Abstract
Background:
Abnormal vascular reactivity and reduced expression of endothelial nitric oxide synthase (eNOS) gene are hallmark of salt-induced hypertension in rats. Although
Objectives:
This study was designed to investigate the mechanism by which oral
Methods:
Forty-eight weaned male Sprague-Dawley rats of weight range 90 to 110 g were randomly divided into 6 groups of 8 rats per group. Group I was fed normal rat chow ad libitum and served as the Normal Diet group. Group II was fed a diet that contained 8% NaCl. Groups III and IV took normal and high-salt diet, respectively, and then received oral
Results:
Our results show that high-salt diet significantly reduced (P < .05) endothelium-dependent relaxation response to acetylcholine and qualitatively reduced eNOS gene expression in the abdominal aorta of the rats. However,
Conclusion:
This study suggests that oral supplementation of
Keywords
Background
Sustained increase in blood pressure is arguably the most important cardiovascular risk factor.
1
High dietary salt intake has been shown to lead to this sustained increase in blood pressure in different animal species and humans.2-4 Vascular function impairment is known to be part of the key mechanisms by which high-salt diet leads to the increase in blood pressure.5-7 In the endothelial cells, endothelial nitric oxide synthase (eNOS) helps in the production of citrulline and nitric oxide from
Although
Methods
Experimental animals
Forty-eight (48) weaned male SD rats of weight range 90 to 110 g were used for this study. The rats were divided into 6 groups of 8 rats per group (Oloyo et al., 2011) by simple random sampling such that the mean difference in weight across the groups was not statistically significant (P > .05). These rats were obtained from the Laboratory Animal Center, College of Medicine of the University of Lagos. The rats were acclimatized for 2 weeks before the commencement of the study. All through the period of the study, depending on the groups, they were either fed with normal rat chow containing 0.3% salt or high-salt diet containing 8% salt (Sofola et al., 2002; Oloyo et al., 2011; Oloyo et al., 2016; Adejare et al., 2017). The rats were housed in transparent cages where they had free access to food and clean water. They were maintained in a well-ventilated environment under standard environmental conditions (28°C-30°C, 12-hour light/12-hour dark cycle).
Ethical statement
This protocol was approved by the Health Research Ethics Committee of the College of Medicine of the University of Lagos (Approval number CM/HREC/12/16/080). All procedures for this study were carried out in strict adherence to the National Institutes of Health Guide for the care and use of laboratory animals. 17
Study design
Rats were randomly divided into 6 groups containing 8 rats per group by an observer unaware of the treatment groups. Group I rats were fed normal rat chow ad libitum and served as the Normal Diet group. Group II rats were fed a diet that contained 8% NaCl.6,7 Groups III and IV rats took normal and high-salt diet, respectively, and then received oral
Grouping of animals.

Abbreviation: L-NAME,
Experimental procedure
Preparation of abdominal aortic rings
Rats were sacrificed by cervical dislocation. Immediately after dislocation, the abdominal region was opened and the abdominal aorta cut out and placed in a Petri dish containing cold Physiological Salt Solution (PSS) at 4°C. The abdominal aorta was freed of fat and connective tissues. The aorta was then cut into ring segments of about 2 to 3 mm. The ring was thereafter mounted between one long and one short stainless steel hooks. The small S-shaped hook was attached to the base of the organ bath, while the long L-shaped rod was attached to the isometric force transducer (top force transducer MLT 050/D; AD Instruments, Bella Vista, Australia) that was attached through MLAC11 Grass adapter cable to a computerized data acquisition system with LabChart-7 pro software (Power Lab-4/24T, model MLT844/P; AD Instruments Pty Ltd., Castle Hill, Australia). During this procedure, special care was taken to avoid rubbing the endothelial surface of the rings. The 20 mL organ bath contained PSS with the composition: 119.0 mol/L NaCl, 4.7 mol/L KCl, 1.2 mol/L KH2PO4, 1 to 2 mol/L MgSO4, 24.9 mol/L NaHCO3, 1.6 mol/L CaCl2, and 11.5 mol/L glucose at 37°C. The pH of the PSS was adjusted to 7.4 and the set-up gassed with 95% O2:5% CO2 mixture. 6
For each ring, a passive tension of 1.5 g was applied and the ring was allowed to equilibrate for 90 minutes in the PSS during which at 30 minutes interval, it was subjected to a dose of 10−6 M noradrenaline. The ring was rinsed after each stimulations. This 90 minutes stabilization was necessary to ensure a consistent response during the experiment. After this stabilization period, the relaxation response to graded doses of acetylcholine (10−9-10−4 M) and sodium nitroprusside (SNP; 10−10-10−4 M) was assessed separately in the absence and presence of L-nitro-arginine-methyl-ester (L-NAME, 10−4 M) and methylene blue (MB, 10−5 M) following pre-contraction with 10−7 M NA. In the presence of the inhibitors, the incubation lasted for 30 minutes before pre-contraction and application of graded doses of acetylcholine or SNP. Endothelium-intact rings were used for acetylcholine-induced relaxations, while endothelium-denuded rings were used for SNP-induced relaxations. All experiments took place under the same environmental temperature and pressure.
Gene expression of eNOS
Dissection
Following cervical dislocation of the rats for gene expression studies, the abdominal region was opened and the abdominal aorta excised. The abdominal aorta was placed on a phosphate-buffered saline (PBS) pre-cooled plate. The adherent connective tissues were then gently removed and the tissue frozen in liquid nitrogen (−170°C) for subsequent analysis. During dissection, special care was taken to avoid unnecessary stretching of the abdominal arteries.
RNA isolation
Ribonucleic acid (RNA) extraction was carried out in the cut segments of the abdominal aorta using AurumTM Total RNA Mini Kit (Catalog #732-6820; Bio-Rad Laboratories Inc., Hercules, CA, USA). The extracted RNAs were then treated with RNase-free DNase and the concentrations determined by ultraviolet (UV) light absorbance with wavelength set at 260 nm.
Reverse transcription polymerase chain reaction
With the aid of an Oligo-dT primer, reverse transcription polymerase chain reaction (RT-PCR) was carried out to synthesize cDNA from the extracted RNA using an Omniscript RT Kit. The synthesized complementary DNA (cDNA) fragments were then amplified using HotStar HiFidelity Polymerase Kit. The primer sequence used for the amplification is as illustrated in Table 2. A thermal cycler (iCycler; Bio-Rad Laboratories, Inc., Hercules, CA, USA) with the operating conditions set at 5 minutes denaturation, 30 seconds at 94°C; annealing, 40 seconds at 57°C; and a 1 minute extension step at 72°C was used to perform the PCR cycles. A final extension step of 5 minutes at 72°C followed the last cycle to complete the procedure. To check for genomic DNA contamination, samples were subjected to the same PCR procedure in the absence of the reverse transcriptase enzyme. The process was finalized by separating the PCR products by gel electrophoresis. The gel prepared consisted of 1.5% agarose. The products were visualized by ethidium bromide staining under ultraviolent light. The house-keeping gene used for the study was glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and all results were presented relative to its expression. Prior to the commencement of the study, database search of GenBank was performed with BLASTN.
PCR primers and PCR protocols.

Abbreviations: eNOS, endothelial NOS; GAPDH, glyceraldehyde-3-PO4 dehydrogenase; PCR, polymerase chain reaction.
Experimental outcomes
The experimental outcomes for this study include the maximum relaxation to acetylcholine or SNP, −Log EC50, and expression level of the eNOS gene in each group. Relaxation responses following pre-contraction with noradrenaline were calculated and used to assess vascular reactivity. Abdominal aortic rings were obtained from each animal and used for vascular reactivity. Results from the first 8 viable rings from the 6 animals were used in the statistical analyses. Abdominal aortic rings that were not viable were discarded.
Statistical analyses
The collected data were expressed as means ± standard error of mean (SEM) and were analyzed using one-way analysis of variance (ANOVA; independent group is categorical vs dependent group, ie the outcome that is continuous) followed by Student-Newman-Keuls post hoc test (because there were mean differences in some groups). A P-value less than .05 (P < .05) was considered significant. For the vascular reactivity part, GraphPad Prism 5 software (GraphPad Software, Inc., La Jolla, CA, USA) was used to generate −Log EC50 values, which were subsequently subjected to statistical analysis.
Results
Effect of salt-load and l -arginine supplementation on relaxation response to acetylcholine
Relaxation responses were significantly reduced (P < .01) in salt-loaded group (maximal response of 32.24% ± 2.74%) compared to Normal Diet (maximal response of 81.29% ± 4.92%). There was a significant (P < .01) increase in −Log EC50 value in the salt-loaded group compared to normal diet (Table 3). In the groups supplemented with oral
−Log EC50 and percent maximum relaxation response of abdominal aortic rings to acetylcholine with or without L-NAME.

Abbreviations: ND, normal diet; SD, salt diet.
Data are presented as mean ± SEM.
Significantly higher (**P < .01) compared with corresponding normal diet group. Significantly lower (‡P < .05, ‡‡P < .01) compared with corresponding normal diet group (n = 6 rings).
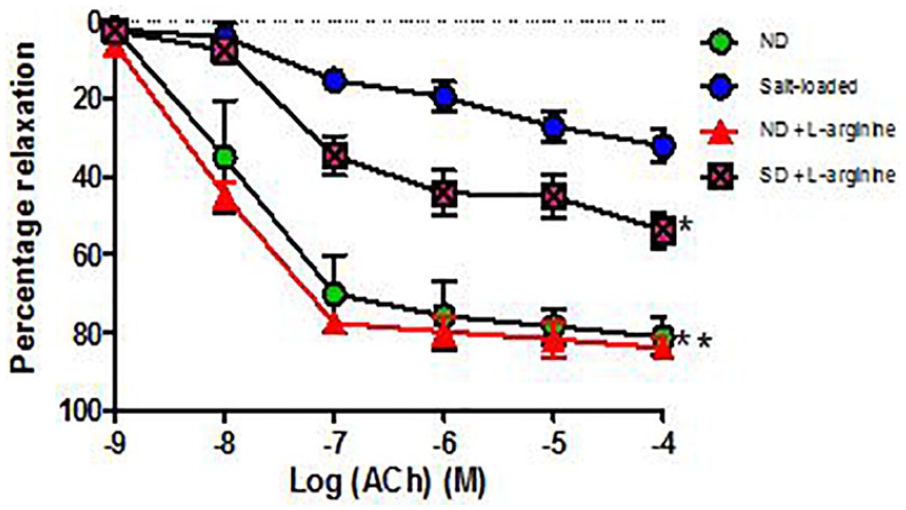
Percentage relaxation response of aortic rings to acetylcholine (ACh) in Groups 1 to 4 (6 rings). Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates

Percentage relaxation response of aortic rings to acetylcholine in Groups 3 to 6 (6 rings). Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates
Effect of salt-load and l -arginine supplementation on relaxation response to acetylcholine in the presence of L-NAME
Figures 3 and 4 show the relaxation responses to acetylcholine following incubation in L-nitro-arginine-methyl-ester (L-NAME). There was no significant change (P > .05) in the maximum relaxation response to acetylcholine in the groups. There was, however, a significant (P < .05) increase in −Log EC50 in the SD +

Relaxation response of abdominal aortic rings to acetylcholine (ACh) in the presence of L-NAME across Groups 1 to 4 (6 rings). Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates
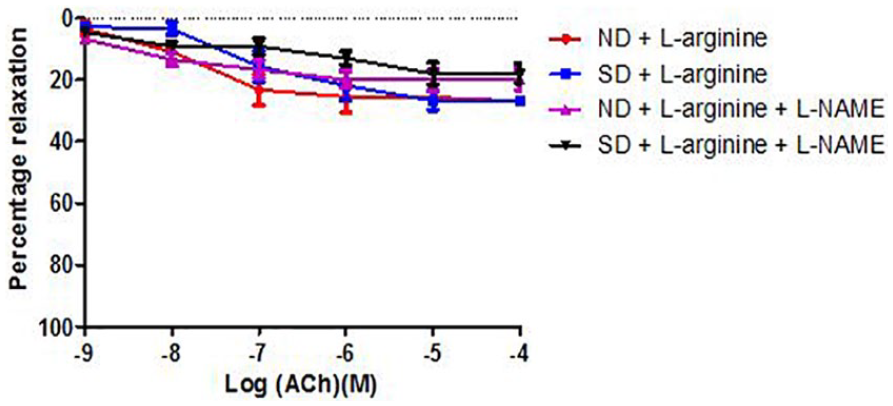
Relaxation response of abdominal aortic rings to acetylcholine (ACh) in the presence of L-NAME across Groups 3 to 6 (6 rings). Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates
Effect of salt-load and l -arginine supplementation on relaxation response to acetylcholine in the presence of methylene blue
Relaxation responses were not significantly different (P > .05) in the salt-loaded group (maximal response of 15.44% ± 2.50%) compared to the Normal Diet group (maximal response of 11.11% ± 1.10%). In the groups supplemented with oral

Relaxation response of abdominal aortic rings to ACh in the presence of Methylene Blue (MB) across groups 1 to 4 (6 rings). Relaxation responses are expressed as percentage of decrease in sub-maximal contraction elicited by NA (10-5M). Each point on the graph represents mean ± SEM.
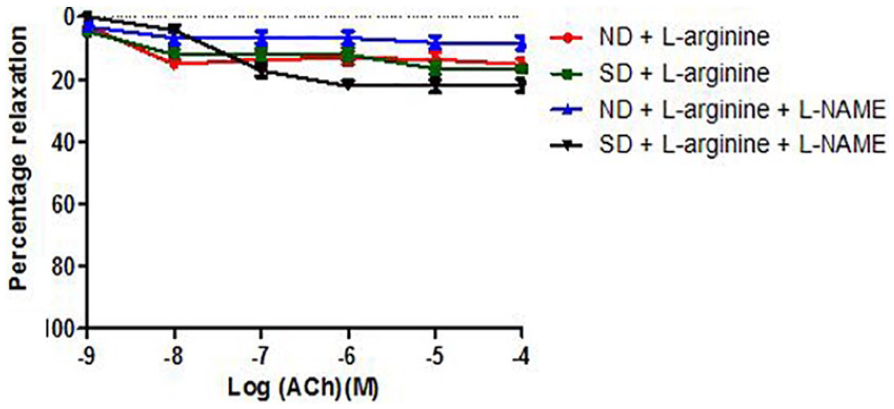
Relaxation response of abdominal aortic rings to ACh in the presence of Methylene Blue (MB) across groups 3 to 6 (6 rings). Relaxation responses are expressed as percentage of decrease in sub-maximal contraction elicited by NA (10-5M). Each point on the graph represents mean ± SEM of 5 rings from different animals. *p <0.05 compared with Control rats.
−Log EC50 and percent maximum relaxation response of abdominal aortic rings to acetylcholine with or without methylene blue (MB).
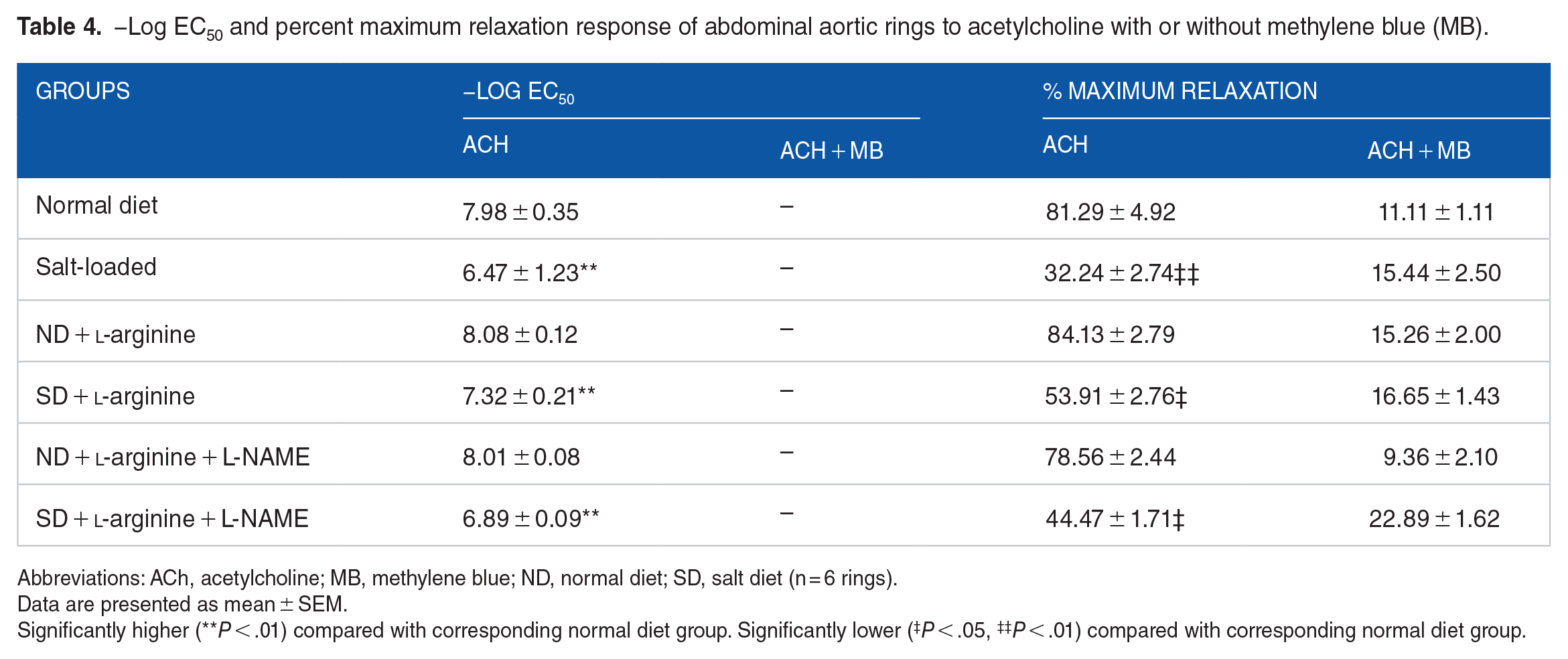
Abbreviations: ACh, acetylcholine; MB, methylene blue; ND, normal diet; SD, salt diet (n = 6 rings).
Data are presented as mean ± SEM.
Significantly higher (**P < .01) compared with corresponding normal diet group. Significantly lower (‡P < .05, ‡‡P < .01) compared with corresponding normal diet group.
Effect of salt-load and l -arginine supplementation on relaxation response to SNP
The maximal relaxation response to SNP (10−10-10−4) in NA-precontracted endothelium-denuded abdominal aortic rings was not significantly different (P > .05) in the aortic rings from salt-loaded group when compared with Normal Diet. Oral

Percentage relaxation response of aortic rings to sodium nitroprusside (SNP) in Groups 1 to 4 (6 rings). Each point on the graph represents mean ± SEM. ND indicates normal diet; SD, salt diet.
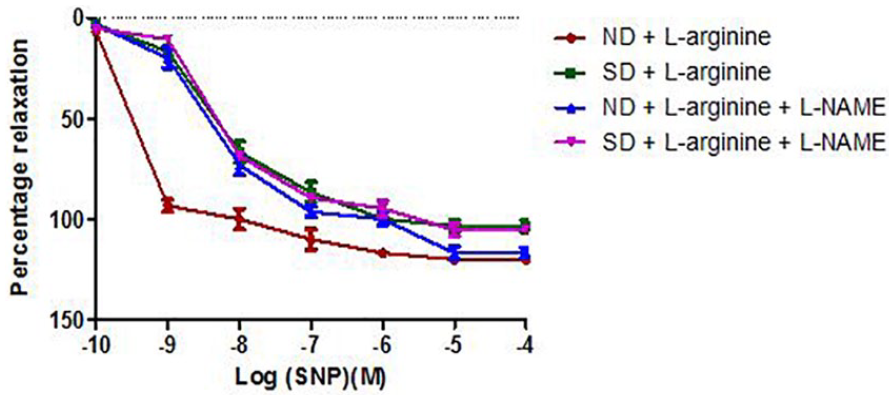
Relaxation response of abdominal aortic rings to sodium nitroprusside (SNP) in Groups 3 to 6 (6 rings). Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates
Effect of salt-load and l -arginine supplementation on relaxation response to SNP in the presence of L-NAME
There was no statistically significant difference (P > .05) in the maximal relaxation response to SNP in the presence of L-NAME across the groups. Oral

Relaxation response of abdominal aortic rings to sodium nitroprusside (SNP) in Groups 1 to 4 (6 rings) in the presence of L-NAME. Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates
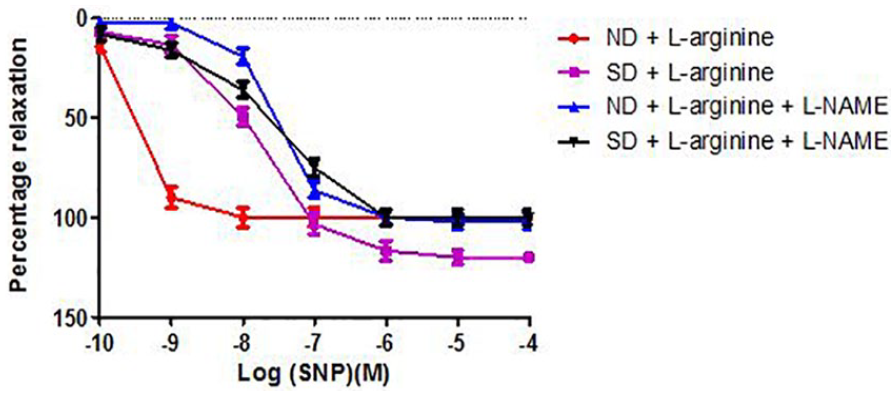
Relaxation response of abdominal aortic rings to sodium nitroprusside (SNP) in Groups 3 to 6 (6 rings) in the presence of L-NAME. Relaxation responses are expressed as percentage of decrease in submaximal contraction elicited by NA (10-5 M). Each point on the graph represents mean ± SEM. L-NAME indicates
−Log EC50 and percent maximum relaxation response of abdominal aortic rings to SNP with or without L-NAME.
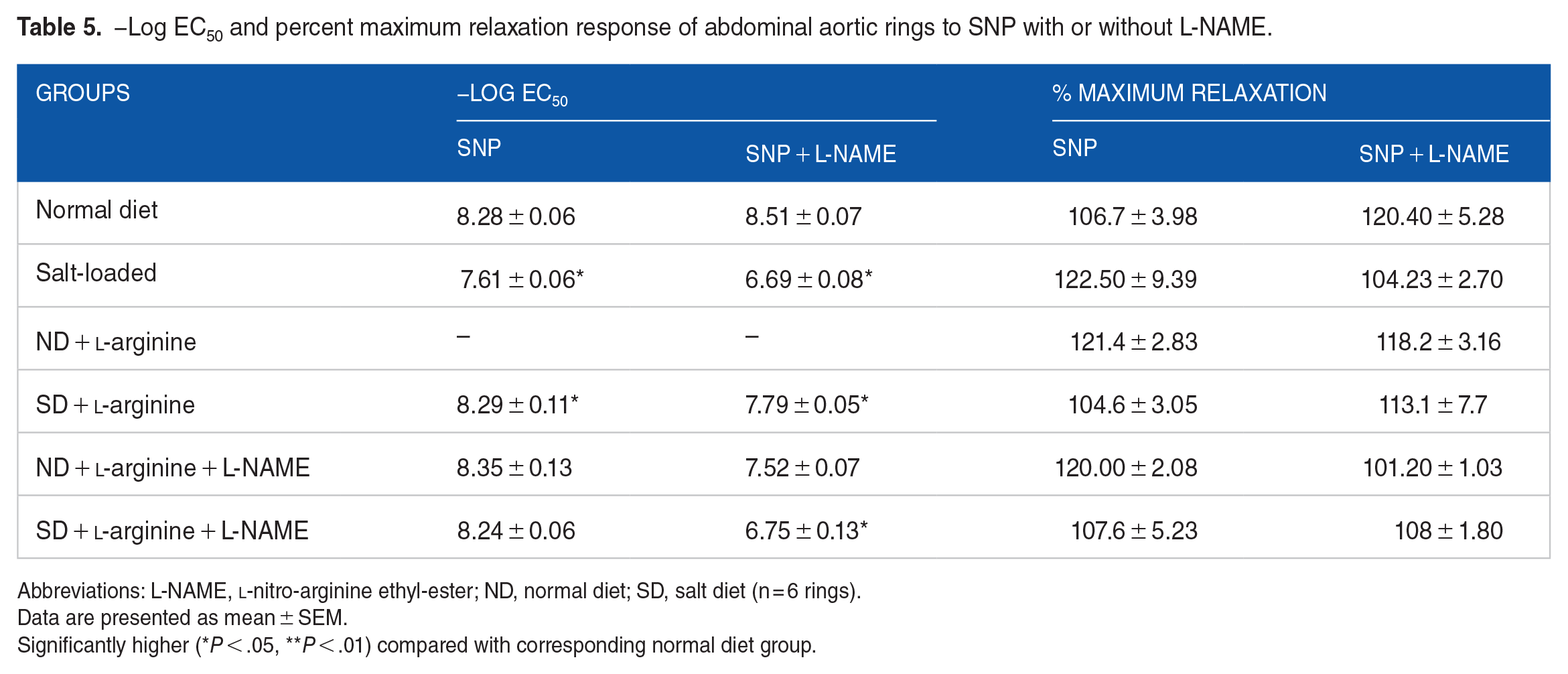
Abbreviations: L-NAME,
Data are presented as mean ± SEM.
Significantly higher (*P < .05, **P < .01) compared with corresponding normal diet group.
Effect of salt-load and l -arginine supplementation on relaxation response to SNP in the presence of methylene blue
There was no statistically significant difference (P > .05) in the maximal relaxation response to SNP in the presence of MB across the groups. Oral
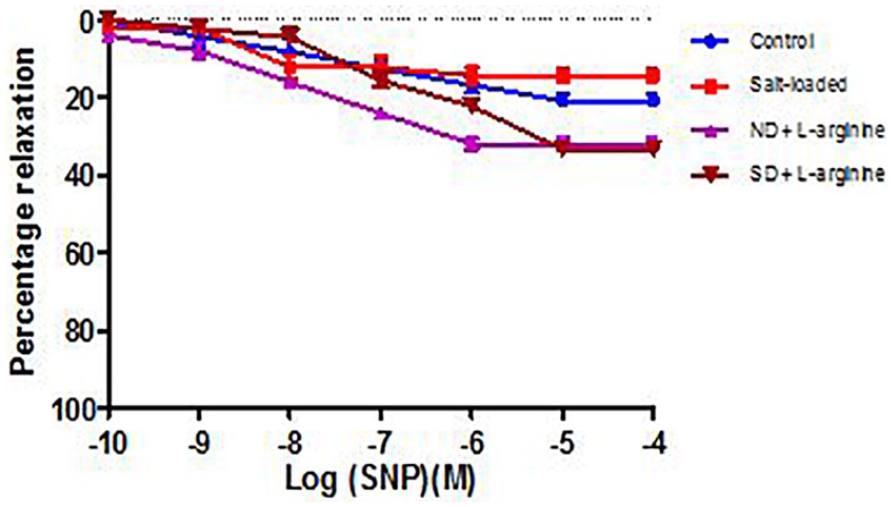
Relaxation response of abdominal aortic rings to SNP in the presence of Methylene Blue (MB) across all the groups (6 rings). Relaxation responses are expressed as percentage of decrease in sub-maximal contraction elicited by NA (10-5M). Each point on the graph represents mean ± SEM.
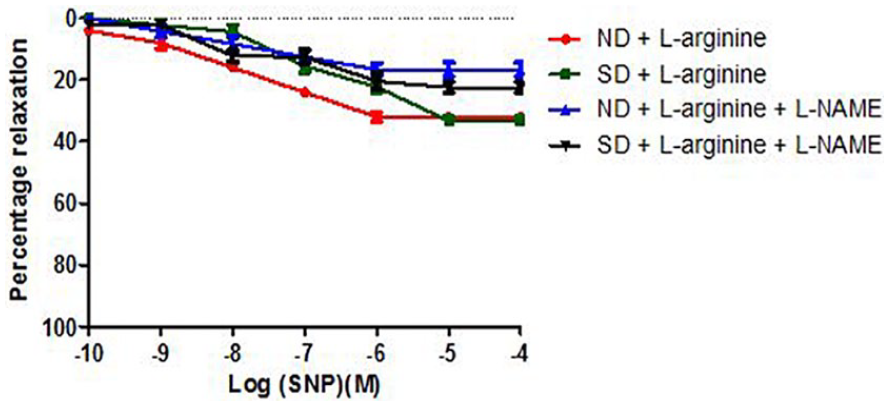
Relaxation response of abdominal aortic rings to SNP in the presence of Methylene Blue (MB) across groups 3 to 6 (6 rings). Relaxation responses are expressed as percentage of decrease in sub-maximal contraction elicited by NA (10-5M). Each point on the graph represents mean ± SEM.
−Log EC50 and percent maximum relaxation response of abdominal aortic rings to SNP with or without methylene blue (MB).

Abbreviations: SNP, sodium nitroprusside; MB, methylene blue; ND, normal diet; SD, salt diet (n = 6 rings).
Data are presented as mean ± SEM.
Significantly higher (*P < .05, **P < .01) compared with corresponding normal diet group.
Effects of salt-loading and oral l -arginine supplementation on nitric oxide level
Nitric oxide concentration appears to be lower in the salt-loaded group compared to normal diet but not significantly (P > .05). Nitric oxide level was significantly higher (P < .05) in the ND +

Nitric oxide level in Groups 1 to 6. Data are presented as mean ± SEM (n = 8). Significantly higher (*P < .05, ***P < .001) compared with salt-loaded group.
Effects of salt-load and l -arginine supplementation on eNOS gene expression
For all the above-mentioned effects to occur, there must have been changes in the expression of a lot of genes in the endothelium.13,18 Of all these genes, arguably, the most important one is the gene that codes for the eNOS enzyme responsible for conversion of
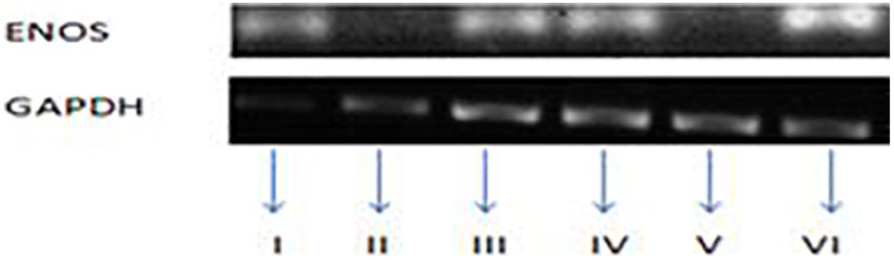
Expression of eNOS mRNA in the aorta from each group of rat as assayed by qualitative conventional PCR (6 rings). Group I = Control, Group II = Salt-loaded, Group III = Supplemented with
Adverse events
The 6 (n = 6 out of 8) animals reported in each group for vascular reactivity were the animals that completed all the treatments. Those that died were replaced and treated separately and accordingly. For the gene expression part, the remaining 2 (n = 2 out of 8) animals were used and 3 rings were cut out from each animal to make 6 rings per group. These rings were then used in the qualitative conventional PCR reactions. Likely influence of female sex hormones was removed by using male rats in the study.
Discussion
In this study, the relaxation response to acetylcholine in the salt-loaded rats was greatly reduced compared to the Normal Diet rats. The sensitivity of the rings to acetylcholine as measured by the −Log EC50 values was also observed to be significantly reduced in the salt-loaded rats. This corresponds with a reduced expression of the eNOS gene in the abdominal aorta of the rats. This implies that high-salt diet impairs vascular relaxation response to acetylcholine, which depends on the presence of an intact endothelium possibly by suppressing the expression of the eNOS gene. These vascular relaxation responses to acetylcholine occurred without significant interference with relaxation responses to SNP. Oloyo et al. made similar observations in the abdominal aortic rings from SD rats fed a high-salt diet.6,7 In line with the results of this study, in Dahl salt-sensitive hypertensive rats, a high-salt diet was reported to impair endothelium-dependent relaxations induced by a variety of vasodilators.19,20 This impairment could best be explained by reduced activity and expression of the eNOS mRNA which results in reduced conversion of the substrate,
Administration of exogenous
It is worthy of note that oral supplementation with
Conclusion
Results from this study suggest that oral supplementation of
Limitation
Only male rats were used in this study to rule out the possible influence of female hormones and thus the conclusion made may not be applicable to female rats with salt-induced hypertension. This is thus a subject of another study. Also, only eNOS gene expression was studied here, and many other genes may, however, be involved in the control.
Footnotes
Acknowledgements
The authors sincerely acknowledge the support of Emeritus Professor Olusoga Sofola and Professor A. S. Akanmu for allowing the use of their laboratories for this work.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AA conceived the idea, carried out the bench work, did the statistical analysis and drafting of the manuscript for publication.
AO helped to develop the original idea, participated in the bench work, provided the materials and equipment used for the work, participated in the drafting of the manuscript for publication.
CA helped to develop the original idea, read through the manuscript and supervised the research work.
SJ contributed to the development of the original idea, read through the manuscript and supervised the research work.
Disclosure
All authors have approved the final article.
