Abstract
The use of mesenchymal stem cells from bone marrow concentrate (BMC) has become an increasingly popular option as an alternative to total joint replacement. Although there is evidence to support the use of BMC injections to improve quality of life for patients with knee osteoarthritis (OA), there is limited evidence to support its use in patients with hip OA. This case series provides the short-term outcomes of 4 hip OA patients who each underwent 4 BMC injections. On average, the last follow-up was administered 3.5 months after the first injection. The results show that patients experienced decreased resting and active pain compared with baseline and mean 72.4% total overall improvement. Patients also reported less difficulty in performing daily activities following the procedure. These encouraging results warrant further research to better understand the effects of BMC injections on hip OA.
Introduction
Osteoarthritis (OA) is the most prevalent type of arthritis, 1 with projections of 25% of the US adult population to be diagnosed by 2030. 2 Symptomatic hip OA affects 8.7% of men and 9.3% of women over the age of 45 in the United States. 3 Hip OA may result from cartilage loss, osteophyte formation, or changes in the shape of the femur head. 4 Patients with hip OA may experience pain and limited mobility and function. 5 These patients often treat surgically with total hip replacement. However, hip replacement surgery is associated with complications such as dislocation, infection, reoperation, inefficiency, and even death.6–8 A recent study showed that 7% to 23% of patients experienced chronic pain after surgery. 6 This high prevalence of chronic pain demonstrates the need for an effective, more conservative alternative to surgical treatment for hip OA.
Surgical alternatives include intra-articular corticosteroid (IACS) injection, nonsteroidal anti-inflammatory drug (NSAID) treatment, and physical therapy. Although IACS injections are performed less commonly for the hip than for the knee, they have demonstrated effectiveness in relieving hip pain from 2 weeks to 3 months after treatment.9,10 However, these injections have proven deleterious to joint cartilage. Oral or topical NSAIDs, which have been shown to relieve pain for the first 2 weeks of treatment, seem largely ineffective in later weeks of treatment. 11 Manual physical therapy is yet another treatment used by some patients with hip OA for pain relief and increased functionality. One study of manual physical therapy showed that patients experienced increased total range of hip motion and decreased pain. 12
A promising alternative to treat hip OA is with bone marrow concentrate (BMC) injections. Bone marrow concentrate is a solution that contains many biofactors, cytokines, and growth factors. However, for therapeutic purposes, it most notably contains platelets and mesenchymal stem cells (MSCs). 13 The MSCs can differentiate into cartilage, bone, and muscle; thus, it is thought that they could play a role in cartilage regeneration. 14 The MSCs also secrete trophic factors that have been shown to aid in tissue regeneration. 15 Several studies show that BMC injections improve patients’ pain and quality of life in patients with knee OA.16–18 A recent study of 10 patients with hip OA who were injected with autologous cultured MSCs from BMC showed that 100% of patients had decreased pain, increased hip function, and increased range of motion between 16 and 40 months. 19 Furthermore, a study that included 216 hips with OA showed that patients had a 31.2% total overall improvement, reduction in pain, and increase on the Oxford Hip Score following one BMC treatment. 20
We hypothesize that treating hip OA with multiple BMC injections may be more effective than treating patients with a single BMC injection. As reported in our knee OA study, 21 if a patient at our clinic requires multiple injections, we advise them to receive injections approximately 14 days apart. This 14-day time period is when there is growth factor secretion from various cell types that participate in the late phases of wound healing.22,23 Transforming growth factor β (TGF-β) is one of these growth factors and has been shown to enhance MSC growth and osteogenic differentiation.23,24 By injecting patients with a steady flux of BMC in a short time period, we believe that patients will experience superior symptomatic relief compared with one treatment.
We found only 2 other studies that have reported the efficacy of BMC injections to treat hip OA. With this lack of literature, the objective of this study was to report our initial patient outcomes regarding the efficacy of BMC injections as an alternative to total hip replacement.
Methods
Patients
This article reports the results from a case series of clinical practice outcomes, where variables were administered prospectively and data were analyzed retrospectively. Patients included in this study underwent 4 BMC treatments for hip OA at a solo practitioner private practice from June 2016 to August 2017. This study compares results to another study of our patients with knee OA who underwent 4 BMC treatments. 21 The patients who underwent 1, 2, or 3 treatments will be reported in a separate study.
Patients in this study underwent 4 successive BMC treatments with follow-up injections at a mean of 45 days after the first treatment, 12.75 days after the second treatment, and 13 days after the third treatment. All patients were directed to have injections approximately 14 days apart; however, scheduling conflicts between patients and physician resulted in average follow-up injections greater than 14 days. The final follow-up questionnaire was administered, on average, 22.75 days after the final injection. The diagnosis of hip OA was based on radiographic findings. All treatments were prescribed on an individual basis, as recommended by a physician. Written informed consent was obtained prior to each treatment. Patients were instructed not to use NSAIDs during treatment, as they hinder tissue regeneration. 25 Patient characteristics can be found in Tables 1 to 3.
Patient characteristics.

Abbreviation: BMI, body mass index.
Patient pain and lower functionality scores before and after treatment.

Mean resting pain, active pain, total improvement, and functionality score by bone marrow concentrate injections received—N = 4 hips.

Procedure
Patients were in the prone position and sterilized with 10% Povidone-Iodine on the skin above the posterior superior iliac spine (PSIS). Next, 4% chlorhexidine gluconate (Hibiclens) was administered with sterile gauze in a circular motion starting at the PSIS. Patients were then anesthetized with 10 -cc of 1% lidocaine and 2 -cc of 8.4% sodium bicarbonate, injected locally on and around the patient’s PSIS. After sufficient local anesthesia was achieved, a fenestrated 11-gauge, 4-in disposable needle was drilled to penetrate the PSIS and extract BMC. A 20 -cc syringe prepared with 1 -cc of heparin (1000 USP units/mL) was used to extract BMC for a total yield of 19 -cc. To maximize stem cell yield and avoid an excess of peripheral blood, the needle was rotated slowly within the ilium cavity and penetrated deeper as required. The BMC was then spun in a centrifuge, and the upper portion without visible red cells was isolated from the centrifuged BMC. About 1 -cc of ropivacaine was added to 5 -cc of centrifuged BMC to ensure that the area after injection was less painful. Ropivacaine has shown limited toxicity to MSCs. 26 The skin was sterilized with 10% Povidone-Iodine followed by 4% Hibiclens. The spun BMC was injected via a 22-gauge, 4-in needle under ultrasound guidance into the hip joint capsule via an anterior approach.
Outcomes
The outcomes of interest for this case series were changes to resting pain and active pain (numerical pain scale), overall improvement (percentage scale), and joint function (scored questionnaire). Variables were chosen for ease of comparison with similar variables reported by other studies of BMC for knee OA treatment.16,17 Data were collected at baseline and following each treatment (Figure 1). The functionality portion of the questionnaire, which assessed degree of difficulty in performing daily activities, was based on 10 of 20 activities assessed in the Lower Extremity Functional Scale, 27 but also included a “not applicable (N/A)” response option. This scale has been shown to be a reliable functionality questionnaire for hip OA as well as an alternative to the Western Ontario and McMaster Universities Arthritis Index. 27 To assess resting and active pain, the numerical pain scale uses a scale of 0 (no pain) to 10 (extreme pain). 28 Finally, the form included a subjective measure of how much overall improvement the patient experienced following treatment on a scale of 0% to 100%.
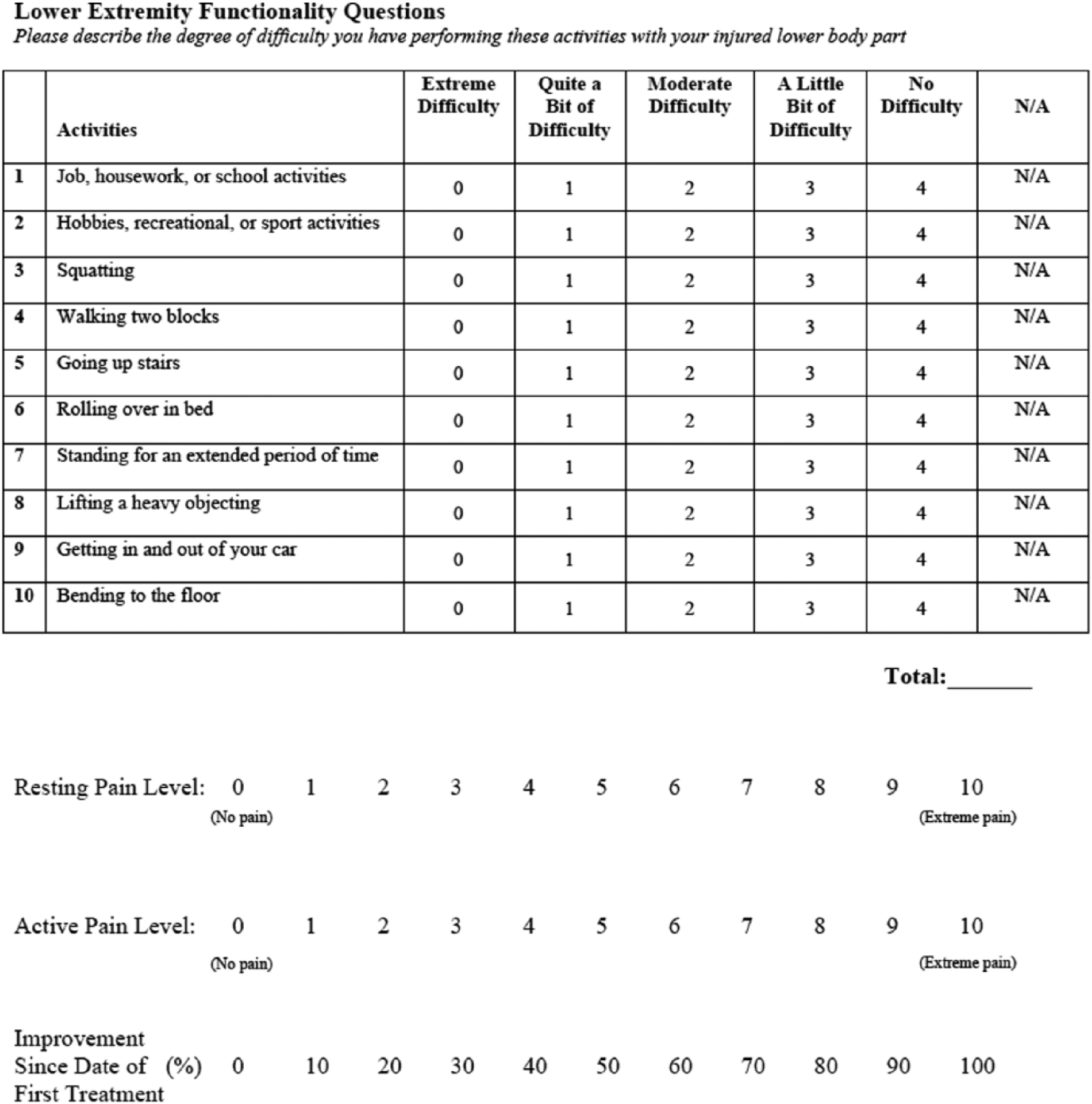
Patient questionnaire.
Cases
Case 1
The first patient was a 75-year-old woman with a 10-year history of progressive right hip pain. She had consulted with 2 orthopedic surgeons, each of whom recommended total hip replacement as her only option. Magnetic resonance imaging (MRI) of the right hip showed severe OA with extensive spurs and near-complete obliteration of the joint space. The patient experienced the most pain when sitting for extended periods of time. She had tried massage therapy and anti-inflammatory gel for pain relief; however, her resting and active pain were each 6/10, and her base functionality score was 33/40.
The patient underwent 4 BMC treatments over a 49-day period, and her symptoms improved with treatment. After the second treatment, the patient reported a 40% improvement, with more joint flexibility. At the follow-up after the fourth treatment, the patient noted a 60% total overall improvement with active and resting pain levels of 2/10. Her functionality score also increased to 37/40. At the final follow-up, the patient reported that she was sleeping better and that she had avoided a total hip replacement.
Case 2
The second patient was a 61-year-old man who had chronic hip pain for 2 years. His pain was most pronounced while standing or with intensive exercise. The patient had undergone chiropractic adjustments, cupping, and physical therapy for pain relief, but his symptoms returned within 1 week of each treatment. Radiographic assessment of the right hip demonstrated severe OA. At baseline, the patient reported a resting pain of 1/10 and an active pain of 7/10. His functionality score was 28/40.
The patient underwent 4 BMC treatments during a 42-day period. After the first BMC injection, he reported an 80% total overall improvement, with no resting pain. At the follow-up after the fourth treatment, the patient reported only infrequent, mild pain (0/10 at rest and 2/10 with activity). Following treatment, he was able to resume exercise and an active lifestyle. His functionality score was 37/40 and reported that he had 94% total overall improvement.
Case 3
The third patient was a 76-year-old man who had a 7-year history of chronic hip pain. He previously underwent a left hip replacement but reported that the recovery had been challenging. Furthermore, he did not want a second hip replacement that had been recommended by his surgeon. The MRI of the right hip showed moderate to severe OA with articular surface irregularity. The patient had physical therapy and massage but his pain had progressed. He underwent 4 BMC treatments over a 54-day period. At baseline, his resting pain was 4/10, active pain was 5/10, and functionality score was 33/40. After the second treatment, the patient reported a 50% total overall improvement with increased hip flexibility and range of motion. At the follow-up after the fourth treatment, the patient reported a 65% total overall improvement with a resting pain of 1/10 and active pain of 2/10. Following treatment, his functionality score was 37/40, and he was able to walk long distances with no pain and enjoyed a significantly improved quality of life.
Case 4
The fourth patient was a 56-year-old man who had a 2-year history of chronic hip pain resulting from his physically demanding career as a contractor. The patient reported that his pain was worsening and limiting his everyday activity. He had an epidural injection with no pain relief. A radiograph of his right hip showed mild osteoarthrosis. His resting pain was 2/10, active pain was 5/10, and functionality score was 17/40 at baseline. The patient underwent 4 BMC treatments over a 146-day period. The patient experienced minor, incremental improvements following each of the first 3 treatments (overall improvement, 30%). He reported improved ability to perform daily activities with less pain during the course of those 3 treatments. After 40 days of the fourth treatment, the patient reported a 70% total overall improvement. His resting and active pain were 1/10, and his functionality score increased to 30/40.
Discussion
All patients in this case series experienced decreased pain and improved functionality compared with baseline. Notably, patient 4 experienced an 80% decrease in resting pain, patient 1 a 67% decrease in active pain, and patient 4 a 76% improvement in functionality score. Patient 2 also reported 94% total overall improvement after treatment. It is encouraging that all patients experienced at least a 60% total improvement following treatment, and 100% reported that they were better able to perform their daily activities. On average, patients experienced successive decreases in resting and active pain after each treatment. Patients also experienced a mean increase in total overall improvement percentage and functionality score after subsequent treatments. Prior to BMC treatment, 2 of the 4 patients had been advised to undergo total hip replacement. Following treatment, neither patient considered surgery.
Most studies using BMC to treat OA to date have focused on knee treatment. Many of these have shown that patients with knee OA have a significantly improved quality of life following BMC injection.16–18 The current study provides evidence that BMC injection under ultrasound can improve patient symptoms of hip OA as well. A recent case series showed symptomatic improvement in patients with ankle, hip, and knee OA using whole, unspun BMC injections. 29 These authors hypothesized that patients’ improvements were more due to the microenvironment of the cells, not the concentration of MSCs. Contrastingly, another study of hip OA used cultured MSCs to increase their numbers prior to injection. 19 Although patients in that study reported substantial improvement in pain and range of motion after 3 weekly injections, there is limited evidence that this technique is more effective than using noncultured cells.
The promise of BMC treatment for OA is due to MSCs’ chondrogenic potential to stimulate cartilage growth. However, recent research has supported that the trophic factors secreted by MSCs have great potential benefit as well. Through paracrine signaling, these trophic factors have been shown to suppress inflammation and increase cartilage regeneration through stimulation of nearby progenitor cells. 30 The excess of inflammation in an osteoarthritic joint may be the pain generator for a patient’s condition. Suppressing this inflammation would also provide a beneficial healing environment for the MSCs and platelets. We hypothesize that patients’ symptomatic relief is due to a combination of these factors; however, additional research is needed to confirm this.
We hypothesize that receiving multiple BMC injections in a short time period might give the hip effective cartilage repair. This explains why our patients with hip OA in the present case series had similar short-term symptomatic relief when compared our patients with knee OA. 21 For example, after 4 BMC treatments our patients with knee OA experienced 67% total overall improvement compared with our patients with hip OA in this study who reported 72.3% total overall improvement. 21 Pain relief between the 2 studies were similar as well. Our patients with hip OA in this study experienced a mean 69.7% and 69% decrease in resting and active pain, respectively, compared with a mean 84.3% and 62% decrease for patients with knee OA. 21 Comparative studies with larger sample sizes are needed to further examine this topic.
This study is limited by the small sample size, the short follow-up, and the absence of nucleated cell counts. The subjectivity of the measured variables may have introduced response bias. Further randomized controlled studies with larger sample sizes and longer follow-ups are warranted to further validate these results.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MD is the primary physician at Darrow Stem Cell Institute, where all study procedures were performed.
Author Contributions
Conceived and designed the experiments: MD, BS. Analyzed the data: MD, BS, BD, SW. Wrote the first draft of the manuscript: MD, BS, BD, SW. Contributed to the writing of the manuscript: MD, BS, BD, SW. Agree with manuscript results and conclusions: MD, BS, BD, SW. Jointly developed the structure and arguments for the paper: MD, BS, BD, SW. Made critical revisions and approved final version: MD, BS, BD, SW. All authors reviewed and approved of the final manuscript.
