Abstract
Renal artery stenosis (RAS) is the leading cause of secondary hypertension. Renal artery stenosis can result in various cardiopulmonary complications mostly through activation of neurohormonal pathways that result in fluid overload and systemic hypertension. We herein describe a 72-year-old man with recurrent rapidly accumulating transudative pleural effusion in a patient with severe bilateral RAS. Patient pleural effusion resolved following stent placement with revascularization of the left renal artery despite absence of improvement of renal function. Patient renal function continued to decline and ultimately treated with fixed hemodialysis.
Keywords
Introduction
Renal artery stenosis (RAS) is the leading cause of secondary hypertension. Atherosclerosis is the leading risk factor. Although the association between the RAS and severe hypertension and pulmonary edema is well known, little has been published on association between RAS and recurrent pleural effusions. We herein present a rare case of 72-year-old man with history of bilateral RAS who developed recurrent rapidly developing pleural effusion treated with repeated pleural taps and chest drain placement. The aim of this article is to discuss the outcome of renal revascularization on the treatment of rapidly reacummulating pleural effusion on the setting of acute renal artery stenosis.
Case
A 72-year-old man with past medical history of type 2 diabetes mellitus, hypertension, and peripheral arterial disease (PAD) presented to the emergency room (ER) with worsening shortness of breath and productive cough of clear sputum. Patient was a known heavy smoker and had extensive PAD including carotid endarterectomy, stenting for bilateral common iliac artery, and superior mesenteric artery stenosis. On arrival to the ER, he was afebrile, hypertensive, blood pressure 195/83 mm Hg, and severely hypoxic. Physical examination revealed diminished air entry to both lungs with normal heart sounds and absent pedal edema. Chest X-ray and computed tomographic scan showed bilateral pleural effusion without consolidation or infiltrate (Figure 1). Initial laboratory workup showed acute renal failure with creatinine 2.8 mg/dL, blood urea nitrogen 63 mg/dL, serum sodium level 137 mmol/L, potassium level 5.1 mmol/L, and brain natriuretic peptide 800 pg/mL and the patient had normal white blood cell count, serum procalcitonin, and troponin. Electrocardiogram showed no ischemic changes. Transthoracic echocardiography demonstrated preserved ejection fraction with estimated ejection fraction 55% to 65%, grade 1 diastolic dysfunction, and small pericardial effusion. Renal ultrasound showed no calculus, mass, or obstruction. Throughout the hospital course, his blood pressure remained elevated. His blood pressure was controlled with 4 different agents including metoprolol succinate 100 mg, amlodipine 10 mg, hydralazine 10 mg thrice daily, and oral clonidine 0.1 mg twice daily. Thoracentesis was performed and fluid analysis was consistent with a transudative effusion that rapidly reaccumulated requiring multiple pleural tap and eventually chest drain placement. The diagnosis of the recurrent pleural effusion remained a dilemma. Patient also had progressive decline in renal function. Further tests included negative viral and hepatitis panels, negative serology for antinuclear antibody, rheumatoid factor, cytoplasmic antineutrophil cytoplasmic antibody (ANCA), perinuclear ANCA, and antiglomerular basement membrane antibody. He had normal serum protein electrophoresis, urine protein electrophoresis, and undetectable level of cryoglobulin. Renal vascular duplex ultrasound and bilateral renal angiogram revealed high-grade stenosis of bilateral proximal renal arteries with 100% occlusion of the right (Figures 2 and 3). Patient had left renal artery stent placement, with severe distal left renal artery disease remaining. Evaluation of kidney function 1 day following renal artery stent placement showed no improvement. Serum creatinine reached 6.05 mg/dL and serum K level was 5.2 mmol/L together with serum Na level 124 mmol/L. Given many metabolic abnormalities and lack of signs of renal recovery, decision was made to start hemodialysis. Right internal jugular nontunneled dialysis catheter was placed. Following stent placement and initiation of first session of hemodialysis, the chest tube fluid output markedly decreased, and by the end of the second day, the chest drain was removed. His blood pressure steadily improved. Both clonidine and hydralazine were discontinued and blood pressure remained adequately controlled. Renal function was assessed on daily basis. Patient urine output remained less than ideal. In addition, his serum creatinine continued to rise off dialysis, indicating lack of clearance. Accordingly, the right-sided temporary hemodialysis catheter was replaced with a permanent right-sided tunneled hemodialysis catheter and patient was started on fixed hemodialysis.

Chest computed tomography on admission shows moderately large bilateral pleural effusion more marked on the R side.

(A) Right renal artery: peak systolic velocity of 218.5 cm/s proximally, 48.5 cm/s at the midportion, and 63.3 cm/s distally. Prolonged systolic upstroke and slightly dampened systolic waveforms throughout the remainder of the renal artery. Ratio to aortic velocity is 1.7. (B) Left renal artery: peak systolic velocity of 197.2 cm/s proximally, 61.4 cm/s at the midportion, and 75.2 cm/s distally. Prolonged systolic upstroke and slightly dampened systolic waveform throughout the remainder of the renal artery. Ratio to aortic velocity is 1.5.
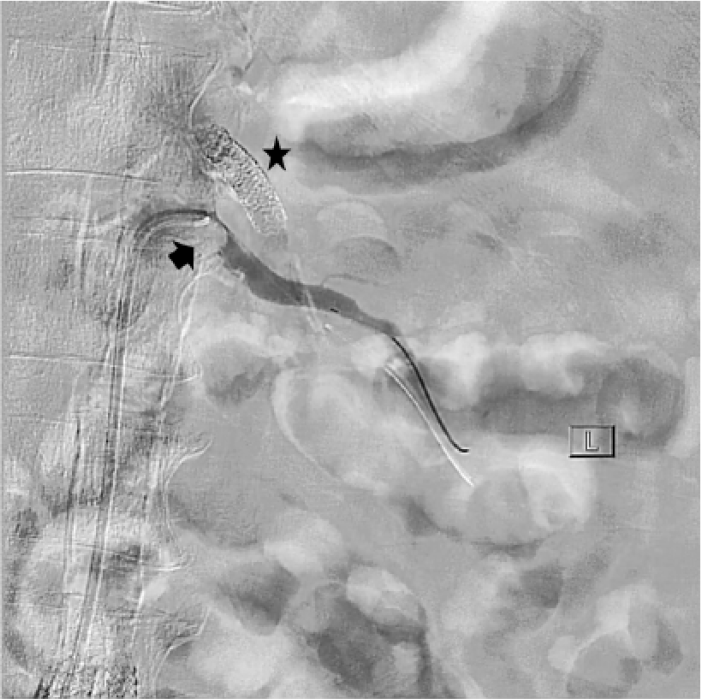
Renal artery angiogram showing severe ostial stenosis of the left renal artery (arrow) as well as previously placed stent along the origin of superior mesenteric artery (asterisk).
Discussion
Renal artery stenosis is identified in around 20% to 30% of individuals with atherosclerosis; it is also the major cause of renovascular hypertension accounting for 1% to 10% of patients who have hypertension. 1
Previous studies have shown a strong association between RAS and severe hypertension, recurrent pulmonary edema, and congestive heart failure.2,3 These complications were collectively grouped under the umbrella of renocardiac syndrome. The association of RAS with rapidly accumulating pleural effusion was rarely reported. To our knowledge, only 1 case report addressed such association. 4
The pathophysiological basis of renal and cardiopulmonary disease is closely linked to each other through various neurohormonal mechanisms. It is proposed that RAS promotes activation of the renin-angiotensin-aldosterone axis causing salt and water retention and peripheral vasoconstriction. All culminate in developing volume overload, hypertension and destabilizing cardiac syndromes. 5 In addition, hypoperfused kidneys have decreased ability to sense elevated systemic pressure causing limited natriuretic response to hypertension thus compounding the volume expansion. 6 Such mechanism explains the development of hypertension, flash pulmonary edema, and worsening of heart failure on the setting of RAS.6,7 However, the mechanism for development of rapidly accumulating pleural effusion on the setting of RAS is not fully investigated.
Akopov et al described a rare association of RAS resulting in rapidly accumulating pleural effusion that was successfully treated with renal revascularization. It was noted that pleural effusion was treated following renal revascularization despite absence of improvement in renal function. 4 To our knowledge, this is the second case report where renal revascularization caused resolution of rapidly accumulating pleural effusion in the absence of noticeable change in renal function. In our case, renal artery stent placement and initiation of hemodialysis were separated by a short time span. Whether the improvement in pleural effusion was secondary to the placement of renal artery stent or because of the initiations of hemodialysis or because of both remains unanswered. It is difficult to attribute the resolution of pleural effusion with certainty to a single factor.
The role of renal artery stenting on treatment of RAS remains controversial. 8 According to guidelines by the Society for Cardiovascular Angiography and Interventions (SCAI), patients with RAS who would benefit from renal artery stenting include those with cardiac disturbance syndromes (acute coronary syndrome or flash pulmonary edema), refractory hypertension, and uncontrolled renal dysfunction. 9 Among patients with these conditions, renal revascularization causes improved renal perfusion, a large loss of sodium and water and resolution of the symptoms and signs of heart failure, as well as improvement in blood pressure and renal function.7,10-12 The role of renal revascularization in treatment of rapidly accumulating pleural effusion needs to be further studied.
Conclusions
This case demonstrates the close relationship between renal and cardiopulmonary physiology. It is highly recommended to screen for RAS among patients with severe hypertension and recurrent pleural effusion. Role of renal revascularization in control of rapidly accumulating pleural effusion remains a puzzle. By reporting this case, we aim to reach to a better understanding of the role of renal revascularization on treatment of pleural effusion.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AA wrote the first draft of the manuscript. MG and DV participated in manuscript development. All authors reviewed and approved the final manuscript.
