Abstract
While some reports are available regarding metachronous gastric metastasis from renal cell carcinoma after treatment, there are few reports of primary lesion detection based on the diagnosis of a gastric metastatic lesion. The patient in this case was an 80-year-old woman who underwent upper gastrointestinal endoscopy after having developed anorexia 2 months earlier. A submucosal tumor with central umbilication was found in the gastric greater curvature. Endoscopic ultrasonography revealed a solid and hypoechoic mass with hypervascularity on color Doppler imaging that proliferated mainly within the submucosal layer. There was partial exposure of the tumor on the superficial layer. Biopsy was performed, as a neuroendocrine tumor was suspected; however, histopathological findings with immunostaining revealed gastric metastasis from clear renal cell carcinoma. Subsequently, contrast enhanced computed tomography showed right renal cell carcinoma and liver metastasis. Thus, molecularly targeted drug treatment was initiated by the Department of Urology. Our findings indicate that a primary lesion can be identified and prognosis can be assumed based on biopsy of the gastric metastatic lesion. Immunostaining of biopsy samples collected endoscopically could help achieve definite diagnosis.
Introduction
Renal cell carcinoma (RCC) is the third most frequently observed tumor in urology and accounts for approximately 2% to 3% of adult malignant tumors. 1 In approximately 25% of patients with RCC, diagnosis is established at an advanced stage, when there is local infiltration or remote metastasis. The mean survival time of patients with RCC showing distant metastasis is approximately 13 months. 2 Metastatic RCCs may occur in virtually all organ systems, but are mainly observed in the lungs, bones, and liver. Meanwhile, metastasis to the gastrointestinal tract, especially gastric metastasis, is rare. In addition, while there have been some reports on metachronous gastric metastasis after RCC treatment, simultaneous metastasis is extremely rare. 3 We herein report a patient with RCC for whom the primary lesion was inferred based on immunostaining of a biopsy sample of a gastric tumor, and the primary lesion was detected concurrently with gastric metastasis.
Case Report
The patient was an 80-year-old woman. She had developed anorexia 2 months earlier. She visited Fukushima Medical University Aizu Medical Center Hospital with the chief complaints of weight-loss, pyrexia, and general malaise. The mass and tenderness were absent in abdomen. Clinical data had some abnormality. We showed increase of the inflammatory reaction (white blood cell [WBC]: 11 500/µL, C-reactive protein [CRP]: 6.77 mg/mL) and hypercoagulable state (fibrin degradation product [FDP]: 8.7 µg/mL, D-dimer 4.2 µg/mL). Liver dysfunction and renal dysfunction were absent (aspartate aminotransferase [AST]: 11 IU/L; alanine aminotransferase [ALT]: 4 IU/L; blood urea nitrogen [BUN]: 10.2 mg/dL; Cre: 0.66 mg/dL). Although it is strange, microscopic or macroscopic hematuria and proteinuria were absent.
Esophagogastroduodenoscopy revealed a subepithelial lesion with a 10-mm central umbilication in the gastric greater curvature. Although magnification did not show irregular epithelium or vessels at the margin around the lesions, loss of gland ducts was clearly observed on the superficial depressed surface (Figure 1). A solid and hypoechoic mass with hypervascularity on color Doppler imaging that proliferated mainly in the submucosal layer was found on ultrasound mini-probe and endoscopic ultrasound (UM3R and GF-UE260; Olympus Co, Tokyo). A partial exposure of the tumor on the superficial layer was found (Figure 2). A biopsy was performed, as a neuroendocrine tumor or metastatic gastric tumor was suspected.

Esophagogastroduodenoscopy findings (white light and narrow band imaging). A subepithelial lesion with a 10-mm central umbilication with a loss of gland duct on the superficial depressed surface was identified.

Endoscopic ultrasonography findings (UM-3R and GF-UE260, Olympus). A solid and hypoechoic mass with hypervascularity on color Doppler imaging that proliferating mainly in the submucosal layer was found.
Proliferation of atypical cells containing round nuclei and irregularly shaped nuclei with prominent nucleoli was observed using hematoxylin–eosin staining. As atypical cells with clear cytoplasm were observed only in a small region of the tumor, poorly differentiated adenocarcinoma or RCC was suspected. Immunostaining (Figure 3) revealed negativity for all epithelial markers, (CK7, CK20, EMA, CK5/6, p63). Results were also negative for neuroendocrine markers (chromogranin A, synaptophysin, CD56) and mesenchymal markers (c-kit, s100, CD34). While lymphocyte infiltration was found, the immunostaining results were negative for Epstein–Barr virus (EBV)-encoded RNA and latent membrane protein 1 (LMP-1). Meanwhile, the MIB index was greater than 50%. Thus, the tumor was assumed to have a high proliferation potency. It was necessary to differentiate clear cell RCC from papillary RCC and chromophobe RCC. Therefore, testing for CD10, which is a proximal tubular epithelial marker, in addition to pan-cytokeratin (AE1/AE3) and vimentin was performed and the results were negative. Test results for transcription factor enhancer 3 (TFE3) were also negative; therefore, Xp11.2 translocation RCC was ruled out. Based on these results, gastric metastasis from clear cell RCC was most likely.

Histological findings of biopsy specimen: (A) HE, (B) CD7, (C) CD20, (D) MIB-1, (E) Vimentin, (F) CD10. These findings suggested the gastric metastasis from clear cell renal cell carcinoma.
Enhanced computed tomography performed to verify the presence or absence of RCC showed a mass of 7 cm (maximum diameter) with early enhancement in the right kidney. Inferior vena cava invasion was also observed. Early arterial dominant phase computed tomography showed a ring-enhanced region in the liver, S7 (37 × 49 mm) and S8 (28 × 42 mm). Metastasis was suggested for a 10-mm mass observed in the right lung, S9 (Figure 4). The patient was diagnosed with right renal cancer (cT3N0M1, cStageIV), and treatment with axitinib, a molecularly targeted drug, was initiated by the Department of Urology.

Computed tomography showed a large mass with early enhancement in the right kidney (C) with inferior vena cava invasion (B). In addition, metastatic lesion in the liver (A) and lung (D) also observed.
Discussion
Metastatic gastric tumors are rare, with malignant melanoma, lung cancer, and breast cancer being reported as the most frequent primary lesion. The usual sites of metastasis from RCC include the lung, liver, and brain; however, metastasis to the stomach is extremely rare (0.65%). Metastasis routes include hematogenous, lymphogenous, renal capsule, renal pelvis, and ureter routes. Among these, the hematogenous route is the most frequently observed. A study reported metastasis in more than 90% of RCC biopsy samples. 4
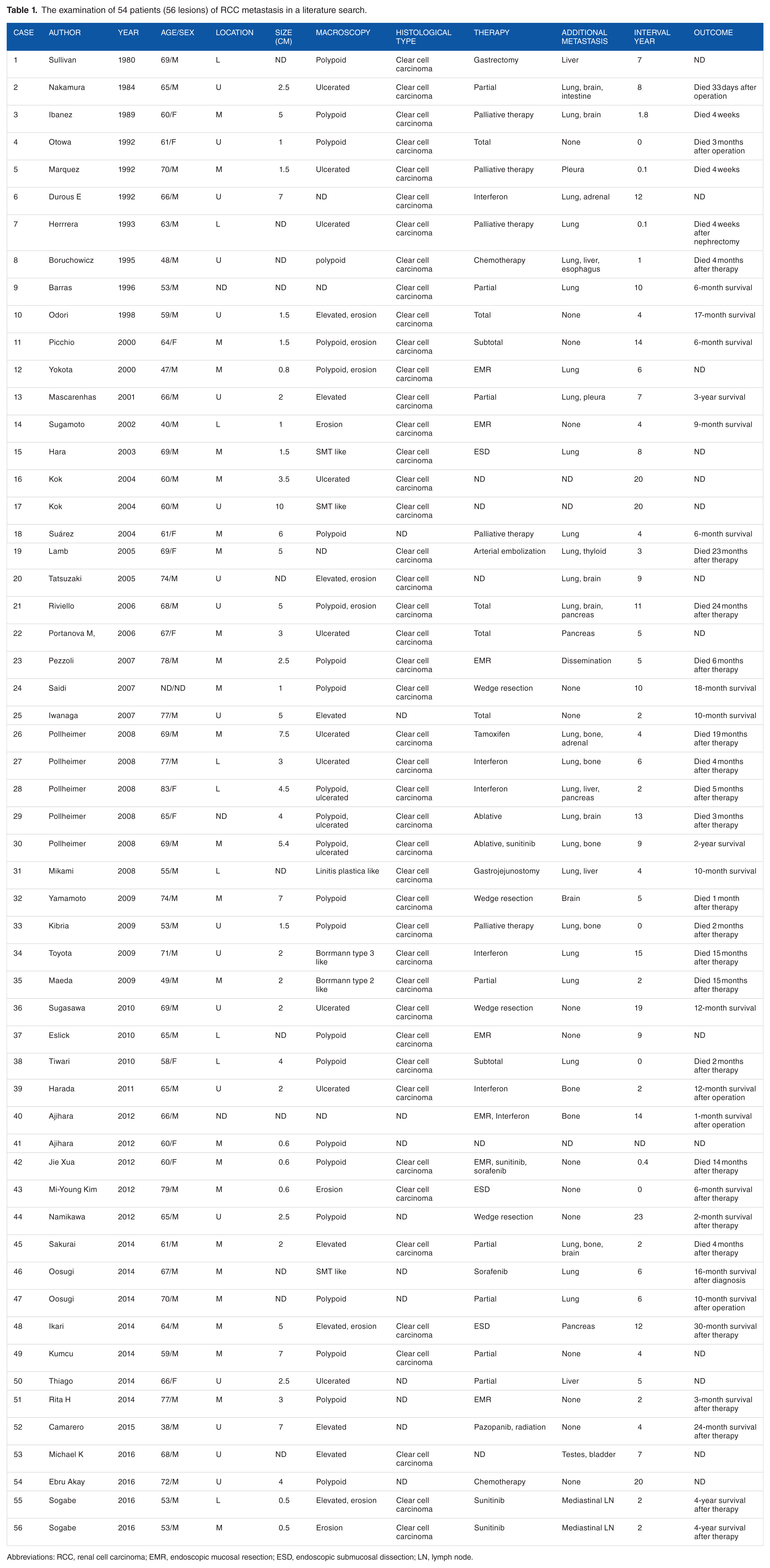
Findings from 54 patients (56 lesions) with RCC metastasis identified on a literature search are shown in Table 1. The mean age of the patients was 63 years, of whom 78% were men. Tumors were most often observed in the middle body of the stomach (44%), followed by the upper body of the stomach (34%). The mean size of the lesions was 3.3 cm (range: 0.5-7 cm). Macroscopic types were varied and included polyp-like lesions, ulcerative lesions, and minor erosion. Ulcerative lesions, which can be called “volcano-like lesions,” were the most frequent. While they were basically hypervascular tumors, a 50-mm polypoid lesion that was found in hemorrhaged tissue from the tumor was also reported. 5 Gastric metastasis from RCC often presents as ulcers or submucosal tumor-like shapes that metastasize mainly to the submucosal layer. The average time from detection of the RCC of the primary lesion to the detection of gastric metastasis is 6.7 years (range: 0-23 years). Thus, they show relatively slow metachronous metastasis. Most reported metastasis cases were detected after nephrectomy; in only 4 patients it was detected at the same time as the primary lesion. 3
The examination of 54 patients (56 lesions) of RCC metastasis in a literature search.
Abbreviations: RCC, renal cell carcinoma; EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; LN, lymph node.
According to the classification proposed by Satomi et al, the growth rate of renal cancer is roughly classified as slow or rapid. 6 Cases with elevated CRP levels, erythrocyte sedimentation rate ≥30 mm/h, γ2-globulin ≥10%, and especially pyrexia have rapid growth, and their prognosis is considered to be poor. Metastatic lesions detected within 2 years after surgery for renal cancer are also classified as rapid-growing. 7 Meanwhile, cases with negative results for the above-mentioned tests are classified as slow-growing. Factors for poor prognosis include protruding gastric lesion, multiple metastases, and gastric metastasis detected within 6.3 years after therapeutic intervention for renal cancer. 8 We retrospectively examined prognoses in patients with metastatic lesions from RCC in previous studies, with a focus on the interval between the detection of the primary lesion and the detection of the metastatic lesion. Among 54 patients (56 lesions), metastatic lesions were detected within 2 years in 15 patients (16 lesions). 3 ,9–16 While therapeutic interventions, including endoscopic therapy and surgical treatment, were performed in all patients, 2 of 3 patients died within a few months after therapeutic intervention for metastatic lesions. In our patients, pyrexia tendency, a high CRP level, and simultaneous detection of a metastatic lesion with a primary lesion were found; hence, a poor prognosis was expected.
Currently, no definite therapeutic strategy for patients with renal cancer with a metastatic lesion has been established. Surgical treatment is recommended as a treatment for metastasis from RCC with an expectation of prolongation of survival time for patients with favorable performance status and a resectable metastatic lesion. However, a favorable prognosis cannot be expected for patients whose tumors are classified as rapid-growing type. MSKCC (Memorial Sloan-Kettering Cancer Center) classification and IMDC (International Metastatic RCC Database Consortium) classification is a classification to predict a prognosis. And it is used for an index to predict the prognosis of the molecularly targeted drugs. According to classifications, our patient was classified in the poor risk in spite of good performance status. So, we must examine it about the adaptation of the invasive treatment carefully. 17 Treatment choices for RCC have recently been increasing, along with the introduction of novel molecularly targeted drugs. Sunitinib, sorafenib, and multi-kinase inhibitors have been used shortly after their introduction, whereas everolimus, temsirolimus, and axitinib have been used recently. More options are now available for the treatment of progressive RCC. Thus, further improvement in the survival rate is expected. In the present case, the patient was treated with axitinib. Small intestinal perforation for peritoneal dissemination occurred after 4 weeks. Therefore, we stopped the molecularly targeted drugs. The patient died 14 weeks after diagnosis.
Renal cancer is characterized by biological characteristics that other malignant tumors do not have. Understanding such features is of particular importance in deciding the therapeutic strategy and evaluating the efficacy of treatment. As with the patient in the present case, a primary lesion can be identified and prognosis can be assumed based on biopsy of a gastric metastatic lesion. Immunostaining of biopsy samples collected endoscopically is particularly important for achieving definite diagnosis of metastatic lesions.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NA and AY managed the patient; NA and AY performed EGD and EUS; AI, GS, AS, YA, YT, YY, TM, RI, SY and TI provided clinical advice; NA and AI collected the data and wrote the paper; AI revised the paper; HH supervised the report; all authors approved the final manuscript for publication.
