Abstract
Cardiovascular diseases (CVDs), which have high morbidity and mortality, have become one of the world’s largest public health concerns. Although primary hospitalization can partially relieve symptoms, many patients continue to have poor prognoses and lowered quality of life after discharge. Cardiac rehabilitation (CR) is an important recommendation for patients undergoing cardiac surgery, as well as those who suffer from severe cardiovascular events or chronic cardiac disease. However, standard, center-based CR options tend to be ineffective and hard to access, leading to low compliance. The development of digital health technologies (DHTs), especially wearables—which can be combined with mobile applications-has enabled home-based CR, that is, remote CR. This model has substantially enhanced CR efficiency and patient adherence. With advancements in artificial intelligence (AI), including machine learning (ML) algorithms and deep learning, large-scale data from wearables and other DHTs can be effectively retrieved and interpreted. Further incorporation of AI into DHTs may provide real-time fitness telemonitoring, accurate risk recognition and prediction, individualized exercise recommendations, and improved patient adherence. In this review, we succinctly highlight several applications of both AI and wearables in remote CR, and evaluate their collective roles in patient CR execution from several perspectives. We hope our review will help advance further integration of AI and DHT into home-based CR, and plan to direct future research toward refining the use of AI in new-era digital CR.
Keywords
Introduction
Cardiovascular diseases (CVDs) are the primary cause of mortality and morbidity globally. CVDs cause up to 38% of premature deaths (in patients aged <70 years) due to noncommunicable diseases.1,2 Although pharmacologic and surgical advancements have saved many lives, poor prognoses, disease recurrence, and high rates of hospital readmission continue to impact many patients. These high rates of readmission can largely be attributed to insufficient recovery of cardiac function and/or physical capabilities, low follow-up adherence, a lack of real-time outpatient evaluation, and suboptimal treatment plans. Over the past several decades, cardiac rehabilitation (CR) has emerged as an effective strategy to reduce mortality, improve prognoses, and enhance quality of life among patients with CVDs.3-6
CR was first proposed as a component of CVD care in a 1995 clinical guideline. This guideline characterized CR as a comprehensive long-term service consisting of medical evaluations, prescriptive exercise, cardiac risk factor interventions, education, counseling, and behavioral modification. 7 The guideline also emphasized that CR/secondary prevention initiatives should include components to help patients decrease their cardiovascular risk, develop healthy behaviors, and improve medication compliance. If all of these steps were taken, the guideline suggested that CVD patients would have decreased morbidity and be able to pursue more active, healthy lifestyles. In 2007, the evaluations, interventions, and expected outcomes associated with CR were all extended. 8 The application and dissemination of CR in the managements and recovery of patients with CVDs is increasing swiftly due to its significant advantages.5,9 Studies have demonstrated that the improved outcomes resulting from CR programme in a real-world population. 10 The earliest type of CR is center-based CR. However, some patients eligible for center-based CR programmes do not complete the entire recovery process because of low referral rates, low adherence rates, and inadequate socioeconomic resources. 11 The unequal distribution of healthcare resources between different countries, limitations in hospital, high infrastructure demand for CR centers, and high cost of CR also contribute to the relatively low patient patriation rates. 12 Thus, CR programmes that are independent of physical health care centers are desperately needed.
The need for home-based CR has progressively increased since the COVID-19 pandemic, as many patients are unwilling to take the risk of being infected with COVID-19 to attend CR in a hospital setting.13,14 New digital health technologies (DHTs) have also robustly supported the development of remote CR. Remote CR has been shown to increase patient adherence to medical guidelines and promote post-event recovery. It also better evaluates the effects of recovery by monitoring health-related data and providing feedback to patients via DHTs, which can encourage patients to cut down on their sedentary lifestyle habits and increase their daily steps. These activities can remarkably improve quality of life and elevate peak VO2. 15 Wearable devices that track daily activity can also help patients understand the relationships between different physical activities and cardiac responses. Patient appears to be more willing to engage in CR, and to increase the amount and intensity of their activities, when they have access to their personal health data. 16 And previous investigations have exhibited the superior or equal results of remote CR than traditional CR.17,18 However, while the advantages of tele-rehabilitation have become increasingly evident, there are also many challenges. These include inaccuracies and inefficiencies in data processing, the substandard quality of raw data, 19 difficulties in interpreting data, and the demands on cardiologists who must identify and analyze real-time information.
Recent advances in AI technology have helped address some of the challenges related to remote monitoring devices.20,21 There are 2 main subfields of AI: multiple machine learning (ML) and cognitive computing. 1 ML is a feasible way to navigate through complex data. 22 ML algorithms can gather and analyze extensive data from different sources, such as clinical measurements and observations, wearable tools, and environmental information, and create coherent models to predict and assess human disorders. Several previous investigations have underscored the superiority of AI algorithms and assistive robots in cardiology.23-25 AI has been used to optimize clinical decision-making by accurately analyzing and interpreting patient data. 26 AI technology has considerable potential in cardiovascular medicine because of the adaptability and flexibility inherent in the algorithms, the large-scale databases we have available for model training, and wide-ranging technology uptake.27,28
In this context, integrating AI with digital technology during CR could provide real-time data, predict clinical anomalies, aid abnormal cardiological signal detection and diagnosis, personalize exercise prescription, evaluate recovery and increase patient adherence to medical guidance. We believe that integrating AI and digital technology will facilitate the emergence of a new era in CR management.
The Beginnings and Evolution of Remote CR
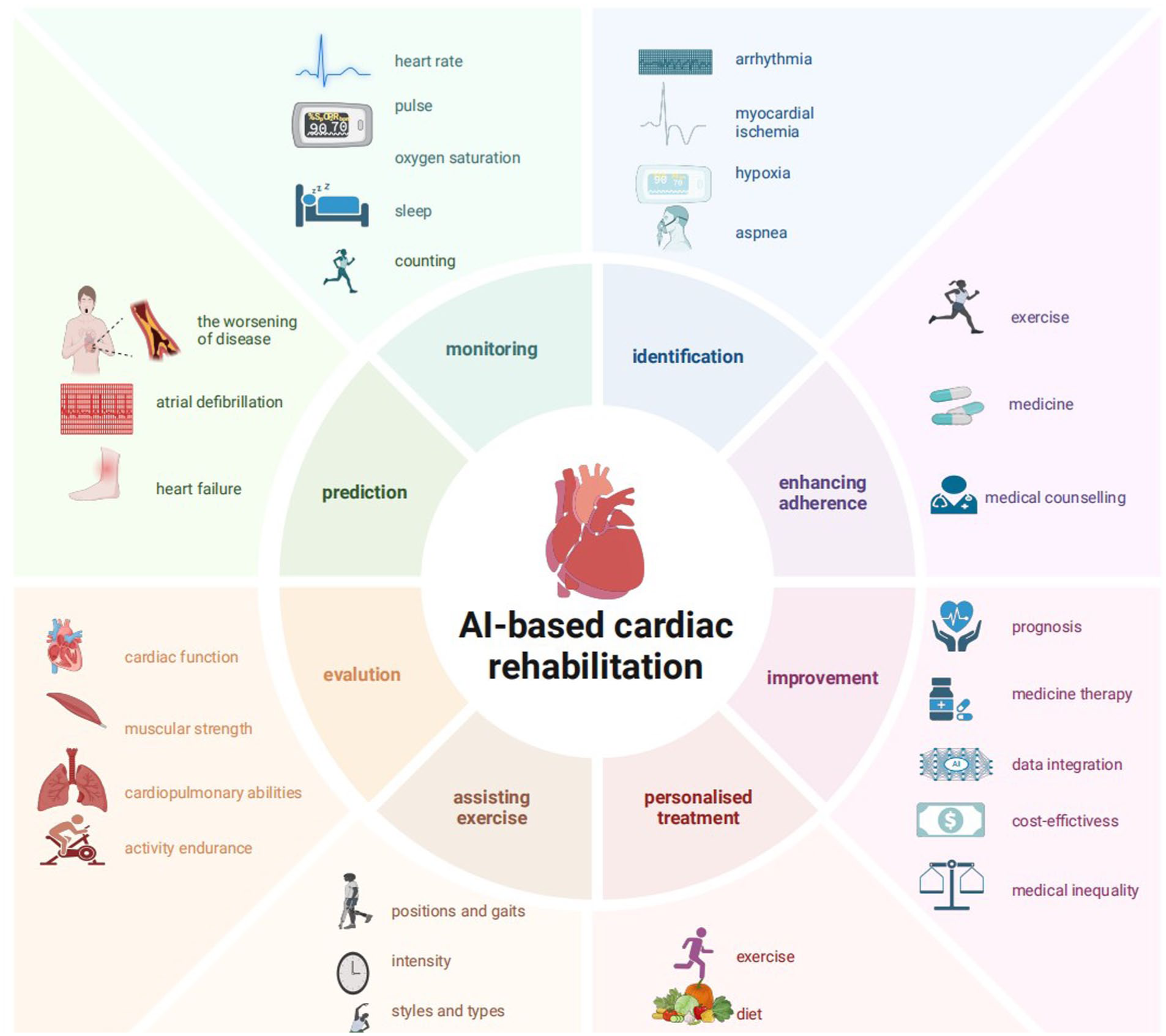
CR is a multidisciplinary programme, and its major components include patient assessment, removal of risk factors, physical activity, the establishment of individualized exercise schedules, dietary counseling, psychosocial management, and vocational support. It is recommended to a broad spectrum of patients, including those who have experienced acute coronary syndrome, those who have undergone cardiac surgery, and those who have stable chronic heart failure (HF) or stable angina.29,30 CR typically includes 3 consecutive phases. Phase I includes inpatient recovery during the primary hospitalization. Phase II refers to cardiologist-supervised outpatient-monitored daily activities for up to 4 months post-discharge. During this phase, patients should consistently engage in exercise, adhere to their medication regimens, and consult with specialists if they need to modify their medical treatment. Subsequently, patients advance to phase III, which is a comprehensive self-management programme. Prior data has substantiated the impact of CR in decreasing hospitalization and mortality rates while increasing quality of life measures.29,31,32 Thus, CR is recommended as an effective intervention for patients with specific CVDs to achieve a better treatment outcome and avoid disease recurrence (Figure 1).9,11,33

The process and evolution of CR. Traditional CR models depend on hospitals or other healthcare centers and are associated with many drawbacks, including geographic limitations, inequitable resource distribution, infrastructure demands, limited data, and the necessity of frequent consultations with cardiologists. Remote CR, which could harness AI and DHTs would overcome the difficulties associated with traditional CR encounters. Various electronics have already been shown to be capable of providing real-time physical data and personalized exercise recommendations independent of complex infrastructure. Patients can obtain medical services from multidisciplinary teams via cloud platforms instead of going to the hospital for medical counseling. The whole CR programme can be divided into 3 continuous phases. Phase I refers to inpatient recovery during the primary hospitalization after cardiac events. Phase II refers to professors-supervised outpatient recovery for up to 4 months after discharge. During this phase, patients can get medical assistance and counseling by multidisciplinary teams in physical, psychological and nutritional conditions. Subsequently, patients advance to phase III, which is a long-term self-management programme in lifestyle maintenance and a constellation of support.
Despite the significant advantages offered by conventional CR in healthcare centers, the adoption rate remains low, likely because of geographic limitations, inequitable resource distribution, substantial infrastructure demands, and the necessity of frequent consultations with cardiologists. Long travel distance to care centers leads some patients to discontinue their CR programmes because of time constraints or inadequate transportation options. The low rates of participation and demands for more convenient CR options have led to the transformation from traditional CR. Specifically, home-based CR, which is sometimes known as exercise-based CR, was developed. The availability of a range of digital telemonitoring devices, including mobile applications, skin-surface patches, and wearable watches, wristbands, and other ECG monitoring devices that harness proprietary algorithms, has collectively facilitated the implementation of home-based CR. Remote CR overcomes the difficulties of traditional center-based CR encounters, resulting in favorable patient outcomes. Multiple wearables can support cardiologists in daily clinical-decision making, and can help provide more personalized therapies for patients in the process of CR.3,34 Remote CR depends on indirect exercise supervision with some telemonitoring devices and cloud-based counseling. Previous evidence has shown that, in the short term, exercise-based CR can reduce all-cause hospitalization and decrease all-cause mortality of people with coronary heart disease (CHD). In the long term, exercise-based CR significantly lowers cardiovascular mortality rates. 31 The popularity of home-based CR has continued to grow because of its convenience and simplicity. Studies have indicated that home-based CR has equal or even superior safety and efficiency in mitigating CVD events compared to traditional CR.17,18,35-37 Patients also express a preference for and satisfactory experiences with home-based CR.26,38
Using DHTs for Remote CR
With the unequal accessibility of traditional CR, there has been increased interest in alternative modes of delivery, strategies to enhance access, and the evidence-based evolution of CR programmes.14,39 The recent development of a constellation of digital devices have helped enable remote CR, as they are portable, simple, and can provide real-time monitoring. These devices rely on technologies, including smartphones, mobile devices, web-based portals, and many other communication platforms and wearables, all of which can help reach more patients and improve compliance. 14 The emergence of digital equipment, also called ‘digital health technology’, has promoted the transformation and development of CR.
Overview of Digital Technology and Common Smart Wearables
Digital technology originated from the fourth industrial revolution and has resulted in great profits throughout society. Over the past few decades, there have been numerous digital technologies developed or co-opted for application in the medical field.40,41 Digital health has grown as a promising approach to harness information and communication technology to facilitate health outcomes. It was originally known as ‘eHealth’, but has now expanded to be ‘a broad umbrella term comprising eHealth, as well as other emerging fields, like the application of advanced computing sciences in “big data”, genomics and AI ’. 42 Digital health devices now encompass a variety of electronic instruments, including smartphones, wearable devices, and social media, as well as environment sensors, all of which facilitate more convenient and efficient remote medical therapy.
Wearables, in particular, represent a promising advancement in digital technology. Wearables are a series of electronic devices with wireless communication skills that are incorporated into accessories or garments worn on the human body. In general, wearables consist of sensors, actuators, and computation components. 43 Miniature, portable, efficient, and diverse in shape, purpose, and application, wearables have proliferated in clinical fields over the past decade.44-46 Wearables can gather patient data by gathering signals and recording them as personalized data. These data can then be analyzed by healthcare professionals to inform disease diagnosis and treatment.
Wearables can be categorized based on several criteria, including application, placement, and power. They can also be categorized by where they are meant to be worn on the body—including head-mounted wearables, upper-body wearables, lower-body wearables, and wrist-held wearables. 44 The common wearables used for CR are depicted in Figure 2. Chest vests, ECG monitors, and skin patches are upper-body wearables that can monitor electrical cardiac activity and hemodynamics. Wrist-held wearables include wristbands and smartwatches, which can continuously track physical activity and heart rate, and smart rings, which can monitor oxygen saturation. Smart socks are an example of a kind of lower-body wearable and can monitor limb activities as well as peripheral blood flow.
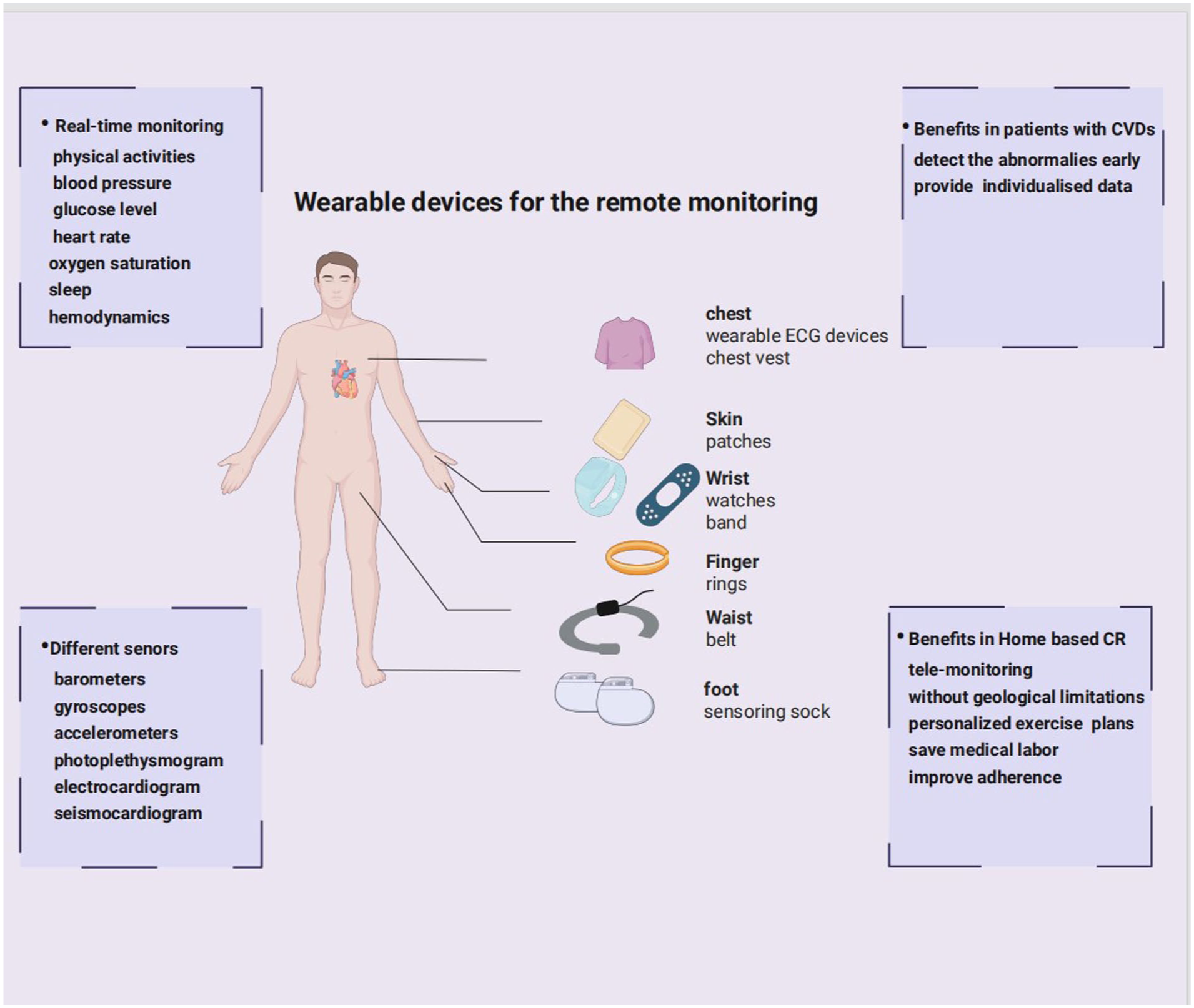
Wearable devices for remote monitoring. There are various wearables available for patients enrolled in home-based CR. Chest vests, ECG monitors, and skin patches can monitor the heart’s electrical activities and hemodynamics. Wristbands and smartwatches can continuously track physical activity and heart rate, and smart rings can monitor oxygen saturation. Smart socks can monitor limb activities and peripheral blood flow.
DHT Application in CR Programme
DHTs such as wearables provide more accurate and timely personal data about patients who are staying far away from the hospital. Lin et al 47 designed a type of smart apparel that incorporated a multi-channel mechanocardiogram. In this system, smart clothing tracked continuous data on cardiac parameters, which was then analyzed and delivered to an associated mobile application. The mobile software received this information and identified characteristic inflection points. Ultimately, the mobile system could predict aberrant cardiac function and identify HF with an accuracy rate of up to 96%. Further, a usability study and technology acceptance test demonstrated that patients, particularly those with CVDs, had positive attitudes about wearing this kind of technology.
In a study exploring the effects of digital devices on CR, initial observations and participant answers indicated a clear preference for and satisfaction with home-based CR, which uses a combination of mobile platforms and wearable technology. Patients using digital devices to guide recovery reported reduced anxiety and improvements in sleep quality. Decreased fat content and increased muscle mass were also noted. However, these devices may lack the capability to deliver precise and efficient interpretations of relevant data. In this system, providers must engage in regular communication with patients and formulate diagnoses based on the supplied information. This process requires a great deal of labor and time, which may diminish treatment efficiency.38,41 However, new AI models may facilitate the identification and interpretation of anomalous results, which could help automatically identify and transmit alarms in real-time and even forecast disease decompensation and diagnoses. 26
Using AI for DHTs
Overview of AI
AI refers to simulated human intelligence, which covers data collection, data reasoning, and data processing and is based on multiple computer systems. ML is a major subfield of AI. ML refers to algorithms capable of generating accurate outputs by identifying and processing vast arrays of data without being programmed or instructed. ML algorithms can also automatically improve and optimize themselves based on accumulated experiences. 48 Ubiquitous ML tools can be divided into supervised learning, unsupervised learning, and reinforcement learning. DL tools are developed based on ML, and both mimic the human brain and can handle multiple-level data. 49 ML and DL are the most ubiquitous computing models and combine an array of algorithms. According the different learning mode, the ML category can also divided into 4 subcategories: supervised, semi-supervised, unsupervised, and reinforcement learning. Supervised learning depends on existing categories or outcomes in the dataset in order to predict related responses, while unsupervised learning aims to discover new interactions among variables without pre-existing labels. 50 The most widely used algorithms include logistic regression, support vector machines (SMM), random forest (RF), and gradient boosting. 51 Reinforcement learning algorithms centered on a constellation of trials and errors. This model is able to gradually train itself by learning from past experience to and better adapt to certain situation, resulting in the best outcomes. 50 DL is extensively used in models for disease prediction. Common DL tools include deep neural networks (DNNs), convolutional neural networks (CNNs), spike neural networks (SNNs), recurrent neural networks (RNNs), and long short-term memory (LSTMs).1,20,46 The characteristic ML process begins with data collection, proceeds to feature engineering, algorithm selection, and model refinement, and ends with model assessment and deployment 28 (Figure 3).

AI categories. There are 2 main subfields of AI: multiple ML and cognitive computing. ML is generally divided into 3 categories: Supervised learning, unsupervised learning, and reinforcement learning. Deep learning are developed based on above algorithms. Common supervised learning algorithms include Artificial Neural Network (ANN), Support Vector Machine (SVM), Random Forrest (RF), and Logistic Regression (LR). Unsupervised learning algorithms include Principal Component Analysis, and Clustering. Reinforcement learning models include distributional reinforcement learning. Finally, deep learning (DL) models include Convolutional Neural Networks (CNN), Deep Neural Networks (DNN), Spike Neural Networks (SNN), Recurrent Neural Networks (RNN), and Long-short term memory (LSTM).
AI Integration Into Digital Health Technology
The incorporation of AI-guided technology into wearable devices is a groundbreaking progress in the healthcare management. Traditional automated ECG interpretation software always showed low accuracy and more error. Recent years has witnessed the remarkable development of AI-based ECG interpretation. With more comprehensive training and precise validation of multiple ML algorithm, the ML-based model inserted into the ECG showed greater accuracy in both diagnosis and discrimination. Additionally, wearable-acquired data can be uploaded to cloud platforms for automated analysis and used to aid in disease prognosis and treatment. The complete data integrating lifecycle includes data collection–uploading information from users or social networks; data processing–filtering unnecessary and redundant data; data transferring, data computing, and data processing-using multiple ML and DL algorithms; data storage-supporting clinical strategies.46,49 Ultimately, AI models could be used to recognize and predict aberrant cardiac activities during the CR process, and send alerts to patients telling them to seek further medical care (Figure 4). A notable study by the Mayo Clinic group 52 used the special algorithm in their smartwatch ECGs to test its precision. The smartwatch analyzed over 125 000 ECGs and the AI-based algorithm could achieve an AUC of 0.885 for recognizing individuals with low LVEF. In addition, in a study by Badertscher et al, 53 the smartwatch assisted by AI algorithm was proved to be inferior in interpreting the results of recorded ECG wave of AF patients in comparison to the doctor. Desirable results were also founded in Diego et al’s 54 experiments. They inserted the AI-based algorithm and manufactures’ algorithm into 5 wearable watches to analyze the rate of inconclusive tracings and the diagnostic accuracy for the detection of AF. As a result, although there is no significance about the accuracy between 2 algorithm, the AI-based algorithm remarkably reduced the rate of inconclusive tracings. These results demonstrated that with the AI application in wearables, the large-scale data was supposed to be addressed more orderly and precisely, more cardiac conditions that are asymptomatic yet potentially life-threatening are likely to be monitored and alerted in an early stage.

Incorporating AI into DTHs during cardiac rehabilitation. DHTs, including wearable devices and inserted sensors, can be worn or inserted into the body. These devices are capable of monitoring individuals’ daily physical activities, vital signs, and cardiopulmonary data. The data collected could be transmitted into mobile applications or wearables. Algorithms could be programmed into these devices to analyze data inputs and develop as well as train themselves. Ultimately, AI models could be developed to recognize and predict aberrant cardiac activities during the CR process, and send alerts to patients telling them to seek further medical care. Health-care providers could also accept the abnormal information from cloud platform. The patients could obtain the related suggestions for CR by online communication or medical visits. Then the patients’ conditions could be re-evaluated and given new and individualized therapies. Besides, the new information and medical data also facilitates the ML model optimization, simultaneously assists digital devices to generate more suitable exercise plannings.
AI Incorporation Into the CR Programme
The development of AI technology allows CR to enter a new era via multiple perspectives.55,56 Firstly, ML models could help care providers to encourage more patients to engage in the CR programme. It was documented that RF models based on the conditional inference trees could forecast the intention of individuals to participate in CR with high accuracy. The findings ascertained that individuals’ ‘willingness to consider the treatment’, ‘perceived self-efficacy’ and ‘perceived need for rehabilitation’ played an important role in motivation, while the ‘barriers to engagement’ and ‘demographic and medical factors’ were less important, which indicated that maybe the barrier is not the main reason for the low participation. 57 Apart from the motivation, good adherence to CR activities and behaviors partially ensure the success of CR implementation. Dimitris et al 58 combined supervised and unsupervised learning to create the model to predict patients’ long-term compliance to CR with a precision of 0.80. They divided individuals into 3 clusters and researched characteristics and clinical data respectively. Patients’ basal features and behavior in the early phase of CR could facilitate the precise prediction. Surprisingly, the patients with low risk and active physical activities continued to be non-adherent individuals in the long-term, because they think they have reached the enough exercise and there is no need for them to be involved in the CR programme. Above findings may suggest the different intervention for these patients. Also, ML could be developed to identify high-risk patients, detect arrhythmia, aid the diagnosis, forecast the prognosis of individuals after adverse cardiovascular events, which promote the CR progression. Romina et al 59 examined multiple ML techniques for attaining the performance of different models in predicting the cardiovascular risks and CR probabilities. In the end, they concluded that the XGBoost algorithm process the highest property in predicting the CR possibility with a normalized mean squared error of 0.008 and an R2 to 92%. Next, the Shapley additive explanations (SHAP) demonstrated that the adherence, oxygen consumption as well as metabolic equivalent task are the most indispensable factors to the CR level and CVD risk. Ismail et al 60 proposed a deep learning model for detecting and classifying arrhythmia using 10-second lead I ECG recordings and achieved high accuracy in recognizing abnormal heart rhythm among various populations in real-world noisy environments, Christina et al 61 developed the model with random forests and neural network algorithms to select the features that have a close relationship with mortality such as age, maximal exercise capacity, accurately predicting the long-term all-cause mortality of CAD patients who are referred to a CR programme. Also, the Linear regression model was used to predict the health care cost in patients with ACS who have undergone the exercise-based CR programme. 24 In Alina et al’s 62 study, They used decision tree induction with random forest feature selection and under sampling to refine models and select the best-performed model to predict the outcomes of patients in the process CR based on the cholesterol level and hypertension, level achieved in 6 minute walk test, and Hospital Anxiety and Depression Score (HADS) anxiety score and HADS depression score, which enhanced the necessity of personalized CR planning evaluated by the risk factors rather than the generalized strategies. Likewise, different algorithms were tested to find the best models for the prediction of outcomes in different treatment paths and help cardiologists to make a better choice about the unique prescription. Furthermore, the Lasso regression model exhibited an average classification accuracy on test set of 0.935. 63 RTW(Returning to work) after CR is also an important part for individuals, especially for those young patients. So the prediction for factors contributing to the RTW is salutary for the optimization of CR goals. Prior studies have utilized different ML techniques to predict the RTW, and found out the best AdaBoost model with 92.4% of AUC. Besides, through these analysis, the team explores that the most common contributors in the models were self-funding, return to drive, age and so on 64 (Table 1).
Machine learning in the field of CR.
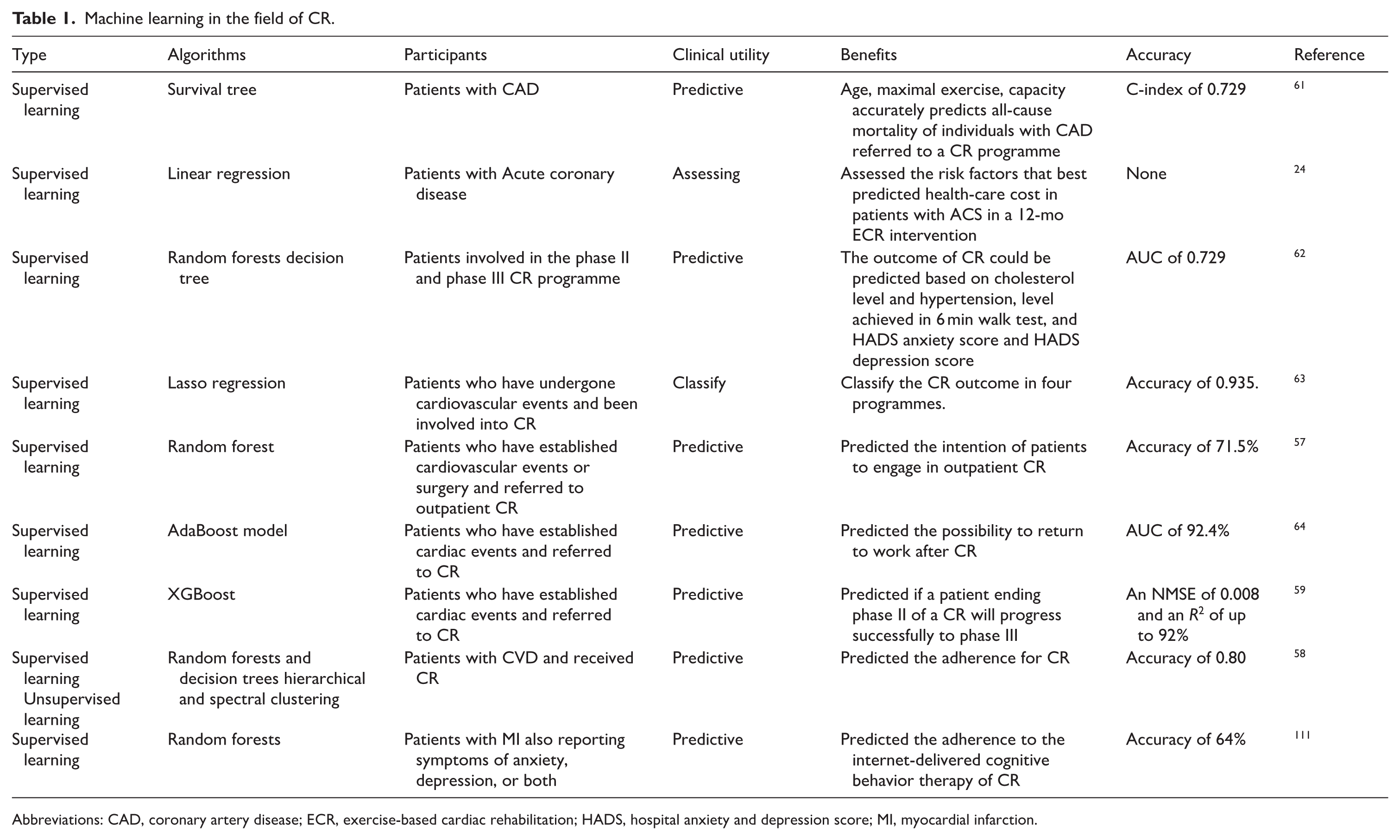
Abbreviations: CAD, coronary artery disease; ECR, exercise-based cardiac rehabilitation; HADS, hospital anxiety and depression score; MI, myocardial infarction.
AI Integration Into DHT for the Implementation of CR
Standard CR models involve regular exercise, dietary counseling, weight loss, blood pressure management, smoking cessation, and psychosocial regulation. Although telerehabilitation has not yet been incorporated into standard care protocols, contemporary digital devices have been shown to benefit CVD patients.65,66 Because of the multifactorial complexity of CVDs, home-based CR also faces great challenges and has not yet been widely adopted. With various AI algorithms applied in the digital remote monitoring devices, some of the current issues associated with CR would be overcome by providing consistent event surveillance, accurate risk prediction and stratification, individualized exercise prescription, better prognosis and high patient adherence (Table 2).
The integration of AI and digital devices into CR.

AI Integration Into DHT for Surveillance and Identification of Cardiovascular Events
Real-time monitoring and early detection of arrhythmia recurrence are of high importance for patients undergoing anti-arrhythmia surgery and start CR stage. The 2 common sensors to record heartbeats are photoplethysmograms (PPGs) and ECGs. ECGs sense the heart’s electrical activities, whereas PPGs detect the blood flow and pressure from the skin 67 Because wearable ECG and PPG devices are convenient and highly accessible, they have been widely used for the classification, prediction, and identification of arrhythmias, including supraventricular arrhythmias, ventricular arrhythmias, atrial fibrillation (AF), and cardiac pacing. 68 Some remote monitoring devices can even enhance the functionality of implantable cardioverter defibrillators (ICDs) and reduce the risk of inappropriate shocks in patients with HF. 69
Because of high accuracy in efficient diagnoses and evaluations, AI has been increasingly integrated into telemonitoring devices.34,70,71 The AliveCor Kardia device, for instance, a type of smartphone/tablet handheld single-lead ECG apparatus, is able to capture patients’ ECGs and automatically interpret the results using an algorithm with a reported sensitivity of 98% and specificity of 97% for AF. The AliveCor Kardia device has also been shown to enhance early diagnoses of AF and reduce the incidence of ischemia and transient ischemic episodes secondary to AF or other factors. 72 An apple watch with inserted CNN algorithm could show great episode sensitivity of 97.5% and duration sensitivity of 97.7% in AF monitoring, which was equivalent to the invasively golden standard-insertable cardiac monitor in effect. 73 These findings suggest that incorporating AI into smart devices can enhance the management of and timely treatments for patients with arrythmias after they are discharged from the hospital.
Other data has also delineated the accuracy and efficiency of AI models in the cardiology space. 74 Progressive advancements have been made in the algorithmic processing of sensor data. For ECG wearables, implemented AI algorithms could enhance reliability in identifying abnormal cardiac electrical activities and may be especially beneficial for patients who are not aware of certain arrhythmia such as cardiac fibrillation or tachycardia. This will help alleviate monitoring burdens amongst both caregivers and patients. 45 Both chest-based and wrist-based ECG device-driven algorithms have been investigated for arrhythmia screening and displayed high-level performance. A wristband-supported single-lead ECG constructed with a deep CNN algorithm exhibited 100% specificity and positive predictive value irrespective of posture and exercise. An AI-driven wristband could also accurately discriminate AF from sinus rhythm. Its accuracy in comparison to the gold standard measurement system was 96.49%, 98.25%, and 98.25% in the supine position, the upright position, and after exercise, respectively. 75 In another trial, a type of chest-worn ECG monitor was designed for AF identification. The recordings were transferred to a smart application and then transmitted to a cloud service for algorithm-controlled analysis. This system resulted in a subject-based accuracy of 97.5%, a sensitivity of 100%, and a specificity of 95.4% compared with 3-lead Holter ECG. 76 Integrating AI and wearables could help drive early intervention and improve patient outcomes. Early detection and precise interpretation of AF would also aid in preventing disease progression. 77 Saeed et al 78 applied LSTM RNNs algorithms in wearable ECG devices to continually monitor cardiovascular abnormalities. The LSTM algorithm showed excellent accuracy in ECG classification for wearables while minimizing computational costs and time expenditure. Another wearable ECG device implemented the SNN algorithm and achieved comparable accuracy in classification compared to previous work. Strikingly, the required energy consumption was significantly lower than that of prior models, and it was more compatible with wearable devices. 79 In the future, wearable ECGs for arrhythmia detection will be developed to become more minimized and use fewer leads. Various algorithms will be trained and developed from larger datasets, which will enhance the speed of clinical decision-making.
AI-Enabled DHTs for Risk Stratification and Prediction
The prediction and risk stratification of CVD patients is an essential aspect in the CR process, promoting the early recognition of severe disorder and better optimizing the CR therapy. Previous literature suggests that ML algorithms, and especially DL algorithms, are increasingly prevalent in AI models that are used to predict and classify CVD risk.24,49,80 Chang et al 81 applied an LSTM model with multi-labeling capabilities to identify STEMIs and other 12 cardiac rhythm irregularities. With regard to STEMI recognition, further real-world testing demonstrated the superiority of the LSTM model over other predictive measures. Wearables also offer enhanced benefits in patient identification and stratification when inserted into mounting algorithms.20,48 Another study found that using a CNN algorithm enabled single lead ECG, which led to the prediction of patients with left ventricular ejection fractions (LVEFs) of 40% or lower, could help screen asymptotic patients for HF. Electrical cardiac activities can also be recorded during a stethoscope examination and then transmitted to smartphone app as for prompt AI analysis and model improvement. 82 Sau et al 83 developed an actionable, explainable and biologically plausible AI-ECG platform including 1 main model and 7 submodels which are validated by five cohorts from a wide range of patients. They revealed that AI-enabled wearable ECG platform showed greater performance in predicting risks of all-cause mortality, future ventricular arrhythmia, future atherosclerotic cardiovascular disease as well as future HF in comparison to authoritative risk assessment system. It could accurately produce the unique patient-specific survival curve and forecast the mortality at different time point.
AI-Empowered DHTs for Individualized Exercise Prescription
Although traditional CR plans demonstrate good outcomes, they may not always be suitable for every patient under same condition as the difference of individual endurance and complications. Thus, personalized and precise exercise prescriptions are needed. The development of AI-integrated DHTs has helped address this barrier. Oliver et al 84 found that, compared with conventional robotics, feedback-controlled robotics, which harness more optimized algorithms, increase training intensity among patients with a history of stroke. They also increased peak cardiopulmonary performance with no increase in rates of adverse events. Literature has suggested that robot-assisted gait therapy is safe for CR after cardiac surgery. It enhances exercise endurance and muscular strength without increasing risks for infections or other adverse events and generate tailored exercise. However, the most satisfying results were obtained after early intervention. 85 In contrast to traditional CR, robot-assisted therapy requires patients to cooperate rather than impose training plans. After being incorporated into 3 strategies, consisting of impedance control, adaptative control, and visual feedback, the robot could sense patients’ contribution to the training and adapt their pattern of training, providing the appropriate associated information to patients via images or videos. This modality could encourage patients to engage in recovery training and provide them with more personalized treatments. 86 Likewise, social robot is another type of AI robot that is mechanically embodied with human or animal features and enabled with social and emotional intelligence. This robot can facilitate remote patient monitoring in cardiovascular treatment by analyzing patients’ situations and creating more personalized recovery strategies. 23 A real-world study found that, compared with conventional CR, robot-assisted CR demonstrated superior patient adherence, physical activity, and cardiac function outcomes. 25 The flourishment of AI technology will ease the difficulties for professionals to manage mount of patients with limited capabilities of exercise and facilitate the personalization of training.
General and specialized exercise training are 2 essential components of remote CR and facilitate enhanced recovery from CVD events.87-89 AI-integrated wearables allow continuous and relatively objective patient data to be monitored. One recent trial used multiple ML algorithms to establish an optimized model to recognize exercise and repetition counting during local muscular endurance (LME) exercise-based CVD rehabilitation. A solitary wrist-mounted sensor was used to collect synchronous data, which were then used to train, validate, and test ML models. The researchers found that using CNN algorithm allowed the telemonitoring sensors to achieve an overall accuracy measure of 96.89%, and the SVM model attained a high degree of accuracy in exercise recognition. Another group found that the CNN model had 90% accuracy in repetition counting without the requirement for peak detectors. 90 The CNN model was also used to extract distinctive features from workout data collected from forearm-mounted wearable devices. It was capable of identifying and classifying 50 gym exercises with 92.1% accuracy. 91 The CNN model-controlled smart sensor allowed for more accurate fitness monitoring, making home-based CR to be more convenient and feasible. To better collect and analyze the large amounts of data from wearables, a multipath CNN (MP-CNN), which combined dynamic CNN (D-CNN) and state transition probability CNN (S-CNN), was established to evaluate and classify exercise activities and nudge patients to correct their actions based on the evaluation. 92 In addition to these examples, the SVM and RF algorithms have been inserted into wearable inertial sensors to classify and analyze limb movement. A combination of acceleration and gyroscope sensors was found to enhance the algorithm-classifier’s accuracy, optimizing movement categorization during rehabilitation. 93
In addition, certain ML algorithms can automate workout prescriptions and enhance their functionality through daily user-uploaded data. They can also make adjustments based on a patient’s current status and establish appropriate exercise goals. 94 Wearable DHTs contain various physical activity or fitness trackers that utilizing the wearable sensors, often worn as a wristband, patches, rings, medical ear buds, fitbit-flex or embedded in a smartwatch or cellphone that collects information of individual daily life trajectories such as heart rate, respiratory frequency, sleep quality and physical activities and produced the optimized individual training plannings.50,95,96 For instance, smart software can record the real-time physical activities of individuals and craft personalized health advice. Related data can also be transmitted to platforms where clinicians can monitor patients’ health indices and make timely treatment adjustments. 13 For instance, the fitbit-flex have the stronger capability to capture free-living physical activities with accurateness and timeliness, even than conventional 6MWT, promoting the designation of individualized physical recuperation. 96 Some health-related smartphone apps can also assess patients’ physiological parameters and formulate personalized exercise regimens, establishing target heart rates, safe heart rates, exercise frequency, intensity, and duration. These digital tools also elevate cardiopulmonary endurance and sports capability of patients with coronary heart disease. A previous study showed that using AI technology promoted improved lifestyles and decreased risk factors such as hypertension, hyperglycemia, anxiety, and depression. 97 In another study, home-based rehabilitation using AI and DHTs offered patients a more tailored training regimen compared to inpatient rehabilitation, enhancing heart rate responses to physical stressors and physiological reserve. Furthermore, wearable-measured heart rate responses better measured the effectiveness of individualized exercise plans among patients going through open-heart surgery. 98 Further study and exploration are needed to optimize the use of AI combined with DHTs in personalized exercise regimens during CR.
AI Incorporated DHTs for Cardiorespiratory Evaluation and Prognosis Improvement
Previous data have indicated the efficacy and feasibility of using AI tools to help wearables measure physical fitness and integrate data during CR.99,100 An experiment has designed effective ML models using the input feature of chronotropic responses in an effort to visualize the functional capacity and track recovery in a CR population during a standardized 6-minute walking test (6MWT). In 129 patients with heart failure, the ML model combined with wearable ECG findings displayed a mean absolute error of 42.8 m (±36.8 m) in comparison to the actual 6MWT distance (6MWD). The 3D model also allowed for the visualization of the relationship between chronotropic response, effort, and 6MWD, paving the way for a transition from hospital-based CR to home-based CR. 2 Chronotropic incompetence (CI) significantly affects the prognosis of HF patients, but identifying and evaluating CI is relatively difficult. The clinically employed method for assessing CI is dynamic exercise testing. Surprisingly, using a wearable recording system (Holter-Actigraphy) easily documented heart rate changes during daily physical activities, analyzed the correlation between 6MWT distance and the percent maximum heart rate achieved during exercise testing, and reduced physical activity intensity in patients with abnormal CI. The wearable recording system thus provided simple methods for detecting HF patients with CI without the complex infrastructure needed for traditional exercise testing. 101
Wearable ECG devices equipped with AI algorithms provide convenient and synchronous patient data, allowing providers to receive more accurate information and, keep in close touch with individuals and learn about their condition to deliver more accurate guidance for recovering. For example, a randomized and controlled clinical trial indicated that patients with stable coronary artery disease at moderate cardiovascular risk who accepted home-based CR achieved higher heart reserve compared to those who underwent hospital-based CR. The NUUBO® system uses Bluetooth wireless technology and AI-based wearables and enables patients to minimize hospital visits while also enhancing treatment adherence and improving cardiovascular performance. 36 Besides, a meta-analysis incorporating 24 randomized controlled trials showed that mobile-based telemonitoring significantly decreased all-cause hospitalization and all-cause mortality of chronic HF patients. 102 The combination of AI and digital strategies offered improvement of outcomes among HF patients and promote the better management of CR.
The restoration of cardiopulmonary function is a crucial metric for evaluating CR outcomes. It is documented that cardiopulmonary exercise testing (CPET) are essential in exercise prescription in the CR programmes due to its decisive role in the targeted heart rate at anaerobic exercise. Therefore the comprehensive assessment and prediction of cardiorespiratory fitness is helpful for the CR prognosis. While CPET is the golden standard in cardiopulmonary function evaluation, its high requirement for the equipment and large cost impede most people from taking part in the CR programme. So a series of formulas have been proposed to calculate the relative parameters of CPET, yet most of them lack precision. Nowadays the application of AI into the CPET shows great potential in automated detection of ventilatory thresholds, improved reproducibility of physiological measurements, assessment and prediction of data by CPET, and identification of abnormal exercise responses. In Zignoli et al’s 103 study, they tested the CNN-based tools for predicting CPET parameters. And results showed that the AI algorithm was able to automatically detect the first and second ventilatory thresholds in CPET with an average mean absolute error of 178 (198) mlO2/min (11.1%, r = .97) and 144 (149) mlO2/min (6.1%, r = .99), which showed great capabilities in comparison to authoritative experts. Next, they conducted deeper research to challenge the regression, generation and explanation of processing CPET data. They used a CNN algorithm adapted for multi-variable time series for the regression of exercise intensity and domain, a conditional generative adversarial neural network for producing fake but realistic combinations of CPET variables, Shapley’s value for evaluating the significance of each variable. The AI model showed high accuracy in classifying exercise intensity. Another study conducted by Atsuko et al 104 designed and tested a ML model and finally verified that the mean absolute error of the targeted heart rate predicted by the ML-based model was 7.7 ± 0.2 bpm, which was more accurate than those of the widely accepted equations. Portella et al 105 utilized the RF-based algorithm to create the multi-classification models to discriminate the abnormal exercise intolerance led by heart, lung or other organs from normal exercise, and the AUC for recognition in cardiological exercise limitation is approximately up to 0.9. A randomized clinical trial was recently conducted to evaluate the effects of home-based CR for CHD patients. In the experimental group, patients were required to download a smart mobile application developed and wear heart rate belts. The researchers learned about patients’ conditions via a cloud platform and communicated with them. At the end of the trial, the intervention group showed superior cardiopulmonary exercise ability after CR. 106 The maximum oxygen consumption measurement during maximal exercise is the gold standard for assessing cardiorespiratory fitness (CRF). Notably, the proposed algorithms were able to directly estimate the CRF of an individual in free-living conditions with accuracy equivalent to laboratory measurements. 107 Oxygen uptake dynamics in response to new metabolic demands are governed by an intricate cooperation of the cardiovascular, respiratory, and muscular systems. Thus, oxygen consumption dynamics could reflect cardiopulmonary function during exertion. Data recorded during daily activities using a hip accelerometer, heart rate monitor, and respiratory bands were extracted and analyzed using a validated RF model. This AI model could determine aerobic system kinetics by integrating related information. 108 These results imply that the combination of AI and wearables augmented the assessment of cardiopulmonary function during CR. Likewise, a qualitative and quantitative meta-analysis indicated that, among patients with cardiac disease, those using wearable physical activity monitoring devices showed improved cardiorespiratory fitness compared with non-wearable users in the maintenance phase of CR. 109
AI and DHTs for Improvement in Long-Term Adherence to CR
Many patients with CVD suffer from psychiatric disorders, including anxiety and depression. In addition to conventional in-person treatment, Internet-based cognitive behavioral therapy 103 may help improve mental health symptoms. However, compliance is a major issue. A group of patients discontinue treatments for a variety of reasons. Intelligent computing, including AI and complex modeling, has been increasingly used to develop individualized rehabilitation plans and increase patient compliance. 110 Prior research has shown that ML models can predict the factors that drive patient adherence. In addition to self-reported cardiac-related fear, sex, and written behaviors can help predict adherence, thereby guiding future AI-driven psychological strategies to enhance compliance among patients with comorbidities. 111 In a previous study, in an attempt to decrease high CR dropout rates, an AI-based toolkit, combined with the Well Beat system, was implemented within a CR programme. This system harnessed multiple AI models to provide personalized recommendations and smartphone messages to encourage patients to continue with the CR programme. At the 6-month follow-up, patients using the Well-Beat system were more likely to complete the CR course, and dropout rates were significantly reduced. Clinicians also provided feedback for the AI system and demonstrated that the approach, self-efficacy, maturity, and main driver barrier offered by the engine were correct over 90% of the time and that the precision of the barrier parameter was approximately 86%. 112
Robert et al 113 recently developed an Internet-based CardioFit programme to customize physical activities for patients with CHD after hospital discharge within a timeframe of 6 to 12 months. They found that, compared to groups provided with usual care, those using the CardioFit programme had improved physical activity and higher health-related emotional well-being scores. These results suggest that AI and digital gadgets could potentially increase the number of patients who are willing to participate in CR and thus broaden its accessibility. 38 Apart from the CHD, the mobile application-guided and wearable ECG device-supported CR was also substantiated outperformance for the patients with AF and going through catheter ablation surgery. Strikingly, patients in the telerehabilitation group had improved physical abilities, higher self-efficacy, and health-related belief scores and more closely complied with the CR programme. The comprehensive telemonitored groups also had decreased AF recurrence after 3-month follow-up. 114 Home-based CR programmes with integrated digital and AI-supportive devices could help patients create a routine for physical activity, which would enhance long-term adherence to care and rehabilitation. Those devices also allow the clinical team to promptly understand data, facilitating quick patient feedback to patients via cloud platform, so enhancing patient compliance with their rehabilitation process and boosting their awareness of their health status.
Challenges and Clinical Perspectives of AI-Based Digital Health in CR
Technical and Clinical Challenges in AI-Driven DHT
Although the FDA (US Food and Drug Administration) does not classify smartwatches as medical devices, smartwatches are regarded as auxiliary equipment’s that are approved more quickly under the Digital Health Software. And some AI models for ECG interpretation, such as the Mayo Clinic’s CNN-based model for recognizing individuals with low EF, have already been authorized by FDA in clinical practice.53,82 Clinical evidence has showed the comparative or even a little superior effects of smartwatch in monitoring events, strong evidence that their use translates into better hard clinical outcomes, such as decreased complications or mortality, and multiple-center RCT researches are still lacking. Besides, the AI algorithm was validated to obsess greater function approaching that of traditional measurements in prediction and identification of cardiac events, yet it hasn’t officially applied to the real-world practice to become the first-line medical tools or replace the golden standard.
Before widely applied into the clinical practice, several challenges of these technology need to be conquered. One major limitation of AI-driven health monitoring is the power consumption of wearable devices. Smart gadgets that provide real-time monitoring require a continuous power supply, which restricts their usefulness in long-term rehabilitation programmes. Although energy harvesting and wireless charging technologies may offer solutions, further innovations are needed. Additionally, the clinical readability and accuracy of data collected from smart devices also need to be improved. Although the high accuracy of them compared with traditional detection tools has been verified by mounting studies, there are still studies reporting concerns over the reliability of PPG-based heart rate measurements. These measurements are often less accurate than standard ECG readings, particularly during movement or in individuals with darker skin tones. 115 On one hand, large-scale data influx results in an increase in superfluous information; on the other hand, these algorithms lack an understanding of physiological details, making AI-driven clinical decisions potentially unreliable. 116 Efforts to refine these algorithms include developing more complex models to filter excess data while maintaining the accuracy of clinically relevant signals.
AI models trained on biased datasets may not generalize well across diverse populations, leading to further disparities. 117 Variability in demographics, health conditions, and treatment responses makes it challenging to develop a universal AI model for CR. Many AI-driven CR programmes rely on supervised learning models, which require large, well-annotated datasets to function. These datasets are often limited in diversity, which leads to predictions biased toward specific population groups. 118 Current AI algorithms also struggle to adapt to individual patient needs, which limits their capacity to provide personalized rehabilitation plans. 119
Another significant technical barrier is the lack of standardization and interoperability among AI-powered digital health tools and healthcare systems. 120 CR programmes can rely on a vast array of different platforms, including hospital databases, cloud-based monitoring systems, and mobile applications, making integration within electronic health records (EHRs) complex. There are also inconsistencies in data formatting, and many devices incorporate proprietary software. 121 AI-driven health monitoring also demands substantial computational power, particularly for deep learning models that are attempting to process datasets in real time. Integrating AI into wearables and portable devices is still limited by hardware constraints. 119 Cloud-based AI solutions can address some of these concerns but rely on stable internet connections. The high cost of AI-integrated DHTs may make them inaccessible to some patients. 122 Lastly, Current AI-based monitoring devices mostly track physical activity and vital signs but do not incorporate important lifestyle and psychological factors such as dietary habits or mental health, both of which drive CR success. Previous studies have shown that automated dietary monitoring can be achieved with multimodal sensing methods, but this technology remains underdeveloped. 123 Early evidence also suggests that AI-based wearables have poor accuracy and specificity in detecting anxiety, underscoring the need for further research before mental health assessments can be incorporated into this model. 124
Ethical and Privacy Concerns in AI-Enabled CR Programmes
The widespread implementation of AI in DHTs raises ethical concerns, particularly around patient privacy, security, and autonomy. Because these systems collect and process vast amounts of sensitive health data, robust safeguards are needed to prevent cyberattacks and data breaches. If compromised, patient health data could be exploited by third parties, such as insurance companies or commercial entities, for non-medical purposes, including premium pricing and targeted advertising. 120 This would threaten individual privacy and raise broader questions about the commodification of health data, which directly violates the principle of “inviolable autonomy.”
The opacity of AI’s decision-making processes also poses a major challenge. AI-driven CR programmes often function as ‘black boxes’, which make it difficult for both clinicians and patients to understand how clinical decisions are being made. This lack of transparency is particularly concerning when AI-generated recommendations contradict human clinical judgment. Explainable AI (XAI) models are being developed to increase algorithmic transparency, which would allow healthcare professionals to better validate AI-driven decisions. 100
The digital divide could further exacerbate disparities in AI-driven healthcare. Patients with limited digital literacy may struggle to engage with AI-powered DHTs, leading to reduced adherence.1,125 Financial constraints can also prevent patients from accessing AI-enhanced wearables or telehealth services.
Future Potential: Addressing Challenges and Enhancing AI-Based CR
Despite these challenges, ongoing advancements in AI and digital health present significant opportunities for improving CR programmes. Addressing sensor accuracy and data validation will be a key future priority. Multi-modal data fusion, which integrates readings from multiple sensors (eg, ECG, PPG, and accelerometers), may help enhance the robustness of AI-driven health assessments. 120 Additionally, federated learning—a ML approach that enables AI models to be trained on decentralized data without compromising patient privacy—may help mitigate issues related to dataset bias. 122
To tackle concerns surrounding algorithmic transparency, researchers are currently developing XAI models that can provide human-readable insights into their decision-making processes. 116 Incorporating XAI into CR programmes can help clinicians and patients to better understand and trust AI-driven recommendations, which will ultimately improve treatment adherence. Enhanced encryption methods, decentralized data storage (such as blockchain-based EHRs), and stricter privacy regulations may also strengthen security protocols. 126 Blockchain technology, in particular, offers a decentralized framework that can grant patients direct control over their personal health data and ensure a tamper-proof audit trail of all health records. This would enhance confidentiality and reduce the risk of data breaches. However, challenges such as data access, scalability, and interoperability still need to be addressed before blockchain can be widely adopted in AI-based CR solutions. 100
In addition to privacy concerns, efforts are also being made to bridge the digital literacy gap. For example, with regard to low medication adherence among patients with poor medical literacy, digital pills that automatically link with smartphone applications or other electronic devices and can thus remind patients to take them are being developed. 65 Ongoing interdisciplinary collaboration among AI developers, clinicians, and regulatory bodies is crucial to ensuring the safe and ethical deployment of AI in CR.
As CR programmes increasingly integrate wearable and implantable devices for real-time health monitoring, an existential question remains: will the self-tracking, AI-assisted patient become a mere data subject, reducing health and well-being to quantifiable metrics, or will AI enable a richer, more context-aware approach to personalized medicine that respects the complexity of the human experience? Addressing these concerns requires both technical refinement and an ethical framework that balances efficiency with human dignity, ensuring that AI-driven healthcare enhances rather than diminishes the lived experience of recovery and rehabilitation. By ensuring that AI-driven healthcare remains patient-centered, ethically guided, and context-sensitive, future CR programmes can harness these advancements to optimize clinical outcomes and empower individuals in their CR journeys.
Footnotes
Author Contributions
All authors made a significant contribution to the review article, whether in the conception, analysis, interpretation or all these areas. All authors participated in the drafting, revising, and reviewing of this article. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work. All authors have reviewed the final version of the manuscript and approved it for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Capital Health Development Research Project (NO.2024-2-2069), the Beijing Municipal Natural Science Foundation (7232043), Hospital High-Level Research Funding (2024AZB3003), the National Natural Science Foundation of China (82100486 and 82270341), High-level public technical talent construction project of Beijing Municipal Health Commission (Leading Talent-02-01), Beijing Hospitals Authority’s Ascent Plan (DFL20220603), and Beijing Hospitals Authority Youth Programme (QMS20210603).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
