Abstract
Background:
Excessive bleeding and coagulopathy frequently occur after cardiac surgery, and Fresh Frozen Plasma (FFP) remains the standard treatment. Prothrombin Complex Concentrates (PCC) present a potential alternative; however, their effectiveness and safety compared to FFP remain uncertain.
Methods:
Electronic databases, including PubMed, Cochrane Central, and ScienceDirect, were searched from inception till April 2025. The risk ratios (RR) and mean differences (MD) were pooled using the random-effects model in Review Manager software for the dichotomous and continuous outcomes, respectively. Leave-one-out sensitivity analysis and GRADE assessment were performed to evaluate the cause of heterogeneity and assess the certainty of evidence, respectively. Publication bias was evaluated visually through funnel plots and statistically through Egger’s regression test.
Results:
This meta-analysis included 9 studies—comprising 4 randomized controlled trials and 5 observational studies—covering a total of 1920 patients. PCC is associated with a significant reduction in chest tube drainage output within 24 hours compared to FFP (MD −170.05, 95% CI, −253.54 to −86.56; P < .0001; I2 = 38%). Similarly, the number of RBC units transfused (MD −1.06, 95% CI, −1.57 to −0.55, P < .0001; I2 = 45%) and number of patients requiring RBC transfusion (RR 0.84, 95% CI, 0.77-0.92; P < .0001; I2 = 47%) within first 24 hours are both reduced significantly in the PCC arm. Other outcomes including stroke (RR 1.03, 95% CI, 0.63-1.69; P = .90; I2 = 0%), thromboembolic events (RR 1.11, 95% CI, 0.76-1.64; P = .59; I2 = 0%), and all-cause mortality within 30 days (RR 1.03, 95% CI 0.73-1.45; P = .87; I2 = 0%) were comparable between both groups.
Conclusion:
PCC significantly reduced the chest tube drainage output along with the number of RBC units transfused and the number of patients requiring RBC transfusion within the first 24 hours. In contrast, stroke, other thromboembolic events, and all-cause mortality were comparable between the 2 groups.
Introduction
Excessive bleeding and coagulopathy are common and serious complications of cardiac surgery, with reoperation for bleeding required in approximately 2% to 6% of cases, and transfusion of allogeneic blood products in up to 80% of patients undergoing complex procedures. 1 Traditionally, Fresh Frozen Plasma (FFP) remains the primary treatment for acquired coagulopathy because it contains all coagulation factors and plasma proteins. 2 However, its use is associated with several drawbacks, including volume overload, transfusion-related acute lung injury (TRALI), delayed onset of action, and variable coagulation factor content.3,4
Moreover, the administration of FFP can exacerbate hemodynamic instability in cardiac patients already at risk of fluid overload. Prothrombin Complex Concentrates (PCC) offer a promising alternative due to their rapid reconstitution, small volume requirements, and standardized, high levels of vitamin K–dependent clotting factors (II, VII, IX, X). 5 Some formulations also include proteins C and S, enhancing their hemostatic profile. 6 PCCs can provide faster reversal of coagulopathy and reduce transfusion burden, especially in patients where fluid restriction is critical. Evidence comparing PCC and FFP in cardiac surgery has expanded in recent years. Studies like Cappabianca et al reported lower 24-hour chest tube drainage and reduced RBC transfusions in PCC groups. 7 However, other studies raise concerns about thromboembolic risks, with events reported in up to 8% of patients receiving PCC.8,9
Due to differences in study design, population characteristics, and outcome definitions, the results remain heterogeneous and inconclusive. Although guidelines are beginning to recognize PCC as an option for coagulopathic bleeding in cardiac surgery, a lack of consensus persists due to limited high-quality evidence and inconsistent results across studies. 10 This meta-analysis aims to synthesize current evidence to determine whether PCC is a superior, equivalent, or riskier alternative to FFP in adult patients undergoing cardiac surgery.
Methods
We carried out this systematic review and meta-analysis in compliance with the criteria issued by the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) protocols
11
and the Cochrane Handbook for Systematic Reviews of Interventions.
12
The procedural framework was registered in advance on Prospero under the ID
Search Strategy
A digitalized and programed search was run on multiple databases, including PubMed, Cochrane Central, and ScienceDirect, up to April 2025. We screened the studies comparing the PCC versus FFP in patients undergoing cardiac surgery of any type. The terms “prothrombin complex concentrates,” “Fresh Frozen Plasma,” and “Cardiac Surgery” were utilized as Medical Subject Headings (MeSH) terms or keywords. The detailed search strategies used in different electronic databases are provided in Supplemental Table S1. Additionally, we manually reviewed the citations of the included studies and review articles to ensure that no important publications were overlooked.
Selection Criteria
The inclusion criteria for this analysis encompassed RCTs and observational studies involving patients aged 18 years or older who underwent cardiac surgery and were predisposed to bleeding or coagulation abnormalities. Eligible studies were required to evaluate PCC as the intervention, with FFP as the comparator, and to report at least 1 common outcome across the included studies. The exclusion criteria included studies involving participants with pre-existing coagulopathic conditions, participants using the anticoagulants 5 days before cardiac surgery, studies with overlapping populations, or patients who received both PCC and FFP, as well as populations undergoing LVAD implantation. We also excluded non-original research articles, including editorials, case reports, cross-sectional studies, and review articles. Pre-existing coagulopathic conditions include genetic disorders like hemophilia, as well as acquired conditions caused by medications such as anticoagulants, or medical illnesses like liver diseases and disseminated intravascular coagulation (DIC). Studies were excluded if they involved overlapping populations, such as those registered from the same institution with identical data collection periods. In such cases, the study with the larger sample size was preferred for inclusion in the analysis.
Study Screening and Data Extraction
All the articles retrieved from the extensive search strategy were introduced to the EndNote Reference Manager (Version X7.5; Clarivate Analytics, Philadelphia, Pennsylvania, 2016). Firstly, duplicates were removed, and then screening was performed using a dual-phase approach. In the first phase, all articles were screened by 2 authors (B.S. and A.S.) independently, based on their evaluation of titles and abstracts. In the second phase, the full texts of appropriate articles were read to confirm relevance. Any disagreements were resolved by mutual discussion with a third investigator (M.H.W.).
Numerical data from the finalized articles were collected using an Excel spreadsheet. Two authors (A.S. and Z.U.A.) independently performed data extraction related to baseline characteristics and outcomes. The baseline data included study design, sample size, number of male and female patients receiving PCC or FFP, mean age, BMI, comorbidities such as diabetes and hypertension, patients undergoing CABG, incidence of emergent surgery, and follow-up days. Outcomes extracted included chest tube drainage output within 24 hours, units of RBC transfused in the first 24 hours, number of patients requiring RBC transfusions within 24 hours post-surgery, post-surgical stroke or TIA, thromboembolic events, and all-cause mortality within 30 days. Any discrepancies in data extraction were resolved through discussion with a third author (M.H.W.).
Risk of Bias Assessment
To conduct an appraisal of bias assessment in the included studies, we employed the Cochrane Risk of Bias 2.0 tool (ROB-2) for RCTs, and the Risk of Bias in Non-randomized Studies of Interventions (ROBIN-I) for observational studies.14,15 The ROB-2 tool contains 5 domains that evaluate the robustness through: (1) randomization process, (2) deviation from intended interventions, (3) missing outcome data, (4), and measurement of outcome (5) selection of the reported results. The ROBIN-I tool comprises 7 domains which assess bias via: (1) confounding factors, (2) selection of participants, (3) classification of interventions, (4) deviation from intended interventions, (5) missing outcome data, (6) measurement of outcomes, and (7) selection of reported results. The bias in the included studies is visually represented using the traffic light plots.
Statistical Analysis
Review Manager Software (version 5.4.1) was utilized for the statistical analysis of the outcomes retrieved from the included studies. Forest plots were generated to consolidate the results. The Risk Ratio (RR) and the Mean difference (MD), along with 95% Confidence Intervals (CI), were pooled for dichotomous and continuous outcomes, respectively. For statistical analysis, the inverse variance method and the Mantel-Haenszel method were employed for continuous and dichotomous outcomes, respectively. A P value of ⩽ .05 was considered statistically significant. Heterogeneity of the included studies was evaluated using the Higgins and Thompson I2 statistics, and studies with more than 50% value of I2 were considered heterogeneous. 16 A sensitivity analysis was performed to determine the source of heterogeneity. Subgroup analysis was also performed based on study design, either RCTs or observational studies. Publication bias in the included studies was assessed visually through funnel plots and statistically through Egger’s regression test. We evaluated the certainty of evidence using the GRADE assessment.
Results
Search Results
We started by searching PubMed, Cochrane Central, and ScienceDirect, retrieving a total of 1650 relevant articles (PubMed = 698, Cochrane Central = 30, and Science-Direct = 922). After removing 412 duplicates, 1238 articles remained for initial screening based on titles and abstracts, resulting in the exclusion of 1052 articles. The remaining 186 articles underwent full-text review, leading to the exclusion of 178 studies and ultimately yielding 9 studies for inclusion in our meta-analysis—comprising 4 RCTs and 5 observational studies.7,17 -24 We analyzed data from a total of 1920 patients. The studies included observational studies and RCTs. Figure 1 provides a PRISMA flow chart summarizing the study selection process.

PRISMA diagram showing the study selection process.
Study Characteristics
Our meta-analysis consisted of 9 studies, among which 4 were RCTs,17 -20 four were retrospective cohorts,7,21 -23 and 1 was a post-hoc analysis of an observational study. 24 Out of the total patient population of 1920, 826 received PCC, and 1094 received FFP. The sample size of the various studies in this meta-analysis varied between 24 and 225 patients, with 66.5% of the patient population being males. The mean age ranged from 61 to 72 years. Table 1 illustrates the baseline characteristics of the included studies. The details regarding the GRADE assessment are provided in Table 2.
Baseline Characteristics of the Included Studies.
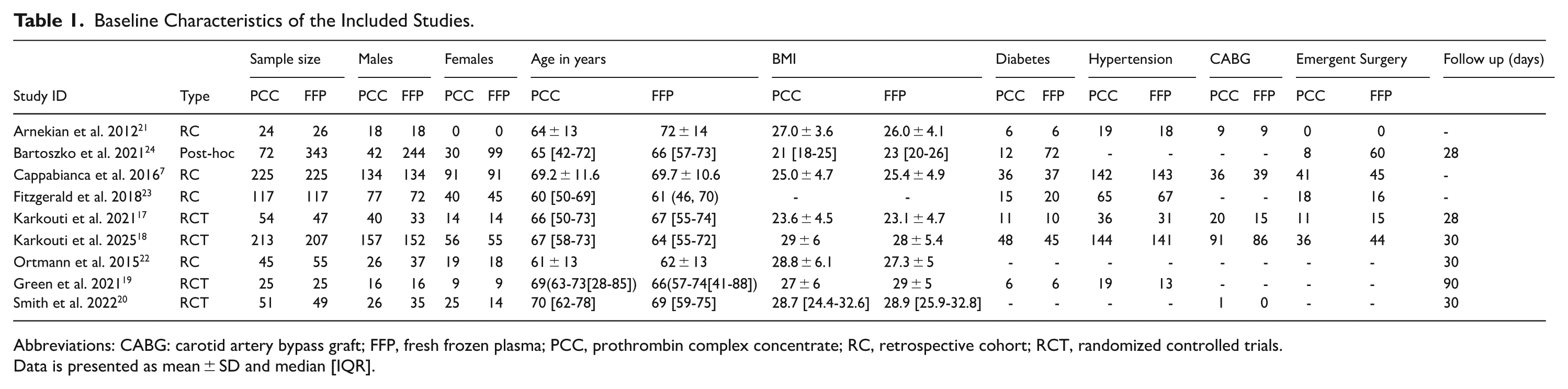
Abbreviations: CABG: carotid artery bypass graft; FFP, fresh frozen plasma; PCC, prothrombin complex concentrate; RC, retrospective cohort; RCT, randomized controlled trials.
Data is presented as mean ± SD and median [IQR].
GRADE Summary of Findings.

The heterogeneity is above 60%.
The 95% confidence interval (CI) is wide and has crossed 1.
Bias Assessment
We employed the ROB-2 tool to evaluate the quality of included RCTs and used ROBIN-I for assessing observational studies.14,15 Among the RCTs, 2 exhibited a low risk of bias,17,18 while 2 others had some concerns due to issues in outcome measurement and deviations from the planned intervention.19,20 In the observational studies’ bias assessment with ROBIN-I, 1 study 21 was rated as having a critical bias risk owing to serious confounding issues. Three studies showed a moderate bias risk, mainly related to confounding, outcome measurement, and selective reporting, with all other areas rated as low risk.7,22,23 One study demonstrated a low bias risk across all categories. 24 The results of the publication bias for RCTs via ROB-2 and for observational studies via ROBIN-I are provided in Figures 2 and 3, respectively.

Risk of bias (RoB 2.0) tool traffic light plot.
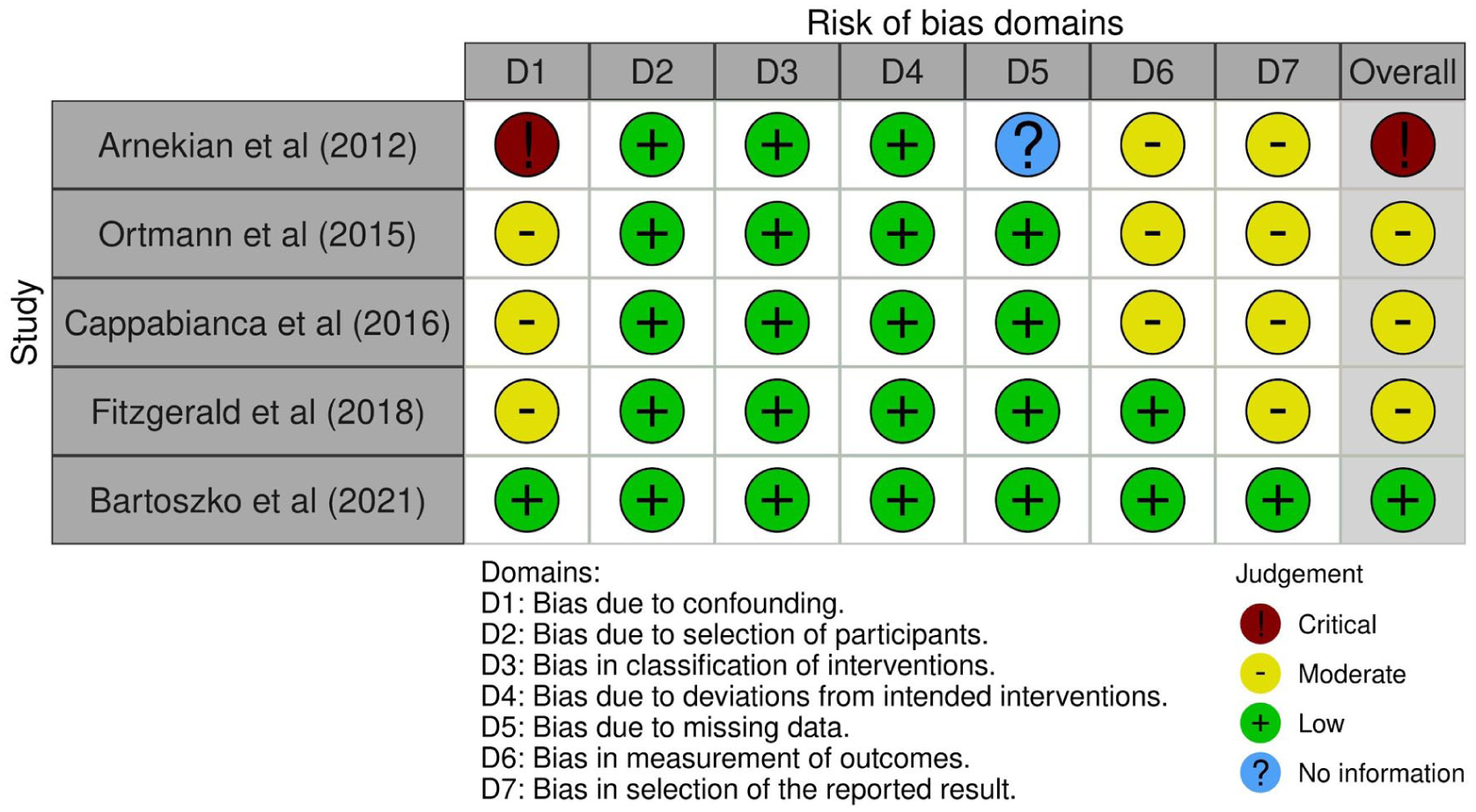
Risk of bias in non-randomized studies - of interventions (ROBINS-I) tool traffic light plot.
Outcomes
Chest Tube Drainage Output
Six studies7,17 -21 pooling 1170 patients (592 PCC vs 578 FFP) reported the chest tube drainage output. It was demonstrated that PCC was associated with significantly reduced chest tube drainage output within 24 hours compared to FFP (MD −170.05, 95% CI, −253.54 to −86.56; P < .0001). Moderate heterogeneity was detected (I2 = 38%, P = .15). On the subgroup analysis, the findings became insignificant in the observational studies subgroup (Figure 4).

Forest plot for chest tube drainage output within 24 hours.
RBC Units Transfused
Five studies7,17 -19,22 pooling 1121 patients (562 PCC vs 559 FFP) reported this outcome. The group treated with PCC was observed to require a lower number of RBC transfusions within 24 hours post-surgery (MD −1.06, 95% CI, −1.57 to −0.55, P < .0001). Moderate heterogeneity was detected (I2 = 45%). On subgrouping, the results remained significant in both the RCTs and observational studies subgroups (Figure 5).

Forest plot for number of RBC units transfused within the first 24 hours.
RBC Transfusion Rate
A total of 8 studies7,17,20 -24 pooling 1869 patients (801 PCC vs 1068 FFP) reported the RBC transfusion rate. We observed that PCC was associated with a significantly lower number of patients requiring RBC transfusions within 24 hours post-cardiac surgery compared to FFP (RR 0.84, 95% CI 0.77-0.92; P < .0001). Moderate heterogeneity was detected between the experimental and control groups (I2 = 47%). Regarding subgrouping, the findings remained significant across both the RCTs and observational studies subgroups (Figure 6).

Forest plot for RBC transfusion rate within the first 24 hours after cardiac surgery.
Stroke or TIA
Eight studies7,17 -23 pooling 1510 patients (758 PCC vs 752 FFP) reported this outcome. The incidence of stroke or TIA after cardiac surgery was comparable between the PCC and FFP groups (RR 1.03, 95% CI 0.63-1.69; P = .90). No significant heterogeneity was detected (I2 = 0%). Regarding subgroup analysis, the results remained insignificant in both the RCTs and observational studies subgroups (Supplemental Figure 1).
Thromboembolic Events
Eight studies17 -24 pooling 1475 patients (605 PCC vs 870 FFP) documented this outcome. No statistically significant difference was observed in the incidence of thromboembolic events between the 2 treatment groups (RR 1.11, 95% CI, 0.76-1.64; P = .59). No significant heterogeneity was detected (I2 = 0%). Regarding subgroup analysis, the results remained insignificant across both the RCTs and observational studies subgroups, as illustrated in Supplemental Figure 2.
All-Cause Mortality
The incidence of all-cause mortality within 30 days of surgery was reported by 9 studies,7,17 -24 pooling a total of 1920 patients (1097 PCC vs 823 FFP). The patient populations treated with PCC demonstrated comparable results to those treated with FFP (RR 1.03, 95% CI 0.73-1.45; P = .87). No significant heterogeneity was observed (I2 = 0%). On subgroup analysis, the findings were consistently insignificant in both the RCTs and observational studies subgroups (Supplemental Figure 3).
Sensitivity Analysis and Publication Bias
A leave-one-out sensitivity analysis was performed to identify the study contributing to heterogeneity when I2 >50%. No publication bias was detected visually on funnel plots, which was further confirmed with Egger’s regression test Supplemental Table S2 and Figures 4 to 9.
Discussion
The management of bleeding in cardiac surgery remains a cornerstone of perioperative care, with profound implications for morbidity, transfusion-related complications, and healthcare costs. This systematic review and meta-analysis investigated whether PCC offers clinical advantages over FFP in managing coagulation among cardiac surgery patients.
FFP is widely used as a hemostatic method in cardiac surgery. It has a well-established efficacy proven over decades, 10 and it is more readily available and less expensive than PCC. Additionally, FFP is considered a safer option due to concerns about the safety profile of PCC, particularly regarding the risk of thromboembolic events and kidney injury. 7 Our findings suggest that PCC may provide a more targeted and effective hemostatic strategy than FFP, particularly in reducing bleeding, chest tube drainage output within the first 24 hours, and transfusion requirements without increasing the risk of thromboembolic or neurologic complications such as TIA or stroke. However, all-cause mortality showed no difference between the 2 groups. All these findings are corroborated by a recent meta-analysis conducted by Ali et al. 25 Our study builds upon the work by Ali et al.’s 25 by incorporating both RCTs and observational cohort studies, thereby enhancing the statistical power and reinforcing the robustness of the results due to increased sample size. The subgrouping analysis by study design also enables us to assess how different study designs influence the overall effect sizes of the endpoints.
One of the most apparent benefits identified in this analysis was the reduction in chest tube drainage output within the first 24 hours following surgery in the PCC group. This outcome, which serves as a surrogate for postoperative bleeding, had a statistically significant mean reduction in favor of PCC. Similarly, both the number of RBC units transfused and the proportion of patients requiring transfusion within the first 24 hours were significantly lower in the PCC group. Previously conducted meta-analyses of observational studies suggested a similar outcome of decreased blood loss and units of RBC transfused.9,25,26 However, on the subgroup analysis, the mean reduction in the chest tube drainage output became insignificant in the observational studies subgroup, which may be due to variability in the patient populations, management protocols, and the lack of randomization.
Higher rates of RBC transfusion have been consistently associated with an increase in adverse outcomes in cardiovascular surgery, including risk of infection, acute lung injury, mortality, and prolonged length of hospital stay.27 -29 Therefore, limiting transfusions through effective reversal agents, such as PCC, may directly improve patient outcomes. These adverse effects may be, at least partially, attributable to fluid overload induced by transfusions, as FFP is typically given in 250 to 300-ml units, and multiple units are often needed to achieve adequate coagulation.28,30 This can contribute to complications such as congestive heart failure and pulmonary edema. 8 In contrast, PCC is effective in volumes smaller than 50 ml, helping protect high-risk patients from volume-related complications. 31 Additionally, the larger volume of FFP raises the risk of hemodilution and associated complications. 32
The decreased RBC transfusion required could also potentially be explained by the concentrated composition of vitamin K-dependent clotting factors (II, VII, IX, and X) forming PCC. FFP preparation involves blood group matching and thawing before administration, whereas PCC can be given quickly to help achieve quicker and more efficient control of blood loss.7,33,34 This rapid administration is crucial for the timely correction of coagulopathy, particularly in patients undergoing cardiac surgery.10,26,29,34,35 By decreasing bleeding, PCC may also reduce the need for re-operation due to hemorrhage, a complication associated with significantly worse outcomes in cardiac surgery populations. 36 Importantly, reduced bleeding also contributes to downstream reductions in transfusion, as reflected in our results.
The risk of stroke or TIA, a major concern with any pro-coagulant therapy, showed no significant difference between PCC and FFP. Although a theoretical concern exists regarding the overcorrection of coagulopathy and thrombosis risk with PCC, our analysis and prior studies 37 show no convincing evidence of such an association in the context of cardiac surgery. Importantly, stroke incidence remains multifactorial in this population, often influenced by aortic manipulation, cardiopulmonary bypass time, and patient comorbidities rather than solely by reversal agents.
Similarly, the analysis revealed no statistically significant difference in thromboembolic complications between PCC and FFP. This finding aligns with real-world registry data 38 and suggests that PCC does not unduly heighten the risk of clotting in postoperative cardiac surgery patients when used appropriately and within guideline-recommended dosing limits. Previously conducted meta-analyses also found no increase in the association between thromboembolic events and the use of PCC.9,25,26
Our meta-analysis found no significant difference in all-cause mortality within 30 days between PCC and FFP recipients. This is a particularly interesting finding as PCC improves several perioperative surrogate markers (e.g., bleeding, transfusion needs), but these advantages did not translate into short-term mortality reduction. There may be several reasons for this, as mortality in cardiac surgery is influenced by a broad array of factors beyond bleeding management. These can include myocardial dysfunction, sepsis, organ failure, and so forth. Furthermore, the included studies may have been underpowered to detect mortality differences due to relatively small event rates. Another reason might be that the follow-up duration of 30 days may not fully capture the long-term benefit or harm of reduced transfusions or thromboembolic events. Nevertheless, neutrality in mortality is important as it confirms the safety of PCC, dispelling concerns about the potential over-correction of coagulopathy.
Limitations
The overall findings of our meta-analysis support the use of PCC in selected patients, but certain limitations must be acknowledged. Some of the included studies were observational, introducing potential confounding and bias. All our outcomes exhibited low to moderate heterogeneity, with consistent effects favoring Prothrombin Complex Concentrate (PCC). Although Egger’s regression test shows no evidence of publication bias, the potential for such bias cannot be completely ruled out, as neutral studies and those with negative results may still be underreported. Similarly, the small sample sizes in some included studies may restrict the generalizability and strength of the analysis. Due to selective result reporting, there is a concern about reporting bias, which could influence how the findings are interpreted, particularly given the neutral mortality results despite improvements in hemostasis.
The findings of this study align with the growing body of literature, suggesting that PCC is a practical, effective, and safe alternative to FFP for managing coagulopathy during and after cardiac surgery. Its rapid action, smaller volume, and lower transfusion requirements make it an appealing option for patients at high risk of bleeding or those sensitive to fluid overload. However, the widespread use of PCC should be approached with caution. Strict adherence to dosage guidelines and thorough screening for thromboembolic risks is essential. Further large-scale randomized trials are needed to validate its long-term safety and cost-effectiveness.
Conclusion
This meta-analysis supports using PCC as a favorable alternative to FFP for patients undergoing cardiac surgery, particularly in reducing bleeding and the necessity for transfusions. While no significant differences were found in all-cause mortality rates or adverse thromboembolic events, the hemostatic advantages of PCC position it as a promising therapeutic option. Nevertheless, clinicians should continue to tailor their reversal strategies to each patient’s unique bleeding risk, the complexity of the procedure, and individual coagulation status.
Supplemental Material
sj-docx-1-cic-10.1177_11795468251391024 – Supplemental material for Prothrombin Complex Concentrate Versus Fresh Frozen Plasma in Patients Undergoing Cardiac Surgery: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cic-10.1177_11795468251391024 for Prothrombin Complex Concentrate Versus Fresh Frozen Plasma in Patients Undergoing Cardiac Surgery: A Systematic Review and Meta-Analysis by Muhammad Hassan Waseem, Zain ul Abideen, Ameer Haider Cheema, Areeba Shoaib, Aiman Waheed, Muhammad Mukhlis, Barka Sajid, Sania Aimen and Pawan Kumar Thada in Clinical Medicine Insights: Cardiology
Footnotes
Author Note
This manuscript has not been published previously and is not under consideration for publication elsewhere
Author Contributions
Study concept and design: MHW and ZUA; acquisition of data: ZUA, BS and AS ;analysis and interpretation of data: AW, PKT and ZUA ; drafting of the manuscript: AS, AHC, AW, SA and MM ; critical revision of the manuscript: MHW, AHC
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available upon reasonable request to the authors
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
