Abstract
Neuroendocrine tumors (NETs, originally termed “carcinoids”) create a relatively rare group of neoplasms with an approximate incidence rate of 2.5 to 5 cases per 100 000 persons. Roughly 30% to 40% of subjects with NETs develop carcinoid syndrome (CS), and 20% to 50% of subjects with CS are diagnosed with carcinoid heart disease (CaHD). The long-standing exposure to high serum serotonin concentration is one of the crucial factors in CaHD development. White plaque-like deposits on the endocardial surface of heart structures with valve leaflets and subvalvular apparatus thickening (fused and shortened chordae; thickened papillary muscles) are characteristic for CaHD. NT pro-BNP and 5-hydroxyindoleacetic acid are the 2 most useful screening markers. Long-acting somatostatin analogs are the standard of care in symptoms control. They are also the first-line treatment for tumor control in subjects with a metastatic somatostatin receptor avid disease. In cases refractory to somatostatin analogs, several options are available. We can increase a somatostatin analog to off-label doses, add telotristat ethyl or administer peptide receptor radionuclide therapy. Cardiac surgery, which mainly involves valve replacement, is presently the most efficient strategy in subjects with advanced CaHD and can relieve unmanageable symptoms or be partly responsible for better prognosis.
Keywords
Introduction
Neuroendocrine tumors (NETs, originally termed “carcinoids”) create a relatively rare group of neoplasms with an approximate incidence rate of 2.5 to 5 cases per 100 000 persons. Mostly they stem from the gastrointestinal tract, in particular from the proximal part of the colon and the distal part of the small intestine. NETs predominantly demonstrate indolent disease biology for many years. They become symptomatic when they are large enough or when they metastasize to the liver (mostly) or the lungs, bones, or other sites (less commonly). 1
Roughly 30% to 40% of subjects with NETs develop carcinoid syndrome (CS), mostly in subjects with liver metastases. Only then vasoactive substances released by the carcinoid are not deactivated in the liver and reach the systemic circulation through the hepatic vein. Approximately 5% of subjects with ovary, lung, or retroperitoneal metastases manifest with CS without having metastases to the liver. Next, 20% to 50% of subjects with CS are diagnosed with carcinoid heart disease (CaHD). 2 CaHD is also called Hedinger’s disease (syndrome). 3
CaHD is the leading source of mortality and morbidity in the CS population. The outcomes of CaHD without treatment are poor, and the 3-year survival rate is 31% versus 68% in subjects with and without CaHD, respectively. 4 Fortunately, the outcomes of subjects with CaHD have significantly improved over the last decades. It is mainly associated with the progress in cardiac imaging modalities, CaHD therapies, as well as with cardiac valve surgery. The median survival accrued from 1.5 years to 4.4 years in the over 20 years. 5
Pathophysiology
The pathophysiology of CaHD is multi-faceted and not entirely known. The long-standing exposure to high serum serotonin concentration is one of the crucial factors, but probably several other vasoactive peptides secreted by tumor cells are engaged (Table 1). 6 The serotonin receptor type 5-HT2B is most abundant in the heart. The stimulation of 5-HT2B increases the mitogenic activity of cardiomyocytes and fibroblasts. It results in the release of proinflammatory cytokines and the transforming growth factor β (TGFβ). 7
Factors playing part in the pathophysiology of carcinoid heart disease.

White plaque-like deposits on the endocardial surface of heart structures with valve leaflets and subvalvular apparatus thickening (fused and shortened chordae; thickened papillary muscles) are characteristic for CaHD. 6 These deposits comprise smooth muscle cells, myofibroblasts, and extracellular matrix (acid mucopolysaccharides, collagen, elastin). In the vast majority (90%!), deposits are found in the right heart. In consequence, we observe the involvement of tricuspid as well as pulmonary valves. The left heart is involved when a high burden of circulating vasoactive peptides exceeds the degradation capacity of the liver and lungs, in case of bronchial carcinoids and subjects with patent foramen ovale (PFO)—even 1/3 of cases. The disease advancement may vary from slightly thickened leaflets and mild valve insufficiency to fixed retraced leaflets with severe regurgitation and mild/moderate stenosis. Rarely, subjects may present with metastases to the heart in extremely advanced disease. 8
Diagnosis
Clinical presentation
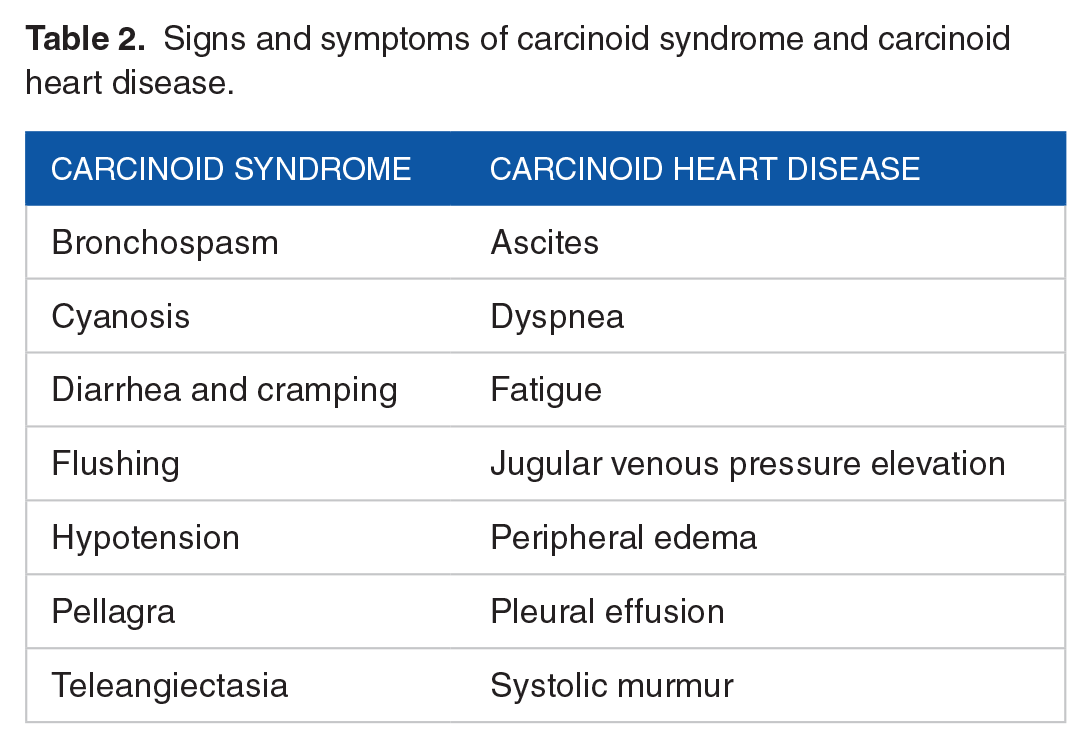
Cardiac signs and symptoms are frequently subtle or absent at the early stages of the disease, and therefore recognition of CaHD may be postponed. Transthoracic echocardiographic (TTE) is the imaging modality of choice in this population. 9 The most common manifestations of the CS, as well as CaHD, are presented in Table 2. However, the CaHD diagnosis might not be so obvious. Over 50% of subjects with moderate/severe tricuspid insufficiency may be asymptomatic or present slightly accented symptoms. Moreover, 1/3 of these subjects may not present any systolic murmur. 10 If present, the systolic murmur is heard best at the left border of the sternum, and its intensity enhances when inspiring. Worth stressing is the fact that peripheral edema, ascites, or pleural effusion may not be caused by heart failure. They may be associated with liver insufficiency, malnutrition, or venous obstruction of the tumor.
Signs and symptoms of carcinoid syndrome and carcinoid heart disease.
Biomarkers
N-terminal pro-B-type natriuretic peptide (NT-proBNP) is the most appropriate marker with validated both diagnostic and prognostic value for heart diseases. 12 In subjects with CS the cut-off value of NT-proBNP in CaHD screening is 260 pg/mL (31 pmol/L). Bhattacharyya et al disclosed that median NT-proBNP value was significantly higher in subjects with CaHD (median: 1149 pg/mL) comparing with CS subjects without cardiac disease (median: 101 pg/mL; P < .001). 13 The sensitivity, specificity, negative predictive value and positive predictive value of NT-proBNP threshold of 260 pg/mL (31 pmol/L) for CaHD recognition were 92%, 91%, 98%, and 71%, respectively.
5-Hydroxyindoleacetic acid (5-HIAA; a product of degradation of 5-HT by monoamine oxidases in the brain, liver, and lungs), chromogranin A (CgA; a glycoprotein produced by NETS) and activin A (a TGF-β superfamily member) are other useful markers in the CaHD assessment. In fact, for diagnosis and follow-up of CS subjects, it is necessary to measure 24-hour urine 5-HIAA or plasma 5-HIAA, and the level >300 mmol of 24-hour urinary 5-HIAA helps identify subjects with the increased risk of CaHD development. Nevertheless, one must remember that certain products rich in tryptophan may cause false-positive results (avocados, bananas, eggplants, kiwis, pineapples, plantains, plums, tomatoes, walnuts). 14 Also, drugs can affect the 5-HIAA measurement and can evoke false-positive results (acetaminophen, caffeine, diazepam, ephedrine, glyceryl guaiacolate, nicotine, phenobarbital) as well as false-negative results (aspirin, phenothiazines, levodopa). 15 Recently, Joish et al published a meta-analysis characterizing 24-hour urinary 5-HIAA levels and mortality in subjects with NETs in the years 2007 to 2017. The mean 5-HIAA value was 149.2 mg/24 h (reference range 3.0-15.0 mg/24 h), whereas the total mortality incidence was 13.0% (95% CI 9.0%-20.0%). Subjects with CS had a mean 5-HIAA value of 161.0 mg/24 h, and the mortality incidence was 8.2%. Interestingly, subjects with CaHD showed a mean 5-HIAA value of 199.2 mg/24 h, and the mortality incidence was 27.8%. The authors disclosed that every 10-unit increase in 5-HIAA level was associated with 11.8% (95% CI 8.9%-17.0%) increase in 12-month mortality incidence. 16
Worth remembering is the fact that serum 5-HIAA level measurement stands another option to screen for CS. The plasma 5-HIAA can serve as a surrogate for urine 5-HIAA. It is more comfortable to be measured than a 24-hour urine collection. 17
CgA characterizes a sensitivity of up to even 100%, but a specificity of only 30% in anticipating advanced CaHD. In consequence, CgA value is a more useful tool for assessing NETs relapse or progression, and CgA is not recommended as a screening test for CaHD. In short, CgA is not an adequate biomarker for CaHD. 1
Also, activin A must be mentioned. Bergestuen et al studied 71 subjects with NETs and 18 subjects as the control group. Activin A threshold ⩾0.34 ng/mL characterized high sensitivity (87%), but low specificity (57%) in recognizing CaHD (P = .006). 18
Imaging modalities
Transthoracic echocardiography (TTE) is the gold standard for the diagnosis and monitoring of subjects with CaHD. The proposed algorithm is summarized in Figure 1. 19 Bhattacharyya et al thoroughly circumscribed features of CaHD in 2D or 3D echocardiography as well as in cardiac magnetic resonance (CMR). 11 TTE must be performed in every subject with CS and with a high risk of CaHD, especially in subjects with increased levels of NT-proBNP or 5-HIAA. In subjects with established CaHD, echocardiography is recommended to perform when the clinical status is significantly changing or every 3 to 6 months. The rigorous use of 2D/3D TTE, color-flow, and spectral Doppler features are required to ensure comprehensive evaluation, especially in the case of pulmonary valve involvement. Moreover, 3D TTE or 3D transesophageal echocardiography are useful in verifying valve involvement as well as in evaluating the right ventricular size and function (Table 3).

Screening algorithm for carcinoid heart disease.
Echocardiographic features of carcinoid heart disease (based on 11 ).

Several echocardiographic scoring systems were developed to quantify CaHD severity. Dobson et al performed a prospective study in 100 NETs subjects with metastases. 20 Six echocardiographic scoring systems were assessed.12,20 -24 All examined systems correlated well with NT-proBNP and serum 5-HIAA values. They were comparable in terms of sensitivity and specificity for CaHD recognition (Table 4). However, in clinical practice, 2 of them are most feasible, that is, systems proposed by Denney et al 21 and by Westberg et al. 24
Comparison of echocardiographic scores in subjects with carcinoid heart disease (modified after 20 ).

Abbreviations: AUC, area under the curve; CaHD, carcinoid heart disease.
CMR imaging may assess the pulmonary valve, recognize cardiac metastases, and verify the right ventricle size and function. Interestingly, Timmermans et al determined the utility of exertional CMR joined with invasive pulmonary artery measurements. They showed that not pressure but volume overload was crucial in the development of the symptoms until the right ventricular function was preserved. 25 Cardiac computed tomography (CT) imaging is also applicable in recognizing heart valves pathology, especially of the pulmonary valve, in assessing right ventricle status, and in the pre-operative imaging of coronary arteries. 26 Somatostatin receptor PET/CT might also be applied in the diagnosis and assessment of CaHD. 27
Treatment
The treatment of subjects with CaHD is multi-faceted due to the necessity to manage simultaneously the systemic cancer disease as well as the heart pathology. When choosing appropriate therapies, the following factors must be evaluated: (1) overall performance status as well as nutritional status and; (2) NETs stage (stable disease, progressive or metastatic disease); (3) function of crucial organs (mainly kidney and liver); and (4) the impact of CaHD on the proceedings of the underlying NETs. In the management of this population, 3 elements can be differentiated: (1) heart failure therapy, (2) intensive lowering of 5-HIAA value below the level of 300 mL/24 h, and (3) cardiac valve surgery. 18
Pharmacotherapy
Presently, long-acting somatostatin analogs are the mainstay option to control CS signs and symptoms and to counteract the development or progression of CaHD. Its efficacy in achieving a clinical and biochemical response is approximately 60% to 70%.28,29 Octreotide and lanreotide are 2 commercially available somatostatin analogs. Short-acting octreotide (half-life: 1.5-2 hours) must be given 2 to 3 times per day subcutaneously or via intravenous infusion. However, long-acting formulations are available such as octreotide LAR (long-acting repeatable), lanreotide long-acting, and lanreotide autogel. 30 As a result, octreotide LAR is administered in doses of 10, 20, or 30 mg every 4 weeks, and lanreotide autogel is prescribed in doses of 60, 90 or 120 mg every 4 weeks. In the case of refractory symptoms, one may consider administering increased doses more frequently. Strosberg et al proved that octreotide dose escalation (40-60 mg per month) resulted in flushing and diarrhea decrease in 56% and 63% of subjects, respectively. 31 Interestingly, Diamantopoulos et al demonstrated that the use of above-label doses of somatostatin analogs in gastroenteropancreatic NETs was associated with improved progression-free survival. The best results were observed in subjects with small intestine/colorectal primaries, Ki-67 below 5%, and no/limited extrahepatic metastases. 32
In certain subjects, especially asymptomatic with small carcinoid burden and 5-HIAA levels within normal limits, initial watchful waiting might be adopted. Nonetheless, in all subjects with substantial serotonin production, it is reasonable to initiate the therapy with somatostatin analogs. It aims to decrease urinary 5-HIAA secretion below 300 mmol/24 h. Moreover, they are now more eagerly used also in asymptomatic subjects. PROMID and CLARINET trials proved that such approach was associated with antiproliferative potential and positive impact on progression-free survival.33,34
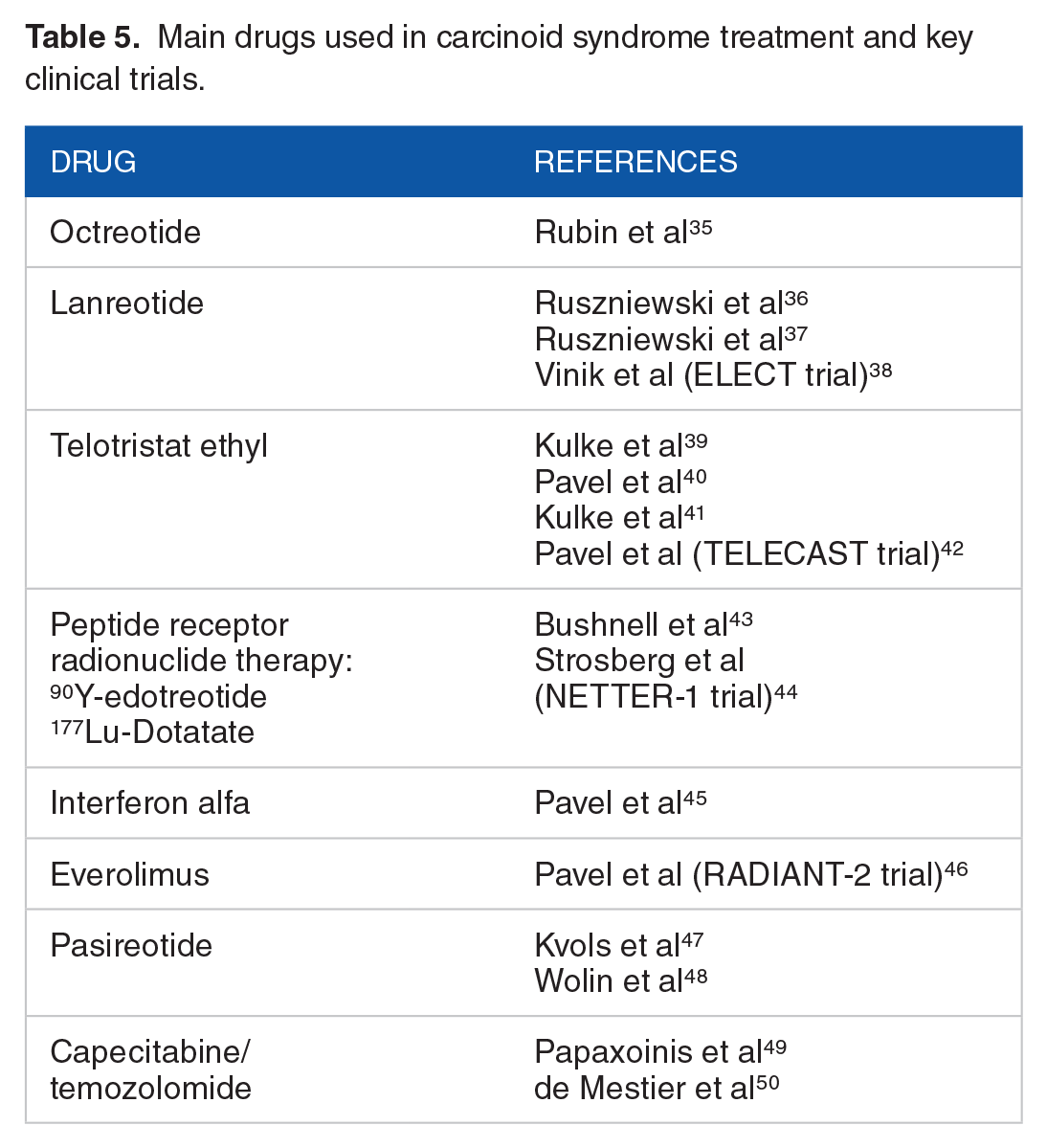
In subjects refractory to somatostatin analogs therapy, one may escalate the somatostatin analog administration to off-label doses, add telotristat ethyl, or initiate peptide receptor radionuclide therapy. When needed, interferon alfa might be administered, whereas everolimus and pasireotide are not recommended. Also, temozolomide and capecitabine (CAPTEM) chemotherapy can be a potent debulking option in pancreatic and lung NETs, and in well-differentiated small bowel NETs with higher Ki-67 indexes (Table 5).
Main drugs used in carcinoid syndrome treatment and key clinical trials.
Telotristat ethyl is the inhibitor of tryptophan hydroxylase, and it is the newest drug registered for the treatment of CS (February 2017). A phase 3 TELECAST trial evaluated the safety profile and effectiveness of telotristat in subjects with CS (main symptoms: nausea, abdominal pain, diarrhea, flushing, and increased urinary 5-HIAA levels) within a 3-month double-blinded treatment period and subsequent a 36-week open-label extension. The primary endpoints were the treatment-emergent adverse events incidence and the percentage change of 24-hour urinary 5-HIAA between the baseline value and after 12 weeks. Subjects were randomized (1:1:1) to receive a placebo, telotristat 250 mg, or telotristat 500 mg 3 times a day. After 3 months, a significant reduction in urinary 5-HIAA was registered. The results of this study confirmed the safety profile and effectiveness of telotristat when added to somatostatin analogs therapy in subjects with CS. 42
Non-cardiac intervention
Transcatheter arterial embolization is an efficient method in decreasing tumor burden and hormone levels in subjects with substantial metastases in the liver. Embolization and chemoembolization result in clinical response in 50% to 100% of subjects with liver involvement. 51 Nonetheless, it must be applied cautiously in subjects with advanced CaHD and right ventricle dysfunction or substantial liver involvement since adverse events (bleeding or liver failure) might appear. Also, total portal vein occlusion, poor overall performance status, and liver failure are perceived as relative contraindications to such therapy. 52
Recently, liver cytoreductive surgery has arisen as a substantial advance in the management of CS. 53 Partial hepatic resection decreases tumor burden and improves the quality of life and overall survival. Nonetheless, such procedures might be challenging as well as life-threatening. They should be performed after heart valve repair. Kinney et al published the perioperative results of 169 subjects after partial hepatic resection due to metastatic NETs between 1997 and 2015 at Mayo Clinic Rochester. 54 The most common adverse events were sustained tachycardia (8.9%), hypotension (5.3%), flushing (0.6%), cardiac conduction abnormalities (0.6%) and acidosis with pH < 7.2 (0.6%). No cases of carcinoid crisis were registered. Reoperation was required in 5.9%, renal dysfunction was observed in 3% of cases, biliary dysfunction—in 3.6%, and death—in 1.2%.
Cardiac surgery
Cardiac surgery, mainly valve replacement, is the most efficient management option in subjects with advanced CaHD. It may relieve persistent symptoms and improve prognosis. 55 Connolly et al observed substantial clinical improvement after valve surgery. At 2 years, the survival rate was 40% compared with 8% in subjects with advanced symptomatic CaHD managed without surgery. 56
Tricuspid valve insufficiency is the most common abnormality in subjects with CaHD, and all subjects with severe tricuspid valve insufficiency and without contradictions should be qualified for valve replacement. In the case of the right ventricular outflow tract (RVOT) obstruction and pulmonary valve insufficiency, subjects are eligible for pulmonary valve replacement and RVOT enlargement with a patch. In subjects with mitral or aortic valve disease, valve replacement or repair may be considered, depending on the severity of the disease. In selected subjects, adjunctive procedures may be performed, such as PFO closure, as well as the excision of carcinoid metastasis. 19 Also, in sporadic cases, quadruple valve replacement/repair might be required. 57
The optimal timing of the valve replacement procedure is not unequivocally identified. In general, subjects with CaHD are qualified for cardiac surgery when they start presenting signs, and symptoms of heart failure (especially right heart failure) is revealed in imaging modalities. Moreover, subjects should characterize post-operative life expectancy above 1 year.19,53
The choice of the implanted prosthesis type must be chosen individually, taking into consideration the bleeding risk, the NET-related overall survival, and possible future therapeutic interventions. In consequence, bioprosthetic valves are mainly preferred since CaHD subjects characterize elevated bleeding risk associated with extensive liver disease and hepatic malfunction. Moreover, in these subjects, the increased probability of future invasive procedures requiring temporary withdrawal of anticoagulants is observed. Also, right-sided bioprosthetic valves are less prone to thrombosis than mechanical ones. The newest generation of bioprosthetic valves is also of higher durability.19,53
Recently, Nguyen et al analyzed the impact of different variables on outcomes in 240 adult subjects undergoing cardiac surgery for CaHD. 58 Tricuspid valve replacement was performed in 99%, pulmonary valve operation—in 84%, mitral valve operation—in 11%, and aortic valve operation—in 10%. In 73% of cases, 2 valves were operated simultaneously. Early 30-day mortality was 9%. Only older age (OR 2.31, 95% CI 1.30-4.11) and higher NYHA class (OR 2.67, 95% CI 1.31-5.45) were linked with early mortality. At 5-years, the mortality rate was 66%, and similarly, it was statistically significantly affected by age (HR 1.55, 95% CI 1.22-1.97), and NYHA class (HR 1.53, 95% CI 1.15-2.03).
Carcinoid crisis is a potentially life-threatening complication in subjects with CS undergoing surgery as well as when exposed to anesthetic drugs or other medicines (eg, vasopressors or opioids). Carcinoid crisis is evoked by the release of substances with biological activity what causes hypotension (rarely blood pressure incline), tachycardia, arrhythmias, flushing, wheezing as well as symptoms from the central nervous system. In prevention in the perioperative period, subjects with CaHD should receive octreotide venous infusion with a flow rate of 50 to 100 mg/h for 12 hours before surgery. The infusion should be maintained throughout the operation and then thereafter until the patient’s state stabilization. If signs or symptoms of carcinoid crisis develop, an additional bolus (100-200 mg) of octreotide must be given and repeated as needed with a simultaneous increase of octreotide infusion rate to 200 mg/h. 30
Follow-Up
Recently, Lesen et al published research on healthcare resource use and costs in subjects with CS and CaHD. 59 Total resource cost was 15 500 € per subject in well-controlled CS cases and increased to 21 700 € per subject in uncontrolled CS cases, what gave the incline of approximately 40% (6200 € per patient). Excluding somatostatin analogs cost, the key drivers for cost incline in CaHD subjects were tumor-related surgical procedures and examinations, mainly echocardiography.
To obtain as best as possible outcomes after the surgery, the optimal follow-up management is crucial. One can recommend the approach as follows: 19
Subjects after valve replacement with a biological prosthesis are urged to continue therapy with warfarin for 3 to 6 months;
After surgery TTE should be performed as early as possible;
After discharge, the first TTE should be performed after 3 to 6 months (at the moment of anticoagulation withdrawal), and then every 6 to 12 months;
Intensive control of CS with target 5-HIAA level below 300 mmol/L is recommended.
Conclusion
CaHD is the leading source of mortality and morbidity in subjects with CS. The CaHD outcomes without the treatment are poor. Fortunately, the prognosis of CaHD subjects has significantly improved over the last decades, which is mainly associated with the progress in cardiac imaging modalities, CaHD therapies as well as with cardiac valve surgery. Although improved systemic therapies have delayed the onset of CaHD in many NETs subjects with CS, new treatment options are required since recently only 1 drug was registered in CS treatment, that is, telotristat ethyl.
Footnotes
Authors’ Contributions
All authors contributed extensively to the work presented in this literature review.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
