Abstract
Contrast-induced acute kidney injury (CI-AKI) is an important consideration in patients undergoing cardiac catheterization. There has been a continuous strive to decrease morbidity and improve procedural safety. This review will address the pathophysiology, predictors, and clinical management of CI-AKI with a concise overview of the pathophysiology and a suggested association with left atrial appendage closure. Minimizing contrast administration and intravenous fluid hydration are the cornerstones of an effective preventive strategy. A few adjunctive pharmacotherapies hold promise, but there are no consensus recommendations on prophylactic therapies.
Keywords
Introduction
Since the advent of transcatheter diagnostic and interventional procedures, there has been a continuous strive for reduced morbidity and improved procedural safety. This continues to be a high priority within the growing field of structural heart interventions. Unfortunately, most procedures require intravascular administration of contrast media, which is associated with contrast-induced acute kidney injury (CI-AKI) and a commensurate increase in morbidity. We will review the pathophysiology, predictors, and clinical management of CI-AKI, in addition to a brief overview of the pathophysiology and a proposed association with left atrial appendage closure (LAAC) and renal dysfunction.
Pathophysiology of CI-AKI
An association between radiocontrast agents and acute kidney injury has been well established. All formulations of contrast media have shown to be cytotoxic in vitro. Endothelial cells experience significant cell damage and/or apoptosis when exposed to contrast media 1 (Figure 1). It is believed that free-iodine is released from the contrast during the procedure and provides a direct cytotoxic effect on the surrounding endothelial cells. 2 , 3 In addition to damaging the surrounding endothelium, the contrast media have a cytotoxic effect on the tubules of the nephron. 4 Cell damage on both fronts leads to oxidative stress and formation of free radicals. The generation of free radicals and reactive oxygen species (ROS) consumes NO and consequently prevents the protective effect of NO as a vasodilator. 1 Tsarouhas et al 5 emphasized that oxidative stress plays a role in CI-AKI; contrast mobilizes the antioxidant defense mechanism. In their study, a transradial approach reduces this risk. The sustained vasoconstriction, which could last for hours, decreases the glomerular filtration rate (GFR), causes medullary hypoperfusion, and increases blood viscosity through the nephron. 2 Medullary hypoperfusion impedes oxygen delivery, resulting in ischemic injury to the tubules, which sustains the vicious cycle. In addition to its cytotoxic effects, the water-soluble contrast media can cause renal damage due to the difference in osmolarity relative to the surrounding tissue. 2 Contrast media solutions with higher osmolarity are associated with higher cytotoxicity. As the contrast media pass through the tubules, it is exposed to increasingly hyperosmolar environments, especially in the medulla. Subsequently, water is drawn out of the tubules into the medulla, leading to an increase in the viscosity of the tubular fluid. 2 This is especially problematic, as there is an increase in tubular pressure, while there is a decrease in the urine flow rate. With the increase in tubular pressure, interstitial pressure increases, further exacerbating the medullary hypoperfusion caused by the previously discussed mechanism. Decreased urine flow rate increases contrast media retention, thus increasing its cytotoxic impact. 2 Mamoulakis et al 6 evaluated the biomarkers that may reflect kidney damage in the blood and tissue samples of the animal model following administration of contrast. Iopromide is a molecule in the contrast media found to cause significant elevation serum creatinine (sCr) 68.2% within 48 hours, and serum symmetric-asymmetric dimethylarginine (SDMA-ADMA) levels. These markers were associated with an increase in renal-cell degeneration and apoptosis.

The proposed mechanism of contrast-media-mediated nephrotoxicity.
In summary, it is the interplay of cytotoxicity and viscosity that forms the pathophysiological basis of CI-AKI (Table 1). As will be discussed later, preprocedural hydration with normal saline (0.9% NaCl) 7 and intravenous (IV) hydration reduces the adverse effects of contrast by flushing of the renal tubules. 2
Literature review—pathophysiology of contrast-induced acute kidney injury.

Abbreviations: CHF, congestive heart failure; CI-AKI, contrast-induced acute kidney injury; CM, contrast media; STEMI, ST-elevation myocardial infarction.
Age-Related Changes in Renal Function
One of the most important risk factors for CI-AKI is age. 8 Changes in renal physiology over time lead to a reduction in renal function (Table 2), which may explain why age is a strong risk factor for CI-AKI.
Literature review—age-related changes in renal function.
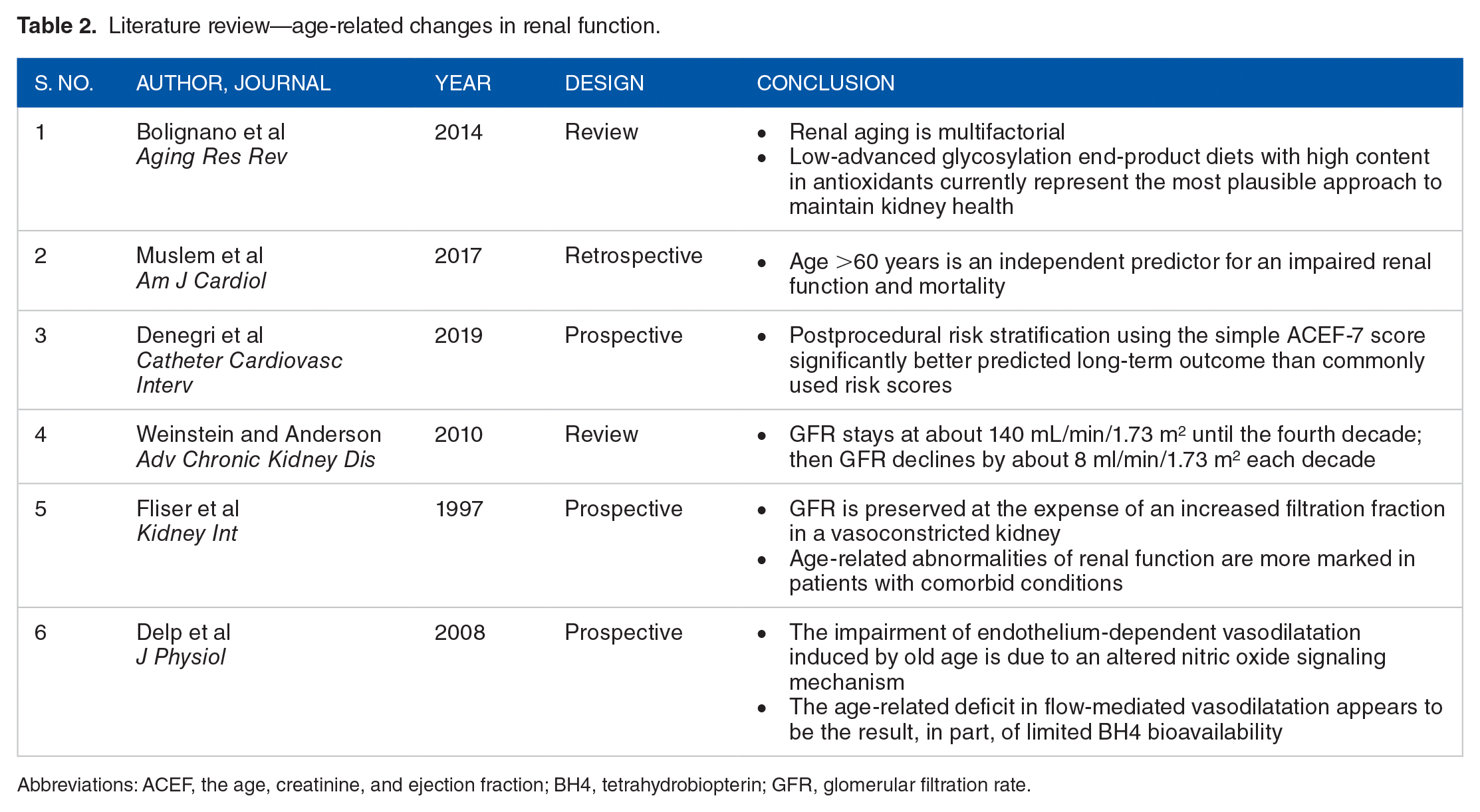
Abbreviations: ACEF, the age, creatinine, and ejection fraction; BH4, tetrahydrobiopterin; GFR, glomerular filtration rate.
Glomerular filtration rate is determined by renal blood flow and permeability of the glomerular filtration barrier. Increasing age is accompanied by a decrease in renal plasma flow due to an increase in vascular resistance of the efferent arterioles 9 (Figure 2). Consequently, the decrease in renal plasma flow causes an overall increase in the filtration fraction. Increased filtration fraction leads to hyperfiltration of the glomerular capillaries, which can damage the filtration barrier and lead to enhanced albuminuria. 10 , 11 Over time, these processes lead to irreversible thickening of the glomerular basement membrane and expansion of the mesangium. 12 A decline in nitric oxide—a vascular vasodilator that inhibits mesangial cell growth 13 —is also seen with advanced age due to oxidative stress and decreased L-arginine availability. 14

Changes in renal pathophysiology with age.
The magnitude of the decline in GFR varies across studies. In a systemic review, the yearly decline in estimated gomerular filtration rate (eGFR) ranged from 0.4 to 2.6 mL/min/1.73 m2. 9 However, most studies used a GFR estimate to examine the natural history of renal function. In a study by Fliser et al 15 using the gold standard method of inulin clearance, the GFR of a healthy group of volunteers with an average age of 68 ± 7 years was 85% lower than a similarly healthy group of volunteers with an average age of 26 ± 3.
The reduction in GFR with age and the pathophysiological basis for this reduction places it at an increased risk of AKI from other insults.
Definition, Clinical Implication, Controversies, and Risk Assessment
Contrast-induced acute kidney injury is defined as an increase in sCr by 0.3 mg/dL or an increase in creatinine to ⩾1.5 times baseline within 3 to 5 days following contrast exposure. 16 Using this definition of CI-AKI, a study of 33 249 patients with acute myocardial infarction undergoing cardiac catheterization demonstrated a dramatic decline in the incidence of CI-AKI from 24.6% in 2000 to 16.5% in 2008. 17 The National Cardiovascular Data Registry (NCDR) Cath-PCI registry data have provided an excellence data source for examining contemporary trends, predictors, and outcomes of CI-AKI in patients undergoing percutaneous coronary intervention (PCI). Data collected between 2009 and 2011 from 985 737 patients in the Cath-PCI registry revealed an overall CI-AKI incidence of 7.1%, with 0.3% requiring dialysis 18 Other studies of the NCDR Cath-PCI registry since have demonstrated similar rates of CI-AKI between 7% and 8%, 19 , 20 likely suggesting a leveling off in this decline in the incidence of CI-AKI.
Although the incidence of the need for dialysis remains low, the occurrence of CI-AKI is a significant sentinel event. It is associated with prolonged hospitalization and increased short- and long-term mortality. 21 Contrast-induced acute kidney injury risk of in-hospital mortality (9.7%; adjusted odds ratio [OR]: 7.8; 95% CI: 7.4-8.1, P < .001). 18 Contrast-induced acute kidney injury is also associated with an increase in postdischarge adverse events. A study linking the Cath-PCI registry to the Center for Medicare & Medicaid Services (CMS) billing data, found a higher postdischarge rate of death, myocardial infarction, and bleeding at 1 year, with the highest rates occurring within 30 days. 19 Patients with CI-AKI requiring dialysis have higher rates (34%) of in-hospital mortality (OR: 21.7; 95% CI: 19.6-24.1; P < .001) 18 and postdischarge adverse events 19 compared with subjects without CI-AKI.
Furthermore, CI-AKI is associated with accelerated progression of chronic kidney disease (CKD). In a retrospective cohort study of 11 249 patients undergoing coronary angiography, patients with CI-AKI were more likely to have sustained reduction in renal function at 3 months. In adjusted analysis, the odds of a decline in renal function were higher in those with moderate to severe CI-AKI (OR: 17.31, 95% confidence interval [CI]: 12.03-24.90) compared with milder AKI (OR: 4.74, 95% CI: 3.92-5.74). 22
It should be noted that although the entity of CI-AKI is relatively well accepted in the cardiology and nephrology community, there is skepticism regarding the true incidence of the disease. Rates of CI-AKI in patients undergoing contrast-enhanced computed tomography exam are lower (5%-6%). Wilhelm-Leen et al 23 found that patients to whom radiocontrast was and was not administered developed AKI at rates of 5.5% and 5.6%, respectively. Moreover, adverse outcomes in these patients is negligible (~0%) compared with the cardiac studies above. 24 A meta-analysis of studies comparing contrasted and noncontrasted radiology exams demonstrated no difference in the rates of CI-AKI. 25 There are multiple potential explanations for these discrepant findings. Patients undergoing cardiac catheterization likely have more comorbidities or present in the setting of acute coronary syndrome with tenuous hemodynamic status, all of which contribute to a higher risk of CI-AKI. Moreover, it has long been known that injection of contrast in the arterial bed or near the renal arteries is associated with a higher risk of CI-AKI compared with IV administration. 24 Atherosclerotic emboli could also partly explain these findings. Coronary angiography had traditionally been done via an access through the femoral arteries, which requires wire and catheter manipulation across the renal arteries. This could lead to embolization of plaque to the distal renal vascular bed and contribute to the onset of CI-AKI. This proposed mechanism is supported by a meta-analysis of studies comparing radial versus femoral artery access for coronary angiography that demonstrated a lower risk of CI-AKI in the radial access group. 26
As discussed below, therapeutic options for CI-AKI are limited. Therefore, it is imperative to identify risk factors and establish preventive strategies to minimize CI-AKI in all patient populations. The most critical risk factor for CI-AKI is preexisting renal impairment. 27 , 28 In patients undergoing cardiac catheterization, moderate to severe CKD (eGFR < 30), ST-elevation myocardial infarction, and cardiogenic shock are associated with more than quadrupling of risk. 18 Other predictors of CI-AKI include diabetes, heart failure, contrast volume, and anemia. 29 Mehran et al 30 and Tsai et al 18 developed two separate validated risk-prediction tools that incorporate the above risks in addition to other variables with modest discrimination. These tools are useful in clinical decision-making with patients and families, in addition to guide the clinician in determining preventive therapies.
Prevention and Management of CI-AKI
Minimizing contrast
As there is no treatment for CI-AKI, prevention is the optimal management strategy. Multiple studies have demonstrated a direct correlation between the volume of contrast administered and risk of CI-AKI. 30 Therefore, it is imperative that the volume of contrast administered is the lowest amount reasonable for a successful procedure (Figure 3). A contrast volume to creatinine clearance ratio of >2.44 is shown to be associated with CI-AKI. 31 Automated injection systems are available to minimize “excessive” contrast injection by the operator; however, these systems have not been shown to reduce the rate of CI-AKI. 32

Proposed algorithm for CI-AKI prevention in cardiac Cath patients.
High-osmolar agents had previously been shown to be associated with high risk of CI-AKI, and therefore, their use has been virtually eliminated for coronary angiography. 33 Iso-osmolar agents were shown to further reduce the rate of CI-AKI compared with low-osmolar agents in high-risk patients with underlying renal impairment undergoing coronary angiography. 34 However, these findings were not replicated in subsequent studies and meta-analyses comparing the two agents, 35 , 36 and consensus documents do not recommend the use of either agent. 37
IV fluid hydration
As mentioned previously, normal saline (0.9% NaCl) flushes the renal tubules and facilitates the transition of the water-soluble contrast through nephrons, thereby reducing its cytotoxic impact. Hydration is the only accepted practice for prevention and management of CI-AKI. In a study of 408 patients presenting with ST-elevation myocardial infarction (STEMI), patients randomized to routine hydration using IV normal saline (1 mL/kg/min) from the onset of the procedure till 24 hours postprocedure had a significant reduction in CI-AKI than the no hydration group (11% vs 21%, P = .016). 38 Although normal saline appears to be superior to half-normal hydration, 39 studies have failed to show superiority of sodium bicarbonate compared with normal saline hydration. 40 , 41
Although adequate hydration is an important preventive strategy, volume overload should be avoided, particularly in patients presenting with left ventricular dysfunction (i.e. preexisting heart failure or acute coronary syndrome). One hydration strategy features the use of an invasive hemodynamic measure as a guide for fluid administration. The POSEIDON (Prevention of Contrast Renal Injury with Different Hydration Strategies) trial randomized 396 patients with preexisting renal impairment (eGFR < 60) and an additional risk factor (diabetes, heart failure, hypertension, and age >75) to left ventricular end-diastolic pressure (LVEDP)-guided hydration strategy or standard fluid administration. Standard group received a bolus infusion of 3 mL/kg for 1 hour prior to the procedure, followed by 1.5 mL/kg/hr for 4 hours. The LVEDP-guided group received the same preprocedural hydration, but the periprocedural and postprocedural hydration were guided by LVEDP. There was a significant reduction in the rate of CI-AKI with the LVEDP-guided group relative to the standard group (relative risk [RR] = 0.41; 95% CI: 0.22-0.79; P = .005, which paralleled a reduction in the 6-month major adverse events rate—a composite of all-cause mortality, myocardial infarction, and hemodialysis (RR: 0.32; 95% CI: 0.13-0.79; P = .008; Figure 3). 42
Pharmacotherapies
Multiple adjunctive pharmacotherapies for CI-AKI prevention have been tested with mixed outcomes (Table 3). N-acetylcysteine (NAC) was hypothesized to reduce the risk of CI-AKI via ROS scavenging and is recommended by Kidney Disease Improving Global Outcomes (KDIGO) for contrast prophylaxis in high-risk patients. However, two of the largest studies to date, PRESERVE and ACT trials, have failed to demonstrate a reduction in adverse renal outcomes with oral NAC. 41 Routine use of NAC for CI-AKI prevention is class III in the ACC/AHA guidelines (Table 4). 43
Literature review—risk assessment, prevention, and management of CI-AKI.
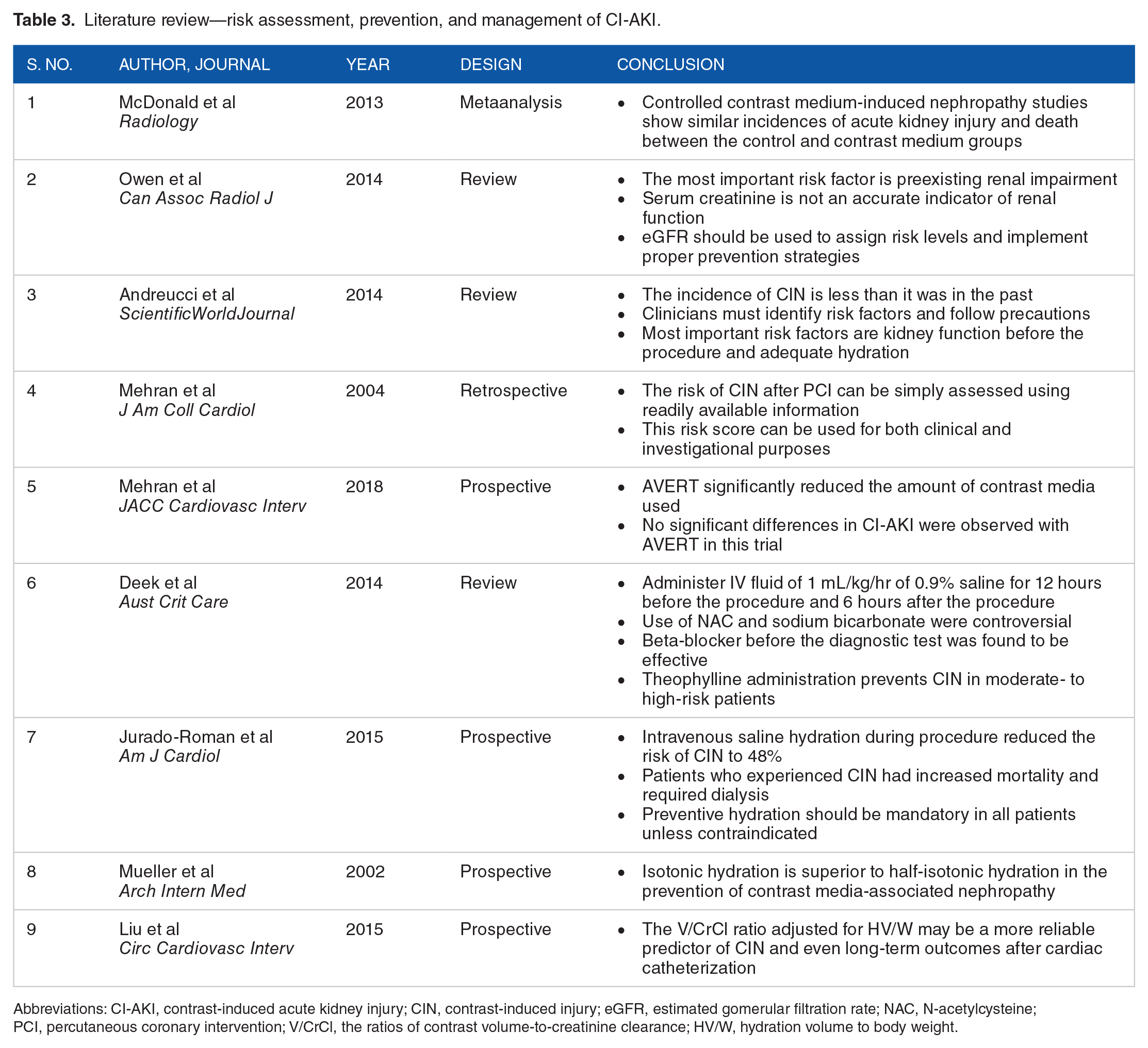
Abbreviations: CI-AKI, contrast-induced acute kidney injury; CIN, contrast-induced injury; eGFR, estimated gomerular filtration rate; NAC, N-acetylcysteine; PCI, percutaneous coronary intervention; V/CrCl, the ratios of contrast volume-to-creatinine clearance; HV/W, hydration volume to body weight.
Literature review—efficacy of percutaneous coronary intervention and associated kidney injury and mortality.

Abbreviations: ANP, atrial natriuretic peptide; BNP, B-type natriuretic peptide; CIN, contrast-induced injury; CKD, chronic kidney disease; CM, contrast media; rhBNP, recombinant human brain natriuretic peptide; STEMI-HF, ST-segment elevation myocardial infarction—heart failure.
Similarly, the anti-inflammatory effects of statin, in addition to other potential hypothesized reno-protective pathways, are thought to position it well as an adjunctive therapy in prevention of renal injury. Two randomized trials of rosuvastatin administered to patients undergoing coronary or peripheral angiography demonstrated significant reduction in the rates of CI-AKI. 44 , 45 Few meta-analyses of randomized trials confirmed these findings, and further demonstrated a significant reduction with high-dose versus low-dose statins. 46 , 47 It is important to note that most patients who are referred for coronary angiography, however, are likely on statin therapy or will be prescribed a statin regardless of the renal benefit. There are no guideline recommendations on the use of statins for CI-AKI prevention.
Studies of renin-angiotensin-aldosterone inhibitor (RASi) on CI-AKI have mixed findings, with harm, benefit, and neutral signals all demonstrated. The CAPTAIN trial of 208 patients with CKD undergoing cardiac catheterization randomized to hold (>24 hours) versus continue RASi failed to demonstrate a significant reduction in CI-AKI in the hold group (hazard ratio = 0.59; 95% CI: 0.30-1.19; P = .16), possibly due to the small sample size and beta error. 48 Current ACC/AHA guidelines do not make any recommendations on the preprocedural management of RASi, and KDIGO guidelines posit that there is insufficient evidence to recommend discontinuation of these agents.
Finally, initial concerns regarding withholding metformin stemmed from isolated case reports of death from CI-AKI and lactic acidosis in patients on metformin undergoing IV contrast studies. However, all reports of lactic acidosis have occurred in patients with severe underlying cardiac or renal disease. 49 There are no ACC/AHA or KDIGO guidelines regarding withholding metformin, and the 2018 American college of radiology guidelines recommends holding metformin only in patients with CI-AKI or GFR <30. 50
Proposed Association of LAAC and With Kidney Injury
The impact of new percutaneous catheter-based procedures on acute kidney injury is not well explored. In particular, the impact of LAAC on renal function is not well explored. Left atrial appendage closure is a transcatheter procedure for stroke risk reduction in patients with atrial fibrillation who are not candidates for long-term anticoagulation. Left atrial appendage closure is thought to reduce risk of stroke by excluding the appendage—where thrombus formation can occur in atrial fibrillation—from the remaining cardiac chambers; however, it also uncouples the appendage from hemodynamic changes in the left atrium. Two important regulatory peptides, atrial natriuretic peptide (ANP) and to a lesser extent brain natriuretic peptide (BNP), are stored and released from the left atrial appendage in response to stretch mechanoreceptors. Levels of both these peptides have been shown to decrease acutely after LAAC. 51 Atrial natriuretic peptide and BNP are key regulatory peptides that cause vasodilation of the afferent arterioles, decrease renal vascular resistance, increase renal blood flow, and consequently increase natriuresis and diuresis. 52 , 53 These down-stream actions of the peptides may have an important reno-protective quality.
In a study of 106 patients with mild underlying CKD undergoing elective coronary angiography or PCI, subjects were randomized to a recombinant form of BNP versus placebo. There was a significant reduction in the primary outcome of CI-AKI (6.6% vs 16.5%, P = .025). Patients with CI-AKI in the BNP arm had less deterioration and faster recovery of eGFR. In multivariable analysis, BNP administration was protective and contrast volume >100 mL was associated with CI-AKI. 54 A study of STEMI patients with heart failure and mild CKD also demonstrated a similar reduction in the rate of contrast-induced injury (CIN) in patients randomized to recombinant BNP infusion (12.28 vs 28.81%, P < .05). 55
In a randomized controlled trial, Morikawa et al 56 randomized 254 patients undergoing coronary angiography to infusion of ANP or lactated ringers. There was a significant reduction in the rate of CIN in the ANP group compared with the control group (3.2% vs 11.7%; P = .015), with a persistently lower creatinine in the ANP group at 1-month follow-up. A randomized study of patients after coronary artery bypass graft demonstrated a significant reduction in creatinine increase rate, in addition to a significant reduction in the need for dialysis and cardiac events in the ANP arm compared to placebo. 57
The rate of CI-AKI in a small study of 355 patients undergoing LAAC was 9%, which is a slightly higher rate compared with patients undergoing cardiac catheterization (~7%). 58 This is despite the IV hydration in almost all patients undergoing LAAC, with a goal of mean left atrial pressure >12 mmHg. In this population, CI-AKI was associated with higher in-hospital bleeding events, out-of-hospital bleeding events, systemic embolic events, and mortality. The higher rate of CI-AKI can be attributed to a patient population with more risk factors. In our single-center experience of 106 patients undergoing LAAC, we noted a significant decline in eGFR at 6 month follow-up (71.9% vs 62.8%; P < .001). Collectively, these studies add traction to a possible reno-protective role for ANP and BNP by reducing the risk of CI-AKI in patients undergoing cardiac procedures. Hence, in addition to the intra-cardiac iodinated contrast administration, LAAC may increase the risk of CI-AKI by reducing circulating levels of ANP and BNP. Additional larger studies are needed to assess the short- and long-term impacts of LAAC on renal function.
Conclusions
Contrast-induced acute kidney injury continues to be a major comorbidity in patients undergoing cardiac catheterization. It is associated with major adverse clinical outcomes and death. Minimizing contrast administration and IV fluid hydration are the cornerstones of an effective preventive strategy. Although a few adjunctive pharmacotherapies hold promise, there are no consensus recommendations on prophylactic therapies. There is a theoretical concern for an increased risk of renal injury with LAAC that is mediated by a reduction in natriuretic peptides. Further studies are needed to investigate this proposed link.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author’s note
Priya Bansal, Haider Al Taii, Houman Khalili, Brijeshwar Maini are also affiliated to Department of Cardiovascular Diseases, Florida Atlantic University, Boca Raton, FL 3348.
Author Contributions
All authors contributed extensively to the work presented in this literature review.
