 38 g/L). Her urine was clear. Her antinuclear antibodies (ANA), RF, anti-double stranded DNA (anti-dsDNA), interleukin-2 receptor (ILR-2), matrix metalloproteinase-3 (MMP-3), anticardiolipin antibodies (aCL) and lupus anticoagulant (LAC) were negative with normal levels of complements C3 and C4. Anti-CCP level was slightly positive at 9.1 U/mL (normal range <5 U/mL). Cortisol levels and thyroid functional tests were also normal.
A computed tomography (CT) scan of the brain with contrast showed bilateral temporal and insular hypodensities that could be ischemic in nature. A brain magnetic resonance imaging (MRI) with and without gadolinium showed ill-defined lesion of high T2 signal involving the external capsule bilaterally (Figure 1A and B). A contrast enhanced MRI T1 weighted sequence showed no significant enhancement of the lesion (Figure 1C), while the apparent diffusion coefficient (ADC) showed a mild increased water diffusivity (Figure 1D and E). On MR perfusion, the cerebral blood volume was not increased within the lesion. MR angiogram of the intracranial vessels showed paucity of the distal branches of the left middle cerebral artery (MCA) (Figure 2), while the reconstructed MR perfusion showed a decreased cerebral blood flow (Figure 3). On MR spectroscopy there was no increase in the choline peak but decrease in the N-acetylaspartate (NAA) peak (Figure 4). The findings were suggestive of nonneoplastic process, ischemic lesions secondary to RA or treatment related changes.

(A) Axial view of brain MRI showing ill-defined lesion of high T2 signal in the in the white matter adjacent to insular cortex (White arrow) containing tiny foci of enhancement, and to a lesser degree on the left (Black arrow). (B) Coronal FLAIR image of the brain demonstrates high FLAIR signal lesion in the left external capsule extending inferiorly into the left temporal lobe. (C) Axial view of a brain diffusion-weighted imaging showing normal findings. (D) The apparent diffusion coefficient map showing mild increased water diffusivity. (E) Axial view of a brain T1 weighted MRI image showing no significant enhancement of the lesion.

3D time of flight MRA of the intracranial vessels demonstrates paucity of the distal branches of the left middle cerebral artery and mild irregularity along the M1 segment of the left MCA (White arrow).

Reconstructed MR perfusion image showing decreasing cerebral blood flow.

Single voxel MR spectroscopy placed within the lesion shows no increase in the choline peak but decrease in the NAA peak denoting neuronal loss.
Electroencephalogram was within normal limits for age. Lumbar puncture with cerebrospinal fluid (CSF) analysis revealed increased white blood cell count of 16 cells/mm3 with evidence of pleocytosis (92% lymphocytes) and increased protein level (0.58 g/L). The IgG index was 0.55 with absence of oligoclonal bands. CSF RF was negative at 8 IU/mL, CSF anti-CCP was slightly elevated at14 IU/mL, CSF CXCL-13 was negative at 3 ng/L, and CSF IL-6 was negative at 8.9 pg/mL. A CSF PCR meningitis panel was negative for common viruses and bacteria causing meningitis. Tuberculosis PCR and measurement of Brucella direct and indirect antibodies in CSF were also negative. A paraneoplastic autoantibody evaluation testing came back negative.
Consequently, the patient was started on 1 g of methyl-prednisolone intravenously once daily for 3 consecutive days with dramatic improvement. Afterwards, she was discharged on oral prednisone 30 mg gradually tapered by 5 mg every 5 days. Prednisone was stopped after 2 months. Upon follow-up 14 months later, the patient was still neurologically stable. She was continued on rituximab 2 g every 6 months and leflunomide 20 mg one tablet daily. No follow-up scans were performed.
Discussion
Cerebral vasculitis, one of the rarest extra-articular complications of RA, is observed in patients with long standing uncontrolled seropositive disease. 4 Most cases of cerebral vasculitis secondary to RA are associated with positive RF and ANA (Table 1). Anti-CCP has been very rarely assessed, being positive in all 3 cases that reported it.5,6 Interestingly, our case reports the lowest titer of anti-CCP in rheumatoid cerebral vasculitis. Table 1 highlights the assessment, treatment, and outcome of RA-associated vasculitis cases that have been reported in the literature so far. In our case, the patient was immunosuppressed by rituximab and leflunomide. Although an adequate immunosuppression was apparent by negative biomarkers and absence of extra-articular manifestations, the immunosuppression was not adequate enough to prevent an episode of CNS vasculitis.
Literature case reports which illustrated cerebral vasculitis as a complication of Rheumatoid Arthritis.
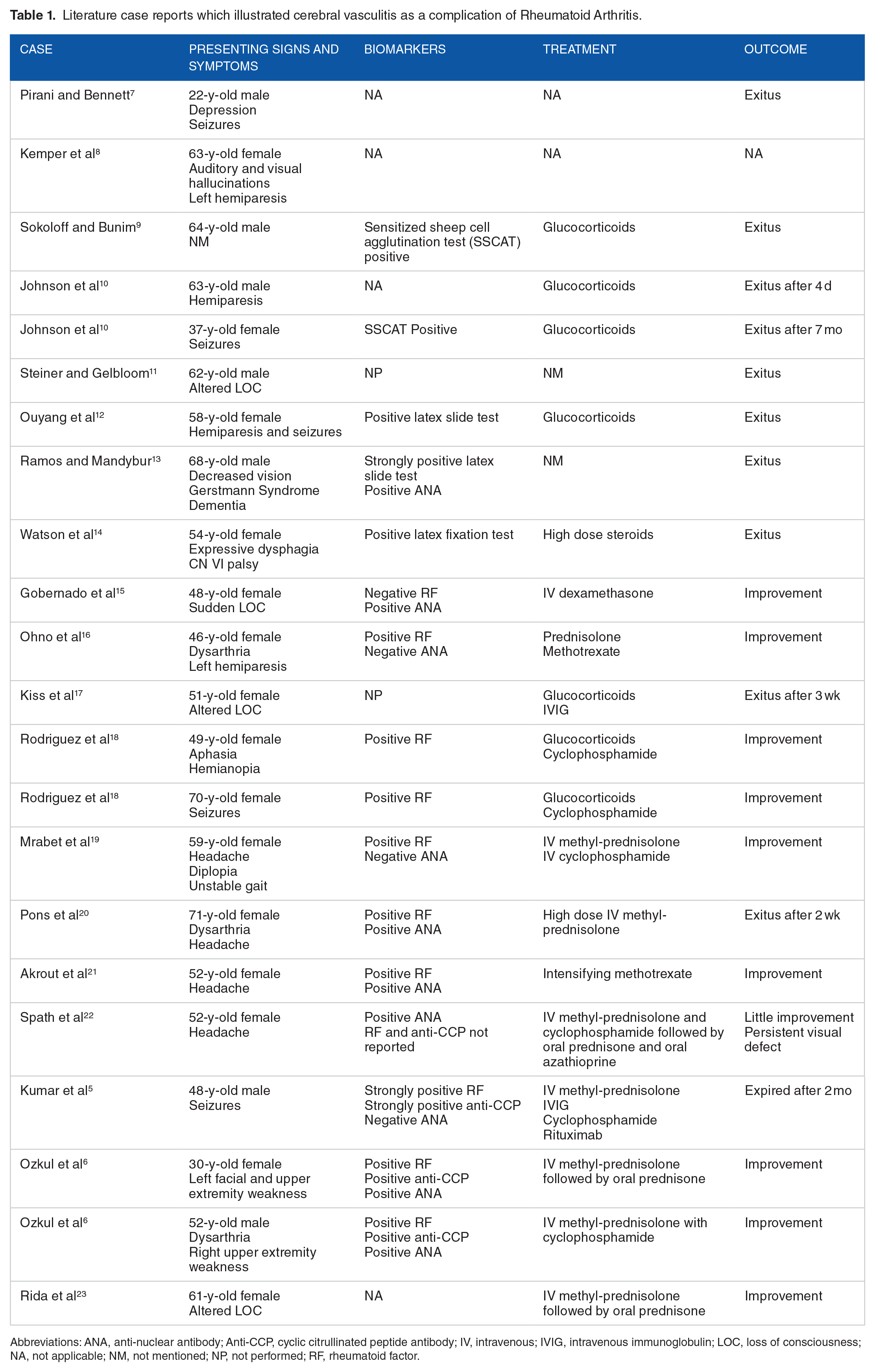
Abbreviations: ANA, anti-nuclear antibody; Anti-CCP, cyclic citrullinated peptide antibody; IV, intravenous; IVIG, intravenous immunoglobulin; LOC, loss of consciousness; NA, not applicable; NM, not mentioned; NP, not performed; RF, rheumatoid factor.
Anti-CCP is associated with several extra-articular RA manifestations even if present at low titer in the absence of RF. 24 In patients with long-standing RA, 1.7% of extra-articular manifestations include vasculitis. 24 Cerebral vasculitis can present as part of a systemic manifestation of vasculitis or as an isolated finding. A positive ANA in RA patients is usually considered as a risk factor. 22 Since the available data is still limited, the exact pathophysiology of RA biomarker, including anti-CCP, induced cerebral vasculitis is still unknown. In our case, the patient had a low disease activity indicated from absence of tenderness and swelling in her joints. The absence of inflammatory markers in the CSF examination indicates that the cerebral vasculitis is secondary to a systemic inflammation.
CNS vasculitis can occur secondary to multiple autoimmune diseases such as systemic lupus erythematosus (SLE), dermatomyositis, and mixed connective tissue disease. 25 Nevertheless, CNS vasculitis can be the manifestation of a systemic vasculitis such as antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis.25,26 As the renal function tests, including a urine microalbumin, was normal in our case, and the patient had an obvious cause of CNS vasculitis, serum ANCA levels were not assessed. Additionally, CNS damage is commonly found in patient with polyarteritis nodosa (PAN) 27 and mainly occurs at late disease stages. 28 Our patient, who was properly followed-up, did not manifest PAN signs or symptoms prior to presentations such as weight loss, fever, peripheral neuropathy, or cutaneous involvement which makes CNS vasculitis secondary to PAN an unlikely differential. Therefore, a whole body CT scan with contrast was not performed.
The diagnosis of rheumatoid cerebral vasculitis is often challenging. A patient with a past medical history of RA, either long standing or newly diagnosed, presenting for signs and symptoms of stroke, headache, and encephalopathy 29 might be developing a CNS manifestation of RA such as atlantoaxial dislocation, hyperviscosity syndrome, reversible vasoconstriction syndrome, rheumatoid meningeal nodules, and pachymeningitis. 30 The most common symptoms of RA-associated cerebral vasculitis are seizures, headache, mental status changes, and cranial nerve involvement 6
Multiple bilateral ischemic lesions or decreased cerebral blood flow involving cortical and subcortical regions could be detected on cerebral MRI. 22 In our case, there was decreased cerebral blood flow secondary to vasculitis which did not lead at the time of diagnosis to ischemia. FLAIR weighted MRI is recommended whereby it can show hyperintensities in the areas affected. Such hyperintense lesions on T2-weighted MRI images and FLAIR images in brain affected regions might hint on the diagnosis of progressive multifocal leukoencephalopathy, 31 especially in a patient treated with rituximab as in our patient. Therefore, further diagnostic investigations are needed. The reduction in the NAA peak is also associated with a demyelination change. 32 In addition, the brain lesions in inflammatory demyelinating disorders might mimic white matter vasculitic lesions. 33 However, MR angiography can differentiate between the two. In addition, an increase in the choline peak is seen in active demyelination secondary to destruction of the myelin sheath. 34 Therefore, the absence of increased choline was against demyelination in this case, as well as the absence of enhancement on the post-gadolinium images.
On MR angiography, a decreased caliber and contour irregularities of cerebral arteries suggest cerebral vasculitis. 22 The other important diagnostic tool is the transcranial doppler ultrasonography which reveals a rise in peak systolic blood flow in cerebral arteries and a drop in cerebral vasomotor reactivity. 22
CSF examination is useful to asses for secondary causes such as infection or systemic inflammation. For example, pleocytosis, increased protein content, or both (as in our case) can occur in CNS vasculitis secondary to systemic vasculitis.35,36 However, CSF may also be entirely unremarkable, with absence of inflammatory markers.26,37,38
A brain biopsy is an invasive procedure that is not so often performed, although it confirms the diagnosis. The biopsy shows inflammatory infiltration of the vessel wall with or without destruction. 22
There is still no consensus regarding the treatment of RA-associated cerebral vasculitis. While steroids have been used in most cases, either as a monotherapy or as an add-on, methotrexate, 21 cyclophosphamide, 6 and IVIG 5 might be good alternatives with variable clinical outcomes. The rapid response to steroids is of an interest in our case.
Conclusion
Rheumatoid arthritis can have several neurological complications, among which is cerebral vasculitis. Even in the presence of adequate immunosuppression markers such as negative biomarkers, the immunosuppression can be inadequate enough to prevent neurological complications such as cerebral vasculitis. Normal lymphocyte count and elevated total serum protein are hints to inadequate immunosuppression. The diagnosis of cerebral vasculitis is often challenging and needs thorough assessment and a high index of suspicion. Steroids are commonly used with a rapid response.
Footnotes
Acknowledgements
The authors acknowledge the help of all colleagues at the American University of Beirut Medical Center.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
IU, AJ, and RH conceived and supervised the study; GEH and RH analyzed data; GEH, FD, and HEO wrote the manuscript; AJ and IU made manuscript revisions. All authors reviewed the results and approved of the final version of the manuscript.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor of this journal.
