Abstract
Left ventricular assist devices (LVADs) have revolutionized the management of patients with advanced heart failure refractory to medical therapy. Current indications of LVADs include Bridge to Transplantation (BTT), Destination Therapy (DT) for long-term use, Bridge to the Decision (BTD) used as a temporary measure, and lastly Bridge to Recovery (BTR). Here, we briefly review the clinical evidence and the molecular mechanisms behind myocardial recovery following LVAD placement. We also share institutional protocols used at 2 major medical centers in the USA.
Introduction
Cardiac myocyte injury may occur due to ischemia and necrosis from limited oxygen supply leading to the loss of cardiac function. The hallmark is ventricular remodeling following an insult resulting from loss of myocytes, myocyte hypertrophy, and alterations in contractile properties of myocytes secondary to β-adrenoceptor downregulation and alteration in calcium homeostasis. 1 In addition, following myocardial injury, the myocytes also experience mitochondrial abnormalities, a disarray of the cytoskeleton cell death (apoptosis, necrosis, autophagy), and extracellular matrix alterations. 2 Off-loading myocardium via an LVAD provides a unique opportunity to reverse these changes.
Clinical Recovery
The benefits offered by LVADs when used as Bridge to Recovery therapy have been well documented in an array of clinical experiments. Several studies have shown that LVADs use results in the successful recovery of myocardium. In the study by Mancini et al, 3 19 patients who had LVADs implanted while waiting for transplantation for dilated cardiomyopathy and coronary artery disease, 5 (26%) patients eventually had the devices explanted after 3 months of use, indicating the potential for myocardial recovery in patients with end-stage heart failure. MacGowan et al 4 also suggested improved functionality of the myocardium when 8 out of 9 patients with prolonged HeartMate II LVADs underwent explantation after a mean duration of 766 ± 343 days.
In the RESTAGE-HF (Remission from Stage D Heart Failure) trial, 40% of patients with nonischemic cardiomyopathy who received an aggressive pharmacological regimen and implantation of HeartMate II LVADs eventually underwent explanation of the devices within 18 months. This demonstrates that physical unloading of left ventricles promotes reverse remodeling leading to myocardial recovery by LVADs. The reverse remodeling by LVADs was found to be more optimized with the use of aggressive pharmacological treatment and regular cardiac monitoring. 5
Several studies aimed to determine the survival rate once the LVAD was decommissioned. In a retrospective review of 515 LVADs patients, Gerhard et al 6 reported that out of 515 patients who received LVADs support, 29 (5%) patients with nonischemic cardiomyopathy showed evidence of myocardial recovery, and 17 (3%) of these patients eventually underwent LVAD explanation. At 73 days after decommissioning, only one patient (6%) developed recurrent heart failure, indicating a long-term device-free survival rate of 94% at the end of 1 year. 6
Monteagudo Vela et al 7 highlighted the reversibility of left ventricular dysfunction achieved by LVADs can be long-term by examining the device-free survival rate at 6 years in patients who underwent explanation after receiving LVADs support for a median of 317 days. In their study, of 470 patients with nonischemic cardiomyopathy with LVADs, 52 (11%) patients underwent explantation. Of these 52 patients, 26 patients with HeartMate II or HeartWare HVAD experienced a long-term device-free survival rate of 87.9% at 1 year, and their survival rate at 6 years only minimally decreased to 77.8%. In addition, the follow-up examination showed an average ejection fraction of 44.25 ± 8.44%, while LVESD and LVEDD were found to be 4.54 ± 0.68 and 5.8 ± 0.46 cm in post-explantation patients at 1 year. 7
Pan et al 8 reviewed 594 patients of HeartMate II and HeartWare HVADs who received LVAD support for a total duration of 500.4 days. Of these, 34 (5%) patients underwent explantation of the devices after experiencing myocardial recovery. The study also identified several positive predictors of myocardial recovery- young age, female sex, low body mass index, and non-ischemic cause of heart failure, highlighting greater benefits of LVADs support when used in patients with nonischemic cardiomyopathy. 8
Analysis from the INTERMACS registry that contains data on commercially approved LVADs implanted in the United States, 1 in 10 LVAD patients experienced either complete or partial myocardial remission with aggressive assessment and frequent weaning evaluations. 9
Boehmer et al 10 analyzed the cohort of subjects in the Intervention in Myocarditis and Acute Cardiomyopathy 2 (IMAC2). Of 373 patients, 14 (3.8%) were identified to be on LVADs support for nonischemic cardiomyopathy. Although the study showed the presence of myocardial inflammation in BTR cohort, lack of fibrosis with lower LV2EDD were favorable predictors of recovery. Out of 14 patients with LVADs, 8 (57%) experienced either complete or partial myocardial injury, and 7 (87.5%) out of those 8 were device free at 19 months of follow-up. 10
Table 1 summarizes favorable characteristics for LVAD explantation.
Favorable characteristics of patients for LVAD explantation.

Molecular Mechanisms
One of the earliest changes that occur in the cardiac myocytes in response to intraventricular wall stress and the burden of volume overload is cardiac hypertrophy. 11 A study by Bruckner et al 12 showed that regression of cardiac hypertrophy by utilization of LVADs is possible by demonstrating an average reduction of myocyte size by 26% (33.1 ± 1.32 to 24.4 ± 1.64 μm) in patients who had LVADs implanted. In addition, reduced fibrosis with less severe alterations in myocardial structure is seen in patients who had LVAD weaned off. 13
Abnormal calcium (Ca2+) handling related to cardiac injury is also associated with decreased contractility. In patients with congestive heart failure, this is evidenced by the depletion of internal Ca2+ stores and impaired function of endoplasmic reticulum Ca2+ ATPASe (SERCA2a). Heerdt et al 14 showed the reverse remodeling of cardiac myocytes supported by LVAD by upregulation of SERCA2a gene expression. LVADs reverse adrenergic remodeling induction in β-adrenergic receptor activity with increased contractile properties of the myocardium. 15
The role of apoptosis in both acute myocardial infarction and congestive heart failure has been studied and demonstrated in the literature. Apoptosis of cardiac muscle cells is known to be a process leading to the progression of heart failure. 16 LVADs result in a fivefold increase in HSP72, an important antiapoptotic factor that prevents cell death. 17 Manginas et al 18 noted an increased number of circulating CD34+ progenitor cells as well as decreased BNP levels in patients with LVADs.
In congestive heart failure, proinflammatory cytokines and chemokines including Tumor Necrosis Factor-α (TNF-α), interleukin-β (IL-1β), interleukin-6 (IL-6) are increased which results in further activation of the renin-angiotensin system leading to cachexia and anemia. 19 In their study, Torre-Amione et al 20 showed a reduction of TNF in patients on LVADs. The suppressor of tumorigenicity 2 (ST2) is a member of the interleukin family, and the soluble form of ST2 is known to affect cardiac fibrosis. 21 Tseng et al 22 showed a significant reduction of sST2, with normalization of levels 3 months after LVAD placement, indicating a reduction in fibrosis.
LVADs also result in the upregulation of genes encoding both sarcomeric and non-sarcomeric proteins. 23 Cardiolipin is a mitochondrial inner protein that is involved in the maintenance of integrity and regulation of the electron transport chain. Post LVAD placement, Heerdt et al 24 demonstrated normalization of cardiolipin composition in ischemic cardiomyopathy, in the setting of reduced mitochondrial stress.
The association between congestive heart failure and the overactivation of the Renin-Angiotensin-Aldosterone System (RAAS) has been well-documented for over a decade. Welp et al 25 showed plasma renin and aldosterone levels fell close to normal in both pulsatile and non-pulsatile LVADs, however, the effect was more pronounced in pulsatile LVADs. Inhibition of the RAAS pathway with ACE inhibitors and ARBs in patients with LVADs has been shown to limit myocardial remodeling, reduce arterio-venous malformations, and improve overall survival. 26
Explantation Protocol at Tertiary US Centers
The RESTAGE-HF trial has represented a major turning point in the pursuit of LVAD explantation through the development of a standardized protocol. This protocol has now become standard practice for many institutions including the University of Washington (UW). The institutional protocol at UW is one that essentially mirrors the protocol designed by the RESTAGE-HF trial. 27
Per this protocol, patients undergo aggressive pharmacologic management based on the HEAAL study. 28 These include ACE-inhibitor or ARB, beta-blockers and aldosterone antagonists, with close follow-up of renal function, electrolyte management, as well as targeted mean arterial pressure (MAP) of >60 mmHg. An initial clinical assessment is then conducted which includes baseline echocardiography and right heart catheterization. Patients are then followed serially at 6 weeks, 3, 4, 5, 6, 12, and 18 months with repeat blood tests and echocardiography. 27
For the assessment of explantation, the functional status of the patient as well as objective parameters gathered from echocardiography and invasive hemodynamic testing are used collectively to determine whether a patient is stable for an explantation attempt. These criteria, as outlined in the RESTAGE-HF trial include the following:
LVEDD < 60 mm, LVESD < 50 mm, LVEF > 45%
PAWP ⩽ 15 mmHg
Resting cardiac index (CI) > 2.4L/min/m2
Maximal oxygen consumption (VO2 max) with exercise >16 mL/kg/min
[LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; PAWP, pulmonary artery wedge pressure].
The institutional protocol implemented by Maine Medical Center (Figure 1) assesses similar parameters for patients who have shown promising signs of myocardial recovery that are considered for weaning off the LVAD support. Five patients since 2014 have successfully been explanted following the outlined protocol. Patients must be on optimal guideline-directed heart failure (HF) medical therapy adopted by the American Heart Association (AHA)/American College of Cardiology (ACC) guidelines for the management of chronic heart failure. After the pharmacological regimen, patients undergo an echocardiogram on a 3 months basis, and those who meet the following criteria such as left ventricular ejection fraction (LVEF) >45%, normal right ventricular (RV) function, absence of significant valvular disease, and significant and sustained reduction in left ventricular end-diastolic dimension(LVEDD) as compared to pre-implantation function, LVEDD < 55 mm, LVESD < 48 mm, are followed through the explanation protocol and scheduled for ramp echocardiogram to optimize the LVAD speed. Once on the ramp echocardiogram, patients are further monitored to demonstrate clinical stability and continued myocardial recovery by tolerating exercise tolerance at decreased speed of the LVAD device. The devices are set to decrease the speed every 5 minutes by 100 RPM(HVAD)/400 RPM(HM2)/200 RPM(HM3) RPM to a no-net flow speed of 2000 RPM (HVAD)/6000 RPM (HM2)/4000 RPM(HM3) in 15 minute duration. After this trial, they are then assessed for LVEF, LVEDD, and LVESD, quantity of valve regurgitation, and aortic valve opening and a right heart catheterization is performed, to lead to the final decision to explant the LVAD device.
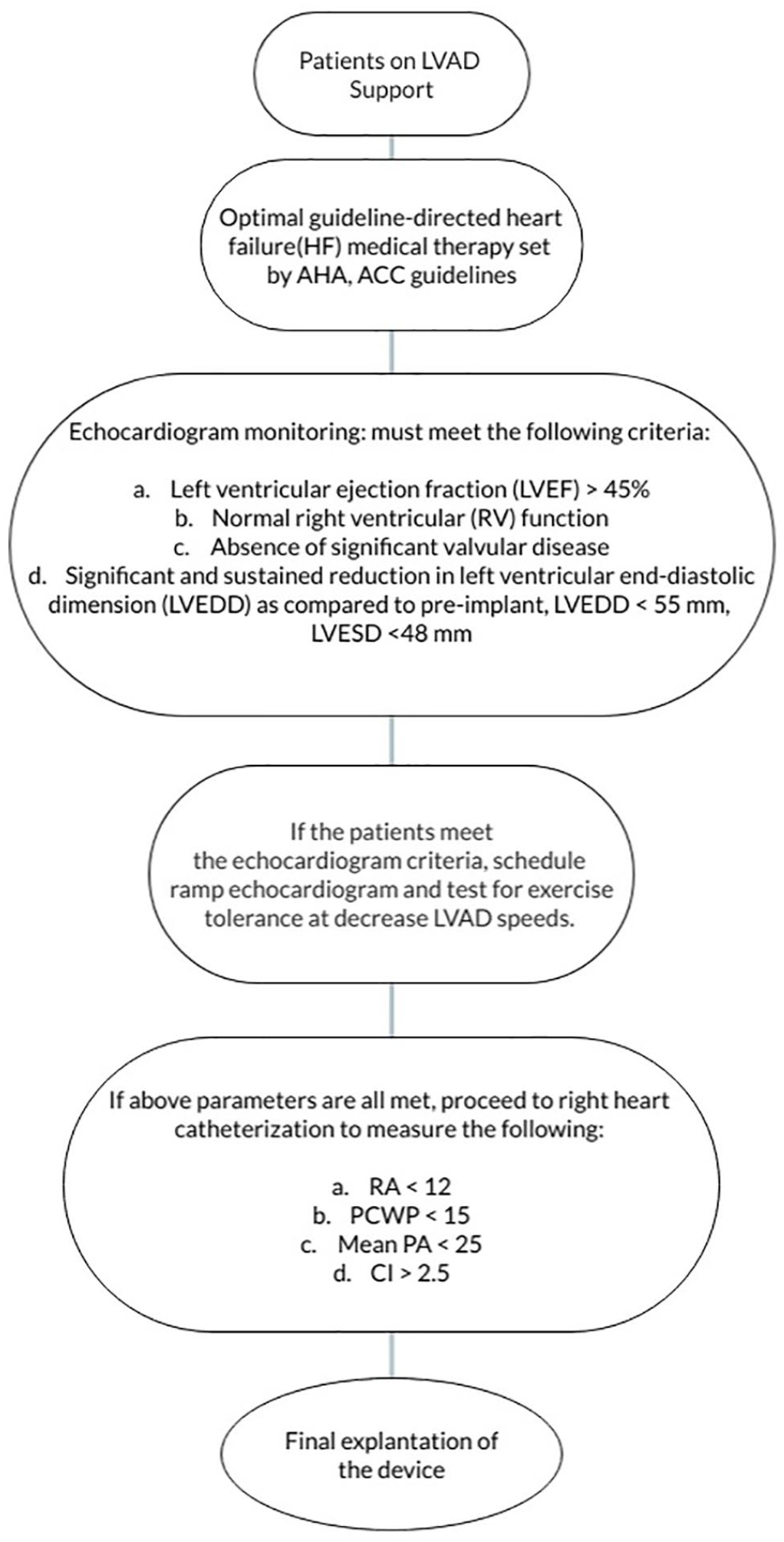
Flow diagram shows LVAD wean to explant protocol by marine medical center.
Conclusion
For more than a decade, the benefit of cardiac myocyte reverse remodeling that occurs by the utility of LVADs was limited to the remission state. However, several recent experimental studies have demonstrated the possibility of long-term recovery rather than temporary remission in patients with severe advanced heart failure after implantation of LVADs in addition to pharmacological therapy. It is important to continue the investigation of LVADs role in myocardial recovery, as LVADs have revolutionized outcomes of advanced heart failure patients as limited numbers of hearts are available for transplant.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Literature review: SA, RC, DS
Drafting of manuscript: SA, RC, DS.
Manuscript revision, intellectual revisions, mentorship: YG, MY, AA, CM, GV.
Final approval: RC, SA, DS, YG, MY AA, CM, GV
