Abstract
Peritoneal dialysis has been a widely accepted modality for treating end-stage kidney disease, but a regular dialysis schedule can be seriously disrupted by various comorbid conditions requiring surgical intervention. A 40-year-old woman who had been receiving peritoneal dialysis was sequentially but separately complicated by pleuroperitoneal communication and ovarian cancer. Despite the need for temporary interruption of her peritoneal dialysis schedule, it was successfully resumed after the relevant surgeries for each disease. Several concerns regarding overall postoperative dialytic management strategies, including how to deal with the peritoneal dialysis catheter during the postoperative period as well as how long peritoneal dialysis should be interrupted, which remain an unresolved issue in the field of nephrology, are also discussed.
Keywords
Introduction
Peritoneal dialysis (PD) has been a widely accepted modality for treating end-stage kidney disease (ESKD).1,2 It allows for minimal variation in the intravascular volume status, a reduction in cardiovascular stress, the favorable preservation of the residual renal function, and independence from frequent hospital visits,1–3 offering a rational approach to dialysis treatment in the broader context of overall health care. 4 Occasionally, the regular PD schedule may be seriously disrupted by various comorbid conditions requiring surgical interventions.
In this report, we describe our experience with one such case in a female patient on chronic PD treatment who was sequentially but separately complicated by pleuroperitoneal communication (PPC) and ovarian cancer, which obliged us to interrupt the procedure temporarily after undergoing the relevant surgeries for each disease. Several concerns regarding postoperative dialytic management strategies that emerged in this case are also discussed.
Case Report
A 40-year-old woman who had been treated with PD for approximately 7 weeks presented with dry cough and dyspnea without chest pain in mid-March 2015. Eleven years before, she had been diagnosed with anorexia nervosa accompanied by chronic kidney disease with elevated serum creatinine (Cr) levels of around 3 mg/dL, for which the patient had received sporadic medical care. In 2007, at 32 years of age, she was referred to our hospital and subjected to contemporary and comprehensive renal care when her serum levels of Cr and potassium were 3.84 mg/dL and 3.0 mmol/L, respectively. Thereafter, her serum Cr levels increased gradually, but slowly, with varying states urine of 1+ to 2+ for protein from 3.68 mg/dL in the beginning of February 2008 to a level of 9.04 mg/dL at the end of January 2015, at which time she underwent the procedure. After placing a Tenckhoff PD catheter through a classic transverse surgical incision, PD using 2 daily exchanges (1 L of Midperiq® 135 ×2; Terumo Co., Tokyo, Japan) with a total dwell time of 8 hours was initiated. The patient’s medications at this point included azosemide 30 mg/day, cinacalcet hydrochloride 25 mg/day, calcitriol 1 μg/day, calcium carbonate 1000 mg/day, ferrous fumarate 305 mg/day, and febuxostat 40 mg/day. Her medical history also included left ovarian cysts with a normal serum cancer antigen (CA) 125 level of 15 U/mL, as determined on a gynecological examination at 38 years of age. A regular checkup with transvaginal sonography (TVS) performed 2 weeks after the commencement of the PD treatment confirmed spontaneous disappearance of the left side cysts but newly revealed an enlarged right ovary measuring 40 × 23 mm with cystic formations.
Chest X-ray at the presentation revealed a blunting of the right lateral costophrenic angle, which is suggestive of right-sided pleural effusion (Figure 1A), but she had neither hypoalbuminemia nor any features for congestive heart failure and/or fluid overload. In addition, Technetium-99m-labeled macro-aggregated albumin scintigraphy combined with single-photon emission computed tomography and computed tomography (SPECT/CT) successfully demonstrated the entry of the radiotracer into the right pleural space (Figure 2), encouraging us to make a diagnosis of PPC. We then tried PD with a reduced total dwell time of 6 hours. Her urine output was maintained at about 1000 to 1200 mL/day, but the daily ultrafiltration volume gradually decreased from approximately 400 to 200 mL despite a prompt but transient decline in the thoracic fluid accumulation (Figure 1B).
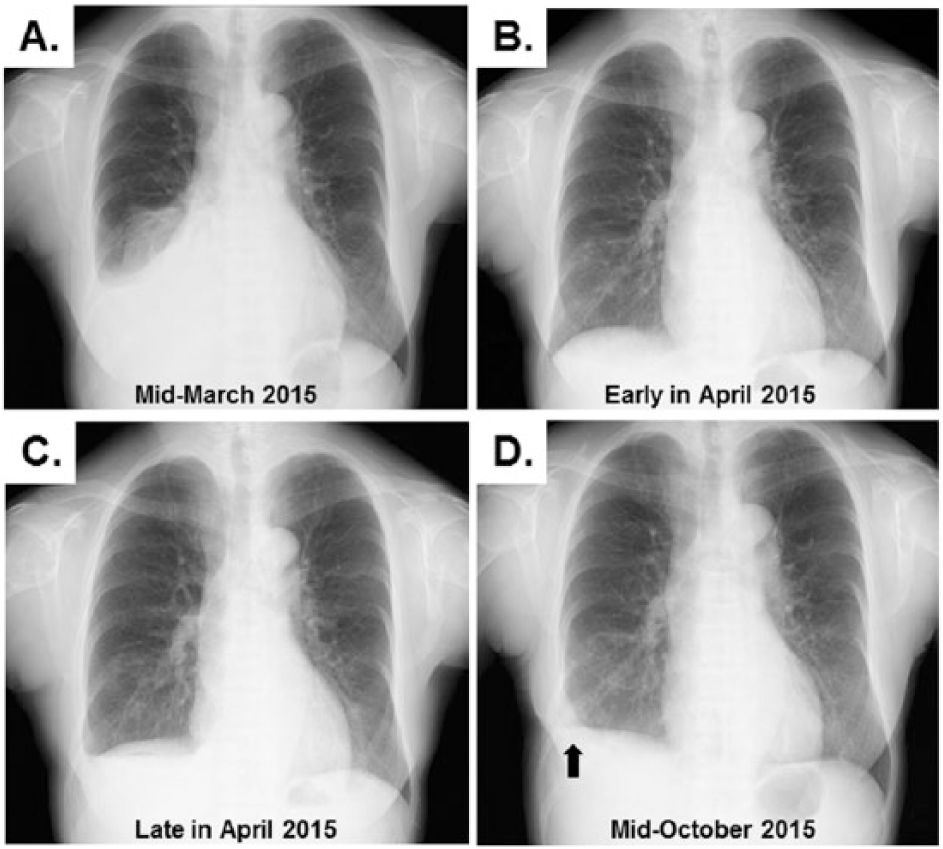
The serial chest X-ray findings during the observation period. The accumulation of fluid in the right thoracic space was shown in mid-March 2015 (A). After switching to a reduced dwell time, the blunting of the right lateral costophrenic angle disappeared in early April 2015 (B). However, the effusion recurred about 3 weeks later (C). Approximately 2 months after the surgical intervention for PPC, fluid collection was no longer detected (D). Note that there is slight tethering of the right hemidiaphragm (arrow), probably manifesting as an anatomical sequela of the surgery.

Findings of peritoneal scintigraphy (A) and SPECT/CT (B to E). An abnormal uptake in the right hemithorax is demonstrated after instilling the tracer into abdomen through the PD catheter (A). Select axial (B and C), coronal (D), and sagittal (E) images of SPECT/CT allow us to confirm precisely the abnormal radioactivity in the region corresponding to the fluid accumulation in the right thoracic cavity (arrows).
At the end of April 2015, the recurrence of pleural effusion (Figure 1C) with an increased serum Cr level of 12.59 mg/dL was confirmed when she complained of general fatigue and appetite loss as well as dyspnea. PD was suspended, and hemodialysis (HD) treatment 3 times per week was commenced through a right internal jugular dialysis catheter. After 4 weeks, during which time she began to feel well along with a decline in her serum Cr level, a forearm arteriovenous fistula was created to provide permanent HD access. Nevertheless, the patient’s mental state started fluctuating, and she strongly requested to resume PD. She eventually accepted surgical intervention for the PPC, so we decided to use PD and HD in combination as preprocedural management strategies. Finally, 5 days of PD (4 exchanges per day; 1 L of Midperiq® 135 ×4; total dwell time of 12 hours) per week followed by HD twice a week led to a favorable volume status, bringing her serum Cr level to 8.43 mg/dL.
At the end of June 2015, video-assisted thoracoscopic surgery combined with right minithoracotomy was performed. Under general anesthesia with separate lung ventilation, the entire right thoracic cavity was carefully examined through thoracoscopy. We identified a few rice grain–sized flaws in the costal part of the diaphragm near the anterolateral part of the central tendon. These flaws were treated with a 3-0 nonabsorbable braided polyester suture in a continuous pattern, and a sheet of absorbable polyglycolic acid felt (Neoveil; Gunze Ltd., Kyoto, Japan) was fixed to the sutured lesion to reinforce the structural stability. 5 The postoperative course was uneventful. Renal replacement therapy was exclusively done with HD (3 sessions per week) for 4 weeks. In this period, the peritoneal catheter was flushed once daily with 500 mL standard dialysate for PD. We then resumed complementary treatment consisting of PD and HD in combination with titrating frequency, exchange volume, and dwell time of the PD, which was well tolerated by the patient. Despite terminating HD in mid-August 2015, there was no recurrence of right hydrothorax (Figure 1D), and her serum Cr levels settled around 9.82 mg/dL with PD using 5 daily exchanges (1.2 L of Midperiq® 135 ×5 [total dwell time of 14 hours]).
At 1 year after surgery for PPC, a follow-up gynecological examination with TVS revealed ovarian cysts with a distinct increase in the size of the right ovary, with the longest axis measuring 70 mm. In addition, she developed an elevated serum CA 125 level of 369 U/mL when about 2 months had passed since she started PD with a modified regimen consisting of 3 cycles of standard dialysate exchanges, each with a 4-hour dwell time, and an 8-hour dwell of icodextrin-based solution (1 L of Midperiq® 250 ×3 and 1 L of NICOPELIQ® ×1; Terumo Co.). Subsequent diagnostic noncontrast-enhanced magnetic resonance imaging revealed prominent cystic mass lesions replacing the right ovary. The patient then underwent exploratory laparotomy, which included total hysterectomy, bilateral salpingo-oophorectomy, and subtotal omentectomy, at the end of July 2016, leading to the diagnosis of stage IA (T1aNXM0) clear cell–type adenocarcinoma in accordance with the American Joint Committee on Cancer tumor-node-metastasis classification and International Federation of Gynecology and Obstetrics classifications for staging ovarian cancer. 6 As perioperative dialysis management, PD was again suspended 1 week before the surgery, and she was placed on the standard HD (3 sessions per week) for approximately 8 weeks. No episodes suggestive of peritonitis were noted, but postoperative hematoma in the pelvis at the hysterectomy site was observed. About 5 weeks after the surgery, disappearance of the hematoma was confirmed when we started flushing the PD catheter as described above. No adjuvant chemotherapy was provided due to the patient’s refusal. In early October 2016, the patient finally became dependent on the complementary treatment consisting of 6 days of PD (1 L of Midperiq® 135 ×1 [3-hours dwell time], 1 L of Midperiq® 250 ×3 [4-hours dwell time each], and 1 L of NICOPELIQ® ×1 [8-hours dwell time]) per week followed by HD once a week. At 10 months of follow-up, there has been no recurrence of the ovarian cancer, and she is still doing well with a daily urine output of approximately 200 mL and serum Cr levels of 11.21 to 11.99 mg/dL.
Discussion
Hydrothorax secondary to PPC is a well-described complication of PD and develops in approximately 2% of all patients with PD.7,8 It may present soon after starting the procedure or after various periods, ranging months to years, of uncomplicated PD. 7 Although the clinical manifestations depend on the magnitude and time of evolution, they often include chest discomfort, cough, and/or dyspnea; however, some subsets of patients with the disease may be asymptomatic despite pleural effusion being found on routine chest radiographs.7,9–11 The mechanism leading to the formation of hydrothorax remains unclear; however, direct communication either through structural defects or laceration of blebs in the fragile region of the diaphragm combined with an increased pleuroperitoneal pressure gradient resulting from abdominal distension has been regarded as the major pathogenic basis for the disease.7–9,12,13 The asymmetric distribution of lymphatics between the right and left diaphragm and the protective effect of the heart and pericardium in covering left structural defects may explain the preponderance of right-sided hydrothorax.7,13,14
Although the workup for the differential diagnosis of pleural effusion in patients with PD is quite similar to that performed in general practice, 15 it should be extensive, and several points may be worth emphasizing. The presence of a pleuroperitoneal leak can be supported by a pleural fluid examination, with indicative findings being an increased glucose level in the transudate and a concomitant normal blood glucose level.16,17 Although the reliability of these findings is limited and there are numerous confounding variables, 13 a pleural fluid-to-serum glucose concentration gradient (PF/S GG) of above 50 mg/dL and/or a pleural fluid-to-serum glucose concentration ratio (PF/S GR) of >1 can be used for the diagnosis instead of the absolute value of serum glucose level.16,17 Alternatively, peritoneal scintigraphy may help confirm such a leak by demonstrating increased supradiaphragmatic radioactivity, as shown in the present patient.7,13,14,18 A permanent shift from PD to HD has been suggested as one therapeutic option for treating PPC.7,8 Other strategies include low volume exchanges and cessation of the procedure for 2 to 6 weeks, although temporary HD may be required as well especially in the subjects with minimal residual renal function.8,13,14 Even short dwell times with or without a dry day may be beneficial.19,20 Surgery or pleurodesis using various sclerosing agents, including talc, tetracycline, doxycycline, bleomycin, autologous blood, and fibrin glue, is reserved for patients who either fail to respond to foregoing conservative management or develop recurrent hydrothorax after a period of obvious success.8,13,14 However, no consensus regarding the optimum therapeutic option has yet been established, with present approaches depending on the patient’s condition and preferences.8,21 Our proposed diagnostic workup is shown in Figure 3.7,8,13–20

Patients maintained on a chronic dialysis treatment are potentially at an increased risk of cancer, which often follows a locoregional pattern, with the kidney and bladder being the most common sites. 22 Epidemiologically established risk factors for ovarian cancer include family history, nulliparity, infertility, and early or late menarche. 23 No specific relationship between ESKD and ovarian cancer has been noted, although cases of ovarian cancer as a comorbid adnexal disorder in patients with ESKD have already been reported anecdotally.24,25 Occasionally, the interpretation of the serum CA 125 level, which has a significant diagnostic impact on ovarian cancer, can be confusing, as it may be a nonspecific indicator of peritoneal irritation, 26 although the diagnosis of the disease in the current case was straightforward after confirming the abnormal imaging findings. Consequently, the clinical scenario of the current patient, characterized by a set of complications including hydrothorax because of PPC and ovarian cancer, may not be surprising. However, the significance of the present case should be evaluated carefully in terms of the management strategies in the perioperative period.
Given the tractive and/or stretching nature of PD characterized by increased intra-abdominal pressure accompanying peritoneal dialysate infusion,14,27 postoperative leakage of the dialysate through the wounds is a matter of concern that needs to be dealt with among patients who receive various surgeries involving diaphragmatic or abdominal wall integrity.8,13,28 None would likely argue against the need to suspend PD after such interventions, at least until the wounds stabilize enough to overcome the increased tension at the corresponding regions resulting from the dialysate dwells. At present, several empirical examples exist in the literature wherein temporary interruption of PD was applied in association with pleurodesis or surgical repair for diaphragmatic defects leading to PPC, followed by resumption at most 8 weeks after the interventions.8,11–13,29 Even restarting PD with low volumes on the operative day has been reported anecdotally. 30 On the other hand, one report has described the impact of gynecologic surgeries on the continuation of PD, reporting on 3 patients who underwent hysterectomy. All patients were shifted to HD in the immediate postoperative period, and PD was successfully restarted after 2 to 4 weeks. 31 Although the optimum regimens for interrupting and resuming PD in this population remains to be standardized, our experiences, which are not very different from the previous clinical practice patterns,8,31 obviously support the opinion that comorbidities in patients on PD necessitating surgical interventions do not necessarily constitute a contraindication of PD. However, whether or not the complementary use of HD and PD with peritoneal resting, 32 which might have reduced the tractive burdens on the operative wounds in the current patient, improves the feasibility and safety of postoperative PD resumption in the ordinary clinical setting remains unknown.
PD catheter dysfunction characterized by various presentations, such as slow fill or drain, catheter tip migration, and obstruction, can interfere with the appropriate dialysate flow required for dialysis.33,34 Traditionally, catheter flushes with dialysate or saline have been performed to prevent blockage and to ensure the functional patency, especially in the break-in period after dialysis catheter implantation, although no prospective study has conclusively shown that this would be beneficial.34,35 Such flushing may also have been practiced with no methodological consensus as a part of the overall postoperative dialytic management strategy among some patients who required transient interruption of the procedure because of various surgical interventions.12,36 We empirically followed this policy as well after our patient received surgical intervention for PPC, but the presence of postoperative hematoma after the gynecological surgery precluded us from promptly initiating catheter flushing to retain a quiescent milieu due to concerns associated with applying mechanical stress to the wounds via peritoneal lavage during the process of stabilization. At present, the validity of the catheter management methods applied to the current patient is unclear due to a lack of substantial information regarding these concerns; however, we may need to focus on the recent proposal indicating that periodic flushing of the peritoneal cavity with dialysate may in fact be irritating and even promote peritoneal reactions with a risk of catheter obstruction through entrapment by omentum tissue.34,37 Of further note is that experiences with the Moncrief implantation technique, in which patients go about their daily lives with the external part of the catheter embedded subcutaneously until it needs to be used, have indicated no increase in the rate of catheter obstruction without peritoneal rinsing. 37 Otherwise, there may be some consensus that flushing the catheter with saline instead of dialysate may be less irritating, although it may theoretically increase the risk of infectious events due to manipulation without any outward draining before infusion. 34
Finally, we believe that the accumulation of more experience with a larger number of patients similar to ours is needed. Gathering further findings will aid in the establishment of optimal overall postoperative dialytic management strategies, including how to deal with the PD catheter during the postoperative period as well as how long PD should be interrupted, which remains an unresolved issue in the field of nephrology.
Footnotes
Peer review:
Two peer reviewers contributed to the peer review report. Reviewers’ reports totaled 293 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a Grant-in-Aid for Research on Advanced Chronic Kidney Disease, Practical Research Project for Renal Diseases from the Japan Agency for Medical Research and Development (AMED).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
TA drafted the manuscript. TY, MK, SN, YK, SI, HY, EN, TM, and TK made contributions to the acquisition of the clinical data. OS, SM, EK, and DN provided a detailed review of the contents and structure of the manuscript, resulting in significant changes to the original document. All of the authors have read and approved the final manuscript.
Disclosures and Ethics
As a requirement for publication, the authors have provided the publisher with signed confirmation of their compliance with legal and ethical obligations including, but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality, and (where applicable) the protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this manuscript is unique and not under consideration for publication or published in any other journals and that they have permission from the rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
