Abstract
Objectives:
Nivolumab, a fully IgG4-programmed death-1 inhibitor antibody, led to improved overall survival compared with single-agent therapy in patients with platinum-refractory recurrent head and neck cancers. In general, nivolumab is used in inoperable patients. To the best of our knowledge, there have been no reports of salvage surgery during nivolumab therapy for patients with head and neck cancer. We report the case of a woman treated with salvage reconstructive surgery during nivolumab therapy.
Method:
Case report and literature review.
Results:
The patient underwent nivolumab therapy for recurrent primary and neck disease after induction chemotherapy, followed by concurrent chemoradiation therapy. The neck disease shrunk, whereas the primary disease temporarily shrunk but later progressed again. Recurrent primary disease led to a narrowing of her airway, and she required airway management. We performed total pharyngolaryngectomy with free jejunal reconstruction, and her quality of life improved. The surgery was performed without complications and the postoperative course was uneventful. She was discharged postoperative day 18 with oral intake function and a safer airway.
Conclusion:
As far as we know, this is the first report of salvage surgery during nivolumab therapy for patients with head and neck cancer. The salvage reconstructive surgery in this case proceeded uneventfully.
Keywords
Introduction
The CheckMate 141 trial showed that nivolumab provided an improvement in overall survival (OS) compared with standard second-line single-agent systemic therapy in patients with platinum-refractory recurrent head and neck cancers. 1 Nivolumab is used for patients who experience tumor progression or recurrence within 6 months of platinum-based therapy. Nivolumab therapy is indicated for and mostly used in patients who have unresectable disease.1,2
Several past studies reported that patients with prior chemoradiotherapy or radiotherapy in combination with cetuximab have a significantly higher risk of surgical complications.3,4
Here, we describe a patient who had recurrent primary malignant disease during nivolumab therapy.
Case Report
A 74-year-old Japanese woman was diagnosed with T3N2cM0 hypopharyngeal cancer. We considered the disease unresectable due to retropharyngeal lymph node metastasis with radiologically proven total encasement of the internal carotid artery (Figure 1). She was treated with induction chemotherapy followed by concurrent chemoradiotherapy (CCRT) as part of a clinical trial of paclitaxel, carboplatin, and cetuximab followed by chemoradiotherapy. The disease was completely resolved 11 weeks after CCRT. However, 24 weeks after CCRT, the primary tumor and neck nodes recurred.
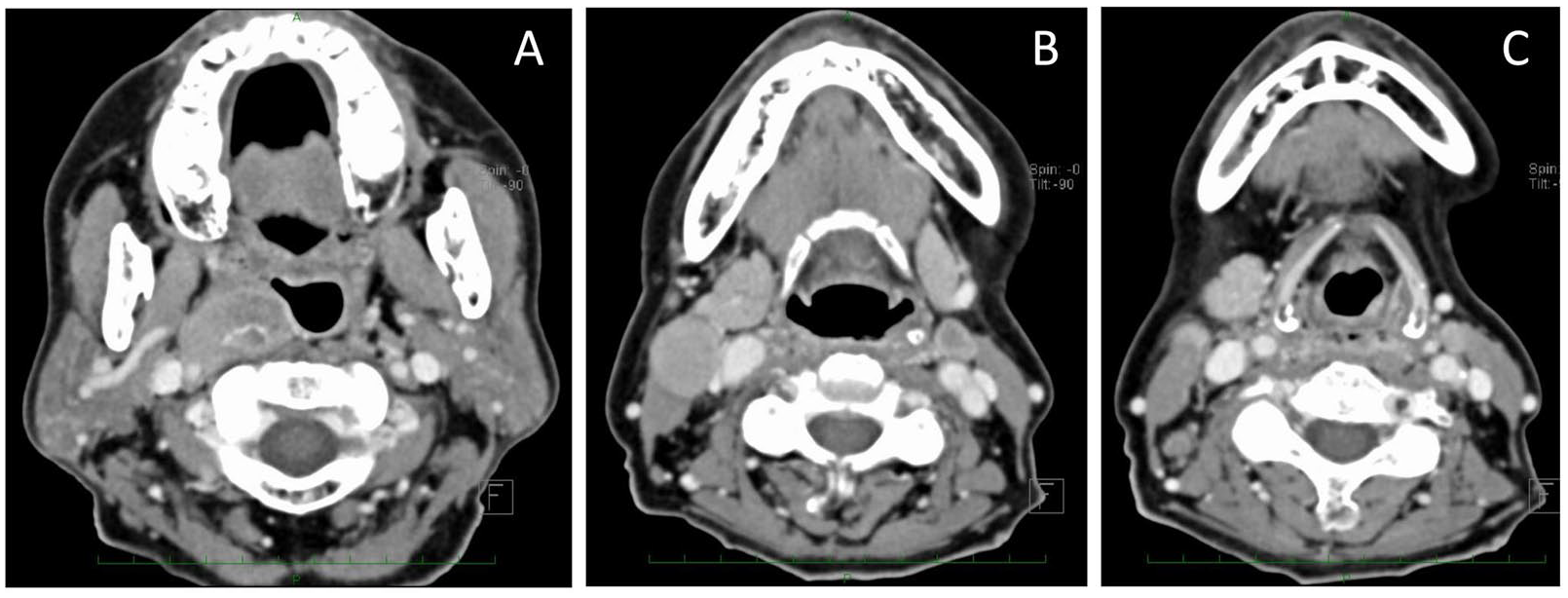
(A) Retropharyngeal node metastasis involved the right internal carotid artery. (B) Bilateral neck metastases were found. (C) The right pyriform sinus and posterior wall were enhanced using a contrast agent.
We considered the recurrent diseases to be platinum-refractory and started nivolumab therapy. Unfortunately, despite receiving 13 courses of nivolumab therapy, the primary disease continued to progress, although the neck nodes shrank and could no longer be detected. The recurrence at the primary site soon led to narrowing of her airway, which required airway management. She could not eat adequately and depended on gastrostomy tube feeding. We could not detect any distal metastases and the retropharyngeal node had also disappeared. Figure 2 shows a summary of the treatment and tumor response until the patient underwent salvage surgery.

Summary of treatment and monitoring of tumor response. (A) Various interventions received by the patient before salvage surgery. Arrows indicate the timing of each intervention. CBDCA indicates carboplatin; CDDP, cisplatin; PTX, paclitaxel. (B) Both primary and neck diseases disappeared at 11 weeks after concurrent chemoradiotherapy. (a) The retropharyngeal lymph node was not detected by positron emission tomography-computed tomography (CT). The CT scans showed recurrent disease at 24 weeks after concurrent chemoradiotherapy. (b) Arrows indicate the swollen retropharyngeal and paratracheal nodes. (c) Recurrent primary disease was detected after 13 courses of nivolumab therapy.
We performed total pharyngolaryngectomy (TPL) with free jejunal reconstruction. The operation time was 6 hours 54 minutes, and blood loss was 315 mL.
There were no particular difficulties encountered during the surgery apart from some adhesions in certain parts as a consequence of previous oncological treatment (Figure 3). In fact, the surgery did not differ from other salvage surgeries. The recurrent primary tumor was completely resected macroscopically; however, malignant cells were present in the margins of the lymph vessels. Microvascular anastomoses were performed uneventfully using the superior thyroid artery and the internal jugular vein.

(A, B) Some adhesions were found, although they did not differ greatly from those observed after chemoradiation therapy. (C) We performed free jejunal reconstruction. (D) We also performed additional caudal resection to maintain the surgical margin.
The postoperative period was uneventful and gastrostomy tube feeding was started from postoperative day 2. No leakage was detected by a barium swallow test on postoperative day 14, and the patient started oral intake of food on postoperative day 15. There were no postsurgical complications. She was discharged on postoperative day 18 with a stable and safe permanent tracheostomy and normal oral intake. We resumed nivolumab therapy on postoperative day 38.
Nivolumab is still regularly being administered and the patient continues to undergo regular follow-up at our outpatient unit. She has not reported any particular symptoms to date.
Discussion
In general, nivolumab is used after the failure of platinum-based chemoradiotherapy (CRT).1,2 The response rate to nivolumab was reported to be 13.3% in patients with recurrent head and neck cancer after platinum-containing chemotherapy. 1 As far as we know, there have been no reports of salvage surgery during nivolumab therapy for patients with head and neck cancer.
In our case, treatment with nivolumab resulted in the disappearance of the recurrent neck disease, although there was recurrence at the primary site. With the expansion of nivolumab usage, resectable recurrent diseases are expected to be detected in more patients with head and neck cancer during or after nivolumab therapy. Salvage surgeries may enable continuous nivolumab therapy and improve patient survival.
Although the patient’s general condition was sufficiently good to allow the surgery, there were some points to consider before salvage surgery. First, we could not predict the potential complications of the surgery as no similar case had been reported for the head and neck. Second, as the effectiveness of nivolumab is expected to diminish over time, TPL might be considered merely as a palliative surgery. On the other hand, the patient soon needed airway management and depended on gastrostomy tube feeding when the recurrent disease was detected. Finally, the benefits and disadvantages of surgery were discussed and the patient gave her consent. No further symptoms had been reported 15 months after the surgery and reinitiation of nivolumab. Nevertheless, further follow-up is needed and is currently ongoing.
Her treatment course was similar to that of other patients who received reconstructive surgery for post-CRT recurrent disease. The surgical findings were also quite similar. We think that other salvage surgeries can also be performed for patients with resectable recurrent disease during or after nivolumab therapy.
It is difficult to assess the effectiveness or safety of surgery during nivolumab therapy because of its infrequency and lack of data in the literature. Past studies showed surgeries during nivolumab therapy were performed safely in patients with cancers involving body sites other than the head and neck. 5 Thus, it is thought that surgery during nivolumab therapy affords a feasible option for patients with head and neck cancer.
While other recurrent or metastatic lesions are controlled by systemic therapy, in cases with limited progression of the cancer in non-head-and-neck sites, salvage surgery for the progressive disease is performed.6-8 After such salvage surgeries, nivolumab or other drugs are resumed as maintenance therapy.7,8 A past study showed that OS was better when the same drug was used for maintenance therapy than when the drug was changed. 9 Although the merits of maintenance therapy for patients with head and neck cancer were not elucidated, it seemed reasonable to resume nivolumab after salvage surgery.
Conclusion
To the best of our knowledge, this is the first report of successful salvage surgery during nivolumab therapy for a patient with head and neck cancer.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NT and TF wrote the manuscript. TS, MT, and TH were treating physicians and helped in drafting the manuscript. TT, WO, and MI helped in drafting the manuscript and revised it critically. RH supervised this case report. All authors have read and approved the final manuscript.
