Abstract
Intestinal diffuse ganglioneuromatosis is a rare, benign, neoplastic condition characterized by disseminated proliferation of neural elements. It has an established association with several systemic disorders including: multiple endocrine neoplasia IIB (MEN IIB) and von Recklinghausen’s disease. However, isolated GNs are very rare and sporadic. We report a case of a 6-year-old patient was referred to the emergency department due to a colon perforation during endoscopy. He then underwent sigmoidectomy and the pathologic studies revealed prominent signs of diffuse ganglioneuromatosis. Unfortunately, the patient died the day after because of the late diagnosis and treatment. Such presentation emphasizes the importance of considering diffuse ganglioneuromatosis, among other causes, in patients with intractable GI symptoms despite medical treatment.
Introduction
Ganglioneuromas are benign tumors typically developing from sympathetic ganglia and adrenal glands, yet their involvement in the viscera is a rare occurrence. 1 Gastrointestinal (GI) ganglioneuromas are benign tumors consisting of 3 subgroups: (1) solitary polypoid ganglioneuromas, (2) ganglioneuromatous polyposis, and (3) diffuse ganglioneuromatosis. The latter is the least common and it is usually associated with multiple endocrine neoplasia (MEN IIB) and, less commonly, von Recklinghausen’s disease, Cowden’s disease, and even colonic adenocarcinoma. 2 Herein, we discuss, an extremely rare case of isolated diffuse intestinal ganglioneuromatosis in a 6-year-old boy with no history of any associated syndrome.
Case Presentation
A 6-year-old boy presented to the emergency department with a 2-week history of abdominal pain, vomiting, fever, and diarrhea. His physical examinations showed abdominal tenderness, his laboratory tests were of no significance except for a fecal occult blood, and the ultrasound revealed a prominent thickening of the descending and sigmoid colon wall. As a result, colonoscopy was performed revealing the emergence of an extremely edematous reddish mucosa and ulcerations covered by exudates lining the sigmoid and the ascending colon (Figure 1A-D). Endoscopic colonic biopsies revealed chronic inflammation with granulation tissue suggesting an inflammatory bowel disease (IBD) or an infection. Consequently, he was put on antibiotics, Prednisolone and Mesalazine.

Colonoscopy showing an edematous sigmoid colon with (A) ulcerated (B) reddish surface (C, D) covered by exudates.
In the following months, the patient did not show enough improvement, while conversely, he developed a persistent abdominal distention and pain in the left iliac fossa. Since our hospital does not own an MR Enterography; another colonoscopy was scheduled. Unfortunately, the colon was extremely friable it perforated, and the kid underwent surgery. At surgery, an old perforated necrotic area was spotted necessitating resection of the whole sigmoid (Figure 2A). The resected specimen was then sent to the pathology department for further examination. Grossly, the colon had multiple perforations, a stricture, and an inflamed and thickened area to the point where it almost closed the lumen (Figure 2B). Histologically, the H&E-stained sections of the sigmoid colon demonstrated extensive erosion of the overlying mucosa, interlacing fascicles of spindle-shaped cells that had Schwannian features, as well as ganglion cells in the muscularis propria and the submucosa of the sigmoid (Figure 3A-C). No signs of atypia were detected. Immunohistochemical stains were positive for S100 and synaptophysin (Figure 4A and B), confirming the diagnosis of diffuse colonic ganglioneuromatosis.
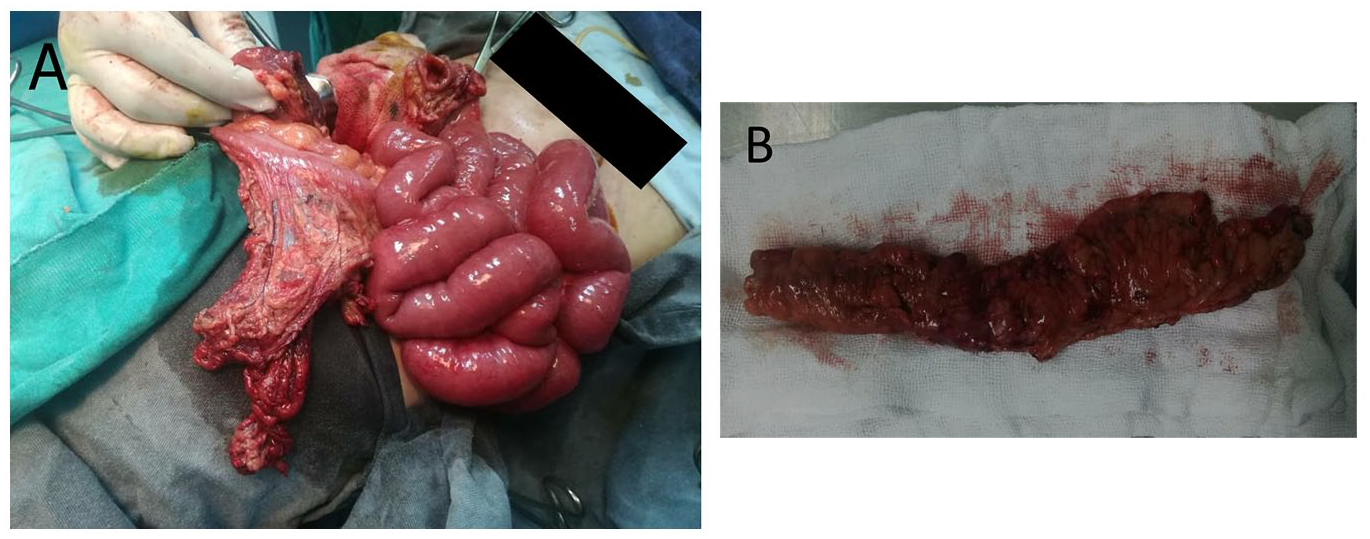
Surgical findings and gross appearance of an (A) edematous (B) perforated sigmoid colon.

(A) Diffuse intestinal ganglioneuromatosis (H&E), (B) clusters of mature ganglion cells within bland, Schwannian stroma (H&E, ×200), (C) diffuse, poorly demarcated proliferation of nerve ganglion cells (H&E, ×400).
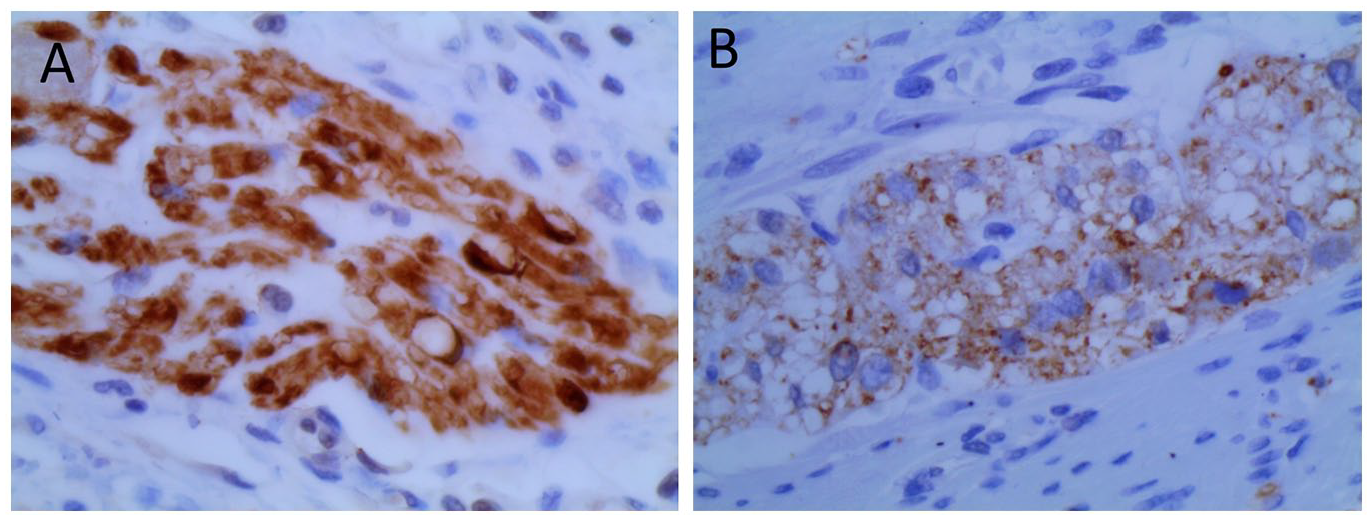
Immunohistochemical staining: (A) S100 staining highlights ganglion cells and abnormal nerve fibers, (B) positive synaptophysin staining.
However, after the surgery he was admitted to the ICU, but few hours later, unfortunately, the kid developed cardiac bradycardia with respiratory depression due to excessive blood loss. He did not respond to resuscitation, and eventually died.
The child’s family history is unremarkable, and he did not display any characteristic clinical hallmarks of systematic disorders such as, marfanoid habitus in MEN2B, cafe’-au-lait spots and cutaneous neurofibromas in NF1. Also, he is not known for any congenital or genetic disease.
Discussion and Conclusion
Ganglioneuromas of the intestinal tract are rare benign neoplasms, defined by an overgrowth of nerve ganglion cells, nerve fibers, and supporting cells. Depending on their formation, they are classified into 3 subtypes: polypoid ganglioneuroma, ganglioneuromatous polyposis, and diffuse ganglioneuromatosis. The most common one, polypoid ganglioneuroma, emerges usually as a sole polyp or few small polypoid lesions. They are small (<2) polyps that are usually adenomatous or hyperplastic, sessile or pedunculated. It may histologically be reminiscent of juvenile polyp, ergo an accurate differential diagnosis is required. 3 Ganglioneuromatous polyposis is generally characterized by more than 20 sessile or pedunculated discrete polyps. Such polyps can be filiform projections containing ganglion cells, or histologically undistinguishable from polypoid GNs. 3 Lastly, diffuse ganglioneuromatosis is a rare, benign, disseminated or poorly demarcated nodular proliferation involving the myenteric plexus.4,5
The latter form may affect any segment of the alimentary tract, even the small intestines, 2 but it is most commonly located in the colon, the terminal ileum, and the appendix.6,7
Diffuse ganglioneuromatosis (DGN) mainly affects the youngsters as a component of multiple endocrine neoplasia (MEN) IIB syndrome, Von Recklinghausen’s disease, Cowden’s disease, Ruvalcaba-Myhre-Smith or Neurofibromatosis-1. Some sporadic cases reported an association between DGN and colonic adenomas or even adenocarcinomas,8,9 but isolated DGN with no underlying pathology is an exceptional finding.
Apart from a certain hypothesis suggesting that a soluble nerve growth factor may be linked to ganglioneuromas development, the exact etiology of diffuse ganglioneuromatosis has not been attributed to any specific gene or mechanism yet.5,9
Individuals suffering from DGN does not show any distinctive clinical manifestations, as it displays a wide array of symptoms based on the tumor’s size and location. These symptoms range from abdominal pain, change in bowel habits, ileus, weight loss, appendicitis and gastrointestinal bleeding; to a more severe conditions such as: acute intestinal obstruction or motility disorders causing megacolon or diverticula.1,7 Also, localized lesions may remain asymptomatic and be found incidentally. 1 With this being the case, DGN can easily be disregarded, clinically, for other GI conditions including: infections or IBD. Our patient’s initial symptoms of diarrhea, fever, and vomiting were not indicative of any specific disease, but the consistency and refractory nature of these symptoms were worrisome and more tests had to be done.
Although the diagnosis of DGN can be assumed radiologically, it is usually left out due to its rarity.2,4 As a result, endoscopic studies composed a more fundamental method, where the tumor can appear as a distinctive thickening of the bowl wall extending from 1 to 17 cm long, with or without wide stricture. 8 In some instances, however, findings can be subtle; therefore, realizing the histopathologic features is becoming more crucial. 8 In regards to our patient, diffuse ganglioneuromatosis was not considered at first, because the edematous reddish mucosa and ulcerations appearing in endoscopy, alongside the microscopic chronic inflammation and granulation tissue masked out the features of DGN and were more suggestive of an IBD. It was not until after resecting the colon and ensuing thorough histologic and immunohistochemical studies on it, the diagnosis was established. In fact, several cases of DGN mimicking Crohn’s have been reported in the literature.6,7
Other considerations in the differential diagnoses of DGN include: Intramural GI schwannomas, Gastrointestinal granular cell tumor (GCTs), Perineuriomas, Neurofibromas. However, ganglioneuromas can be distinct from other neurogenic spindle cell lesions by the existence of ganglion cells. 10
Moreover, the tumor is well differentiated with no evidence of malignancy. It is characterized by an exuberant, poorly demarcated, nodular, or diffuse proliferation of nerve fibers, ganglion cells, and supporting cells of the enteric nervous system in the muscularis propria and other layers. 11 These morphologic features are sufficient to make a diagnosis, but immunohistochemical markers of neural differentiation can be applied to highlight the ganglion cells.5,11
The treatment of choice for the diffused form of GN is surgical resection of the diseased bowl, that is because conservative methods have proven to be of low effect.4,9 Our 6-year-old was taking the medication to treat the suspected IBD, but unfortunately, the dismissing of DGN delayed the surgical treatment and contributed to the deterioration of his condition.
To conclude, we believe that such case emphasis the importance of considering the diffuse form of intestinal ganglioneuromatosis as a cause to otherwise unexplained generic GI symptoms with an unusual progression trajectory. Also, to act upon that by giving great attention to endoscopic and histologic studies, and conducting surgical treatment when deemed necessary.
Footnotes
Author Contributions
Dr. Haya Mohammed Kheir and Prof. Zuheir Alshehabi conceptualized and drafted the initial manuscript and revised it, performed the histological analysis and provided the figures. Nour Haidar and Dr. Mouhannad Dayoub gathered information from the literature, and helped drafting the manuscript. Dr. Hanin Mansour and Prof. Ali Ibrahim, participated in reviewing the final draft and supervised the case progression and treatment and provided the endoscopic figures. Asst. Prof. Ammar Omran carried out the surgical procedure and drafted the case presentation. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for Publication
Written informed consent was obtained from the parent of the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
