Abstract
Background:
The heritability of salt-sensitive hypertension and the heightened susceptibility of offspring to maternal perinatal high salt diet (HSD) indicate that hypertension may originate early in life. However, the mechanism underlying this phenomenon remains unclear. We hypothesized that perinatal exposure of dams to HSD will increase inflammation, impair vascular function and elevate blood pressure (BP) in the adult offspring.
Methods:
Pregnant rats were fed a normal (0.3%) or high (8%) salt diet during pregnancy and the offspring from each group were weaned at 4 weeks of age and placed on normal salt diet (NSD) for 12 weeks. BP measurement, vascular reactivity studies, and ELISA assay for C-reactive proteins (CRP), Tumor Necrotic Factor (TNF-α), and Interleukin-6 (IL-6) were carried out. Data were analyzed using student t-test. The significance level was set at P-values ⩽.05.
Results:
The offspring of dams exposed to perinatal HSD exhibited elevated BP parameters compared to those from dams on NSD. Although the maximum contractile response to noradrenaline was similar in both groups (P > .05), the maximum relaxation response to acetylcholine was significantly reduced in offspring of HSD-exposed dams (P < .01), indicating impaired endothelial function. Furthermore, perinatal HSD led to increased levels of CRP, TNF-α, and IL-6 in the offspring, indicating heightened systemic and vascular inflammation.
Conclusion:
Findings from this study show that maternal perinatal HSD increased biomarkers of inflammation, impaired endothelial function and elevated BP in the adult offspring. These findings suggest maternal perinatal consumption of high dietary salt renders the offspring more susceptible to hypertension in adulthood.
Keywords
Introduction
Hypertension is a global public health concern because it is the most important risk factor for cardiovascular diseases which is the leading cause of death, globally. An excess of dietary salt is the most important modifiable risk factor that contributes to the development of hypertension leading to cardiovascular pathologies. 1 Salt sensitivity increases the risk of death whether a person has high blood pressure or not, and it has been reported that, salt-sensitive hypertension is associated with higher morbidity and mortality due to cardiovascular diseases than hypertension caused by other factors. 2 About 27% to 38% of adult Nigerians are hypertensive3 -5; and recent findings from our research group showed that about 54% of hypertensive and 35% of normotensives adult Nigerians are salt-sensitive respectively. 6 Yet, there is a continuous increase in the consumption of high dietary salt in the Nigerian population. 7 A more worrisome trend is the continuous increase in child and adolescent hypertension8,9 which suggest hypertension has an early origin in life. However, the contribution of a high salt diet to the increase in child and adolescent hypertension is not clear.
The mechanisms involved in the blood pressure (BP) elevating effect of a high salt diet are not fully understood. However, experimental studies have suggested impairment of cardiovascular and renal functions as well as dysregulation of fluid volume and autonomic system.10,11 More recently, systemic and vascular inflammation as well as their immunomodulation have been implicated in the pathogenesis of hypertension. For instance, in several cross-sectional and longitudinal studies, plasma level of C-reactive protein (CRP)—a marker of systemic inflammation has been shown to be higher in hypertensive when compared with normotensive subjects.12,13 Specifically, plasma level of CRP correlates positively with urinary sodium concentration in hypertensive subjects.14,15 Higher level of serum CRP has also been reported in normotensive offspring of hypertensive parents when compared with those of non-hypertensive parents, suggesting that systemic inflammation may precede hypertension.16,17 C-reactive protein is an acute phase protein that can stimulate monocytes to release proinflammatory cytokines such as interleukin-6 (IL-6), interleukin-1 beta (IL-1β), and tumor necrosis factor alpha (TNF-α) while also acting on endothelial cells to express intracellular adhesion molecule (ICAM)-1 and vascular cell adhesion molecule (VCAM)-1. Animal studies have also shown that excess salt alters the innate immune system, promotes tissue inflammation and exacerbates autoimmune disease in mice.18,19 A high salt diet has been linked to enhanced inflammation and target organ damage in hypertension, 14 as TGF-β has been shown to be over expressed in the kidney of animals fed a high salt diet. 20 In fact, pathogenesis of hypertension is believed to be closely intertwined with high dietary salt intake and the immune response to the ensuing systemic and vascular inflammation.21,22 Vascular wall inflammation plays a key role in the pathogenesis of several cardiovascular diseases including hypertension.21,23,24 The p38 mitogen activated protein kinase (MAPK) pathway has been implicated in vascular inflammation. For instance, the activation of p38 MAPK has been reported to play a pivotal role in the production of proinflammatory cytokines such as IL-1, IL-6, and TNF-α. 25 Likewise, nuclear factor kappa B (NF-κB) and IL-6 signaling pathway has been implicated in vascular inflammation, 23 suggesting an intricate interplay between the p38 MAPK and NF-κB pathways through IL-6.
Events during early life have been shown to permanently alter phenotypes in adulthood,26,27 hence having impact on the risk of developing future adult diseases.28,29 Cardiovascular health has been found to have many of its origins in early development as the cardiovascular system is largely affected by early environmental perturbations especially during perinatal events. 27 The effect of maternal phenotype and environment such as stress and nutrition on the offspring cardiovascular and renal health and diseases has been established.27,30 During the in-utero period of life, the fetus relies heavily and at times solely on maternal contribution to the fetal environment. For instance, maternal nutrition has been suggested to have a direct influence on the offspring immunity. This is evident by nutrient-driven epigenetic changes to developing T-regulatory cells as well as placenta and breast milk transfer of immune factors (cytokines and chemokines) to the offspring. 31 Experimental studies have also shown that perinatal events such as low protein, 32 hypoxia,33,34 low iron, 35 and high salt diet36,37 have a negative influence on blood pressure later in life. Suggested mechanisms by which maternal high salt diet causes hypertension in the offspring include; impairment of RAS activation in response to salt loading,38,39 increase in pressor response and / or enhanced vascular reactivity to angiotensin II (AII), 40 left ventricular hypertrophy and increased angiotensin I receptor (AT1R), 41 remodeling of heart and arteries, 37 and endothelial dysfunction. 36 However, it is not clear how maternal perinatal high salt diet increases systemic and vascular inflammation (an important mechanism for pathogenesis of salt sensitive hypertension); impairs endothelial and vascular function and thereby renders the adult offspring more susceptible to the development of hypertension. Therefore, this study is designed to test the central hypothesis that perinatal exposure to high salt diet will increase biomarkers of systemic and vascular inflammation, impair endothelial function and elevate BP in the adult offspring.
Materials and Methods
Animal Grouping
All animal procedures conducted in this study adhered to the guidelines set forth by the Animal Care and Use Research Ethics Committee (ACUREC) of the College of Medicine, University of Lagos (CMUL) regarding the utilization of laboratory animals. Six pregnant Sprague-Dawley rats were randomly assigned to either perinatal normal (0.3%) or high (8%) salt diets groups from the first day of pregnancy until term. Offspring were weaned at 4 weeks and 6, (3 males and 3 females) rats were randomly selected from each of the dams’ dietary groups and maintained on a normal salt diet throughout the experimental period. The offspring were grouped according to their maternal diet during pregnancy, so that offspring of normal salt (NS) dams are NS group and offspring of high salt (HS) dams are HS group. Subsequent experiments were conducted on the offspring when they reached 12 weeks of age.
Blood Pressure Measurement
At the conclusion of the 12-week period, the blood pressure of the animals was measured using previously described methods.42,43 In brief, blood pressure was measured terminally via an invasive method under anesthesia induced by a combination of 25% urethane and 1% α-chloralose (5 mL/kg body weight) in the rats. An incision was made on the trachea, and a tracheal tube (PE250) was inserted for ventilation. Two loops of thread were used to secure the tracheal tube in place. The carotid artery was exteriorized and cannulated using a PE50 cannula filled with 1% normal saline. The cannula was connected to a pressure transducer (model SP 844, Physiological Pressure Transducer, AD Instruments), that was attached through MLAC11 Grass adapter cable to a computerized data acquisition system with LabChart-7 pro software (Power Lab-4/24T, model MLT844/P; AD Instruments Pty Ltd., Castle Hill, Australia). Heart rate was determined by counting arterial pulses, and recordings were sampled at a frequency of 5/seconds. 44
Vascular Reactivity Study
Preparation of abdominal aortic rings involved sacrificing rats via cervical dislocation. Immediately following dislocation, the abdominal region was opened, and the abdominal aorta was excised and placed in cold Physiological Salt Solution (PSS) at 4°C. After removal of fat and connective tissues, the aorta was sectioned into ring segments (2 -3 mm). The ring was thereafter mounted between one long and one short stainless-steel hooks. The small S-shaped hook was attached to the base of the organ bath, while the long L-shaped rod was attached to the isometric force transducer (top force transducer MLT 050/D; AD Instruments, Bella Vista, Australia) that was attached through MLAC11 Grass adapter cable to a computerized data acquisition system with LabChart-7 pro software (Power Lab-4/24T, model MLT844/P; AD Instruments Pty Ltd., Castle Hill, Australia). During this procedure, special care was taken to avoid rubbing the endothelial surface of the rings. The 20 mL organ bath contained PSS with the composition: 119.0 mmol/L NaCl, 4.7 mmol/L KCl, 1.2 mmol/L KH2PO4, 1 to 2 mmol/L MgSO4, 24.9 mmol/L NaHCO3, 1.6 mmol/L CaCl2, and 11.5 mmol/L glucose at 37°C. The pH of the PSS was adjusted to 7.4 and the set-up gassed with 95% O2:5% CO2 mixture. For each ring, a passive tension of 1.5 g was applied, and the ring was allowed to equilibrate for 90 minutes in the PSS during which at 30 minutes interval, it was subjected to a dose of 10−6 M noradrenaline. The ring was rinsed after each stimulation. This 90-minute stabilization was necessary to ensure a consistent response during the experiment. After this stabilization period, contractile response to graded doses of Noradrenaline (10−9-10−4 M) was established. Afterward, relaxation response to acetylcholine (10−9-10−4 M) was assessed separately in the absence or presence of L-nitro-arginine-methyl-ester (L-NAME, 10−4 M; 30 minutes) following pre-contraction with 10−7 M NA. All experiments took place under the same environmental temperature and pressure
Cytokine Assays
Blood samples were collected from the cannulated carotid artery into plain tubes. The tubes were then centrifuged at 3000 rpm at room temperature for 10 minutes; the resulting serum was immediately frozen at −80°C for further analysis. Serum levels of CRP, IL-6, TNF-α were measured using Enzyme-linked-immunosorbent serologic assay (ELISA) techniques as per the manufacturer’s instructions (Elabscience Biotechnology Co., Ltd., Wuhan, P.R. China).
Statistical Analyses
All measurements were presented as mean ± standard error of the mean (SEM). Statistical analysis was performed with the aid of Prism 10 (GraphPad) software. For group comparison, unpaired data with normal distribution were analyzed using student t test. Non-normally distributed data were analyzed using a non-parametric test (Mann Whitney test). For drug concentrations between the groups, one-way ANOVA followed by Student’s Newman-Keuls post-hoc test or 2-way ANOVA was used. Statistical significance was determined at P < .05.
Results
Perinatal Exposure of Dams to a High Salt Diet Promotes an Increase in Systolic, Diastolic, and Mean Arterial Blood Pressure of the Offspring
Systolic, diastolic and mean arterial blood pressures were significantly higher (P < .010; .025 and .0079) respectively in the offspring of Dams fed high salt diet when compared with the offspring of dams fed normal Salt diet. However, there was no significant difference in the pulse pressure and heart rates of offspring from dams fed high or normal salt diet. This is illustrated in Table 1.
Systolic BP, Diastolic BP, Mean Arterial BP, Pulse Pressure, and Heart Rate of Adult Offspring of Dams Fed a Normal or High Salt Diet During Pregnancy.

n = 6, data presented as mean ± SEM. The bolden values are the P-values of the significance symbols on the BP values immediately above them in the table. For example 0.10 is the level of significant increase in SBP of HS offspring when compared with NS offspring. Same with 0.25 for DBP and .0079 for MABP.
Significant increase in systolic BP when compared with NS offspring.
Significant increase in diastolic BP when compared with NS offspring.
Significant increase in mean arterial BP when compared with NS offspring.
Perinatal Maternal Exposure to High Salt Diet Did Not Alter the Contractile Response to of the Abdominal Aorta to Noradrenaline in the Offspring
The percentage contractile response to graded doses of noradrenaline was compared in the aortic ring from the offspring of dams from the 2 groups. There was no statistically significant difference (P > .05) between the percent contractile response to noradrenaline in the rings of the offspring of NS Dams when compared with the HS Dams. This is shown in Figure 1.

Contractile response of abdominal aorta to noradrenaline in the offspring of dams exposed to perinatal high salt and normal salt diet.
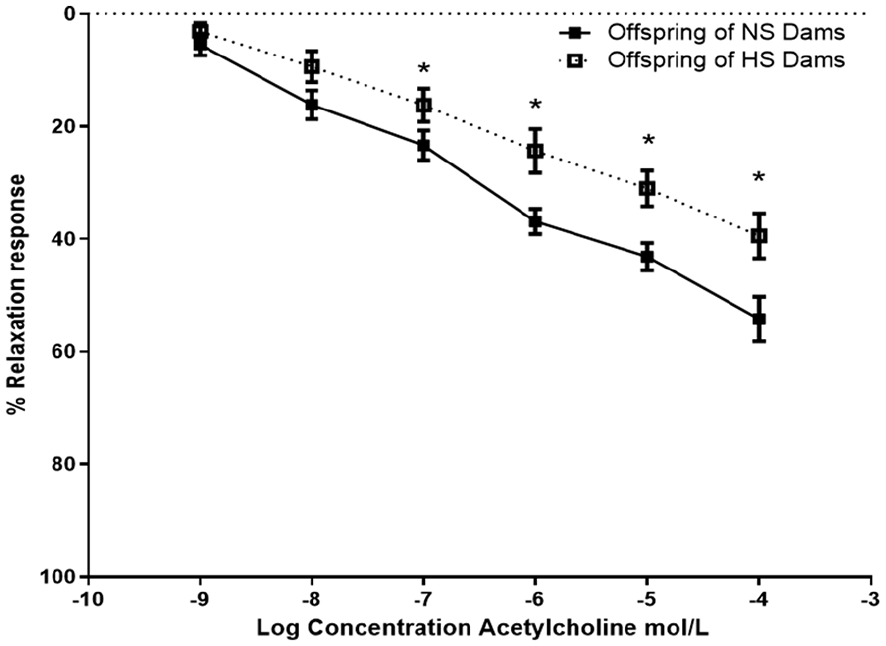
Perinatal Exposure of Dams to a High Salt Diet Reduced the Relaxation Response of the Abdominal Aorta to Acetylcholine (ACh) in the Offspring
The relaxation response at various graded doses of acetylcholine was markedly lower (P < .05) in the aortic rings of the offspring of Dams fed the high salt (HS) compared to their Normal Salt (NS) counterparts. The magnitude of the difference appears to be higher with increasing concentrations of acetylcholine. This is illustrated in Figure 2.

Relaxation response of abdominal aorta to acetylcholine (ACh) in the offspring of dams exposed to perinatal high salt and normal salt diet.
Effect of Perinatal Exposure of Dams to a High Salt Diet on the Relaxation Response of Abdominal Aorta to Acetylcholine (ACh) in the Offspring in the Presence of L-NAME
The possible influence of endothelial nitric oxide synthase (eNOS) inhibition on the vascular function of offspring of dams exposed to maternal perinatal high salt diet was assessed via incubation of aortic rings from the offspring in L-nitro-arginine methyl-ester (L-NAME). The relaxation response at different graded doses of ACh was markedly lower (P < .05; P < .001) in the aortic rings of the offspring of Dams fed the high salt (HS) compared to their Normal Salt (NS) counterpart. The magnitude of the difference appears to be higher with increasing concentrations of acetylcholine. This is illustrated in Figure 3.
Relaxation response of the abdominal aorta from offspring of Normal Salt (NS) and High Salt (HS) Dams to acetylcholine (ACh) in the presence of L-NAME—an eNOS inhibitor.
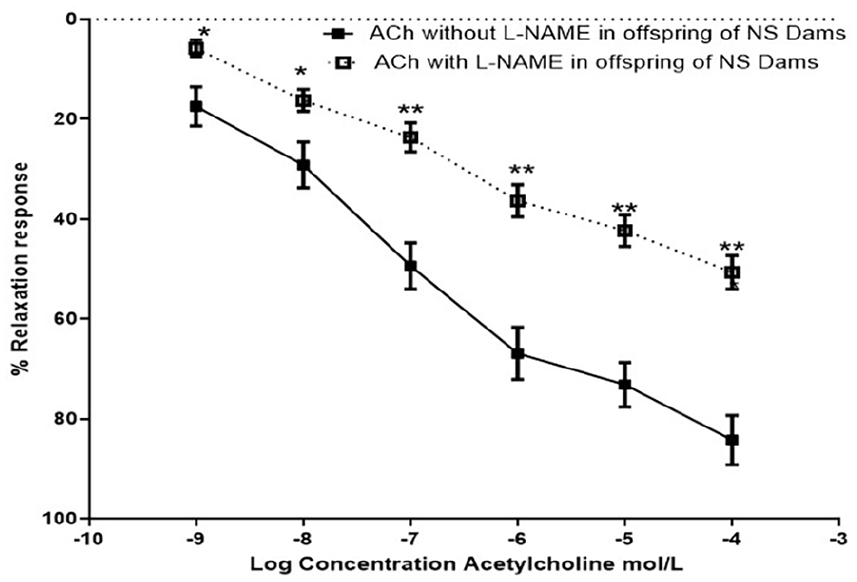
Abdominal Aorta Relaxation Response to Acetylcholine (ACh) in the Presence of Endothelial Nitric Oxide Synthase (eNOS) Inhibition With L-NAME in the Offspring of Dams Fed a Perinatal Normal Salt Diet
The relaxation response to acetylcholine in the presence of L-NAME in the aortic rings of the offspring of Dams fed a normal salt diet was significantly lower (P < .05; P < .001) when compared to the relaxation to ACh without eNOS inhibition with L-NAME. This reduction was observed at every graded dose of the ACh used, however, the magnitude of the difference appears to be higher with increasing concentrations of acetylcholine, Figure 4.
Relaxation response of the abdominal aorta from offspring of Normal Salt (NS) and High Salt (HS) Dams to acetylcholine (ACh) in the presence of L-NAME—an eNOS inhibitor.
Abdominal Aorta Relaxation Response to Acetylcholine (ACh) in the Presence of eNOS Inhibition With L-Nitro Arginine (L-NAME) in the Offspring of Dams Fed a Perinatal High Salt Diet
The relaxation response to acetylcholine in the presence of L-NAME in the aortic rings of the offspring of Dams fed a high salt diet was significantly lower (P < .05) when compared with the relaxation to ACh without eNOS inhibition with L-NAME. This reduction was observed only from the upward 10−7 mol/L concentration of ACh. However, the magnitude of the difference appears to be higher with increasing concentrations of acetylcholine. This is illustrated in Figure 5.

Relaxation response of the abdominal aorta from offspring of normal salt (NS) and high salt (HS) Dams to acetylcholine (ACh) in the presence of L-NAME—an eNOS inhibitor.
Percent Inhibition of eNOS by L-NAME in Abdominal Aorta From Offspring of Dams Fed a Perinatal High or Normal Salt Diet
The percentage of eNOS inhibition by L-NAME in the abdominal aorta from offspring of dams fed a perinatal HSD was significantly reduced when compared with that from the abdominal aorta of offsprings of dams fed a perinatal NSD. The percentage inhibitions from each group as well as the difference in percentage inhibition between NS and HS groups increase with increasing concentrations of acetylcholine. This is illustrated in Figure 6.

Percentage inhibition of eNOS by L-NAME in the abdominal aorta from offspring of dams fed a perinatal normal salt (NS) or high salt (HS) diet.
Perinatal Exposure of Dams to a High Salt Diet Elevates Serum Level of C-Reactive Protein (CRP) of the Offspring
The serum level of CRP in the offspring of HS dams was significantly higher (P < .001) than that of the NS dams’ offspring. Figure 7 illustrates the levels in the 2 groups of rats.

Serum levels of C-reactive protein (CRP) of the offspring from dams exposed to perinatal normal salt (NS) and high salt (HS) diet.
Perinatal Exposure of Dams to a High Salt Diet Enhances the Serum Level of Interleukin-6 (IL-6) of the Offspring
The serum level of IL-6 in the offspring of HS dams was significantly higher (P < .046) compared to the level in their HS counterparts. This is illustrated in Figure 8.

Serum level of interleukin 6 (IL-6) of the offspring from dams exposed to perinatal normal salt (NS) and high salt (HS) diet.
Serum Level of Tumor Necrotic Factor—Alpha (TNF-α) is Increased in the Offspring of Dams Exposed to a Perinatal High Salt Diet
The serum level of TNF-α in the offspring of dams fed a perinatal high salt diet was significantly higher (P < .001) when compared with those from dams fed a perinatal normal salt diet. This is illustrated in Figure 9.

Serum level of Tissue Necrotic factor—Alpha (TNF-α) of the offspring from dams exposed to perinatal normal salt (NS) and high salt (HS) diet.
Discussion
This study investigated the effects of perinatal maternal high salt diet consumption on biomarkers of systemic and vascular inflammation, endothelial function and blood pressure in the adult offspring that were placed on postnatal normal salt diet. We show that exposure of pregnant dams to a high salt diet throughout the course of pregnancy renders the adult offspring more susceptible to systemic and vascular inflammation, which is evident by elevated serum concentration of CRP, TNF-α, and IL-6 which are biomarkers of inflammation. Likewise, we demonstrated endothelial dysfunction as a reduced relaxation response of abdominal aorta to acetylcholine (an endothelial dependent vasodilator) in adult offspring of dams exposed to perinatal high salt diet. Furthermore, we showed the endothelial dysfunction impairing effect of perinatal maternal salt diet is NO-dependent, as inhibition of eNOS by L-NAME impaired relaxation response to ACh more in the abdominal aorta from offspring of dams fed a normal high salt diet compared to those from dams fed a perinatal high salt diet. This suggests that the availability of NO may be reduced or its contribution to endothelial-dependent relaxation is impaired in the abdominal aorta of the adult offspring of dams placed on perinatal HSD.
In this study, perinatal exposure of dams to a high salt diet resulted in elevated BP parameters such as systolic, diastolic and mean arterial BP. However, pulse pressure and heart rate are similar in the 2 groups of offspring. The observed elevated BP parameters in the adult offspring despite being fed a postnatal normal diet indicates a potential perinatal modification of certain blood pressure regulatory mechanisms in the offspring due to the maternal perinatal high salt diet consumption. The rise in MABP of the offspring occurred without a notable alteration in the contractile response to noradrenaline. However, the increased BP parameters in the offspring was accompanied by impaired vascular relaxation response to acetylcholine (ACh) with or without endothelial nitric oxide synthase (eNOS) inhibition. The reduction in the ACh-induced vascular relaxation, independent of nitric oxide synthase inhibition, suggests endothelial function impairment in the offspring of dams fed a perinatal high salt diet. Despite the inhibition of eNOS activity by L-NAME, a sizable relaxation response to ACh still exist in the blood vessels of the offspring from both normal- and high-salt-fed dams. This finding is consistent with our previous findings 45 and it suggests the involvement of other vaso-relaxing factors 46 in the mechanism of the offspring’s vascular function impairing effect of perinatal maternal high salt diet. Findings from this study also revealed that eNOS activity inhibition by L-NAME was more pronounced in the blood vessels of offspring of dams exposed to maternal perinatal normal salt diet than those from dams exposed to a perinatal high salt diet. This markable reduction in the magnitude of the difference in the relaxation response of blood vessels from offspring from dams fed a high salt diet with or without eNOS inhibition indicates an endothelial function impairing effect of maternal perinatal high salt diet.
Although we did not measure the level of nitric oxide in the experimental animals in this study, the reduction in ACh-induced relaxation in the blood vessels of offspring of dams fed a perinatal high salt diet suggests a reduction in endothelial nitric oxide production or a decrease in bioavailability of nitric oxide in the vascular endothelium. These 2 scenarios are possible as studies have demonstrated a reduction in nitric oxide production in animals fed a high salt diet consequent to reduced expression of vascular eNOS genes 47 and protein. 48 Reduction in the bioavailability of nitric oxide in animals fed a high-salt diet has also been reported. 49 This reduction in nitric oxide bioavailability has been linked to increased reactive oxygen species and oxidative stress in animals fed a high salt diet. 50 Despite not feeding the offspring a diet high in salt content directly, the perinatal consumption of a high salt diet by the dams affected the offspring vascular and endothelial functions. These observations are consistent with findings from other studies. Impaired ACh-dependent vasorelaxation has been reported in offspring of dams exposed to perinatal L-NAME-induced hypertension, offspring of mice 33 and rats 34 exposed to late gestational hypoxia, low iron 35 and offspring of preeclamptic women.51,52
In addition to the disruption of endothelium-dependent relaxation in offspring, perinatal exposure to a high salt diet also heightened serum levels of CRP—an important biomarker of systemic inflammation, Interleukin-6 (IL-6), and Tissue Necrotic Factor—Alpha (TNF-α)—markers of vascular inflammation. As observed in this study, the increased MABP in offspring of dams fed a high salt diet could induce endothelial stretching, leading to upregulation of mRNA levels for various pro-inflammatory cytokines, including IL-6 and TNFα, as demonstrated by Loperena et al. 53 The multifaceted mechanisms of IL-6 action in cardiovascular disease pathophysiology stem from its dual role as both pro- and anti-inflammatory. 54 In cardiovascular diseases, IL-6 is synthesized by monocytes, macrophages, endothelial cells, fibroblasts, and vascular smooth muscle cells. 55 While classical IL-6 signaling exhibits anti-inflammatory properties, trans-activation is associated with pro-inflammatory characteristics. 56 Notably, IL-6 has been identified as a significant mediator of salt-stimulated hypertension, 57 with its plasma levels correlating positively with high blood pressure progression.57,58 Furthermore, IL-6 is a key mediator of acute phase reactions, and its levels are linked to CRP levels. 59 Both IL-6 and CRP are utilized clinically as inflammation biomarkers. 59 Clinically, elevated circulating levels of IL-6 and CRP have been associated with increased all-cause mortality, 60 atherosclerosis, 61 and unstable angina. 57 Putatively, the elevated levels of IL-6 and CRP as biomarkers of inflammation suggest that inflammation may precede the vascular dysfunction observed in the offspring consequent of the perinatal exposure of the dams to a high salt diet. Possibly, perinatal exposure of the dams to a high salt diet programed the offspring through genetic or epigenetic means for future salt-sensitive hypertension.
The increase in serum level of Tumor Necrotic Factor-α (TNF-α) in adult offspring of dams fed a perinatal high salt diet further demonstrate the predisposition to inflammation of offsprings exposed to perinatal events such as high salt diet. Tumor Necrotic Factor-α is a critical pro-inflammatory cytokine that plays an important role in the pathophysiology of salt-dependent hypertension. 54 TNF-α was among the first pro-inflammatory cytokines linked to heart diseases, with cardiac myocytes and macrophages producing TNFα, which can operate in an autocrine and paracrine manner to perpetuate inflammation. 62 Renal expression of TNFα has been reported to contribute to hypertension in Dahl salt-sensitive rats. 63 Likewise, etanercept, a TNFα antagonist, has been shown to reduce fructose-feeding induced hypertension, 64 prevent vascular dysfunction, and blunt hypertension induced by angiotensin II, 65 as well as lower blood pressure in an autoimmune model of chronic inflammation. 66
The significance of the elevated biomarkers of inflammation, impaired endothelial function and elevated BP parameters in this study is reinforced by the existence of an NO-mediated link between inflammation and endothelial dysfunction. Nitric oxide is a key player in maintaining vascular health, however, inflammation reduces the bioavailability of NO by impairing its production and increasing its uses and breakdown67,68 thereby precipitating an elevated basal vascular tone—a hallmark of endothelial dysfunction.
Conclusion
Findings from this study suggest that perinatal feeding of dams with a high salt diet increased biomarker of inflammation, impair vascular function and elevated BP in the adult offspring that are placed on postnatal normal salt diet. The implies that pre- and perinatal events such has a high salt diet consumption predisposes adult offspring to hypertension and cardiovascular dysfunction.
Strengths and Limitations of the Study
This study derived its strength from a design that closely mimics what would be expected in a clinical setting. Studying fetal programing of salt-sensitive hypertension in rat model-an altricial animal gives a reproducible insight into how perinatal events such as a high salt diet predisposes to future development of hypertension in the adult offspring. However, this study could derive further strengths from mechanistic studies that differentiate endothelial dysfunction from vascular smooth muscle dysfunction, molecular experiments that evaluate vascular expressions of genes and proteins implicated in vascular function impairment as well as downstream signaling mechanisms of biomarkers of inflammation. Additionally, lack of data on sex-based differences in the inflammation and vascular function is an important limitation of the study.
Footnotes
ORCID iDs
Author Contributions
AOK conceptualize the study and is teh Principal Investigator in the grant that funded the study. SMO, ENO, KBI and EUA conducted the experiments. AAA analysed the data, prepared the manuscript first draft and is a co-investigator in the grant that funded the study. OAS co-conceptualize the study with AOK and he was a co-PI on the grant that funded the study. All authors reviewed and consented to the publication of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the University of Lagos Central Research Grant (CRC NO.—2018/08).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
