Abstract
Background:
Complex Regional Pain Syndrome type 1 (CRPS-1) is severely debilitating and painful disease that is difficult to treat.
Objective:
The objective was to evaluate the long-term residual disability of patients with CRPS-1 following parenteral neridronate treatment.
Design:
This is real-life retrospective observational study.
Methods:
Patients affected by CRPS-1 of the upper limb were treated with neridronate infusions (400 mg over 10 days) between February 2017 and December 2021 for whom clinical and demographic information was collected. From November 2022, patients treated ⩾1 year previously were recalled for clinical evaluation. A dedicated instrument (DASH questionnaire, Disabilities of the Arm, Shoulder and Hand) was employed to assess residual disability. Multivariate logistic regression analysis was used to investigate predictors of disability.
Results:
Forty-nine patients aged 61.1 ± 11.5 years and 73.5% female with CRPS-1 of the upper limb were included. Disease duration before treatment was 9.9 ± 8.0 weeks, and the mean length of follow-up was 4 years (47.7 ± 22.0 months). The disease had fully resolved in 46 patients (93.9%) for whom the diagnostic criteria were no longer recognized. According to the DASH score, 38 patients (77.6%) resulted free of functional limitations, whereas 11 patients (22.4%) were still suffering from disability. The DASH score was positively correlated with residual visual analogue scale (VAS; Spearman’s Rho = 0.61; P < .001). Predictors of residual disability were younger age (odds ratio [OR]: 0.77, 95% CI: 0.63-0.93; P = .012) and delay between disease onset and treatment (OR: 1.45, 95% CI: 1.13-1.96; P = .004).
Conclusions:
In this real-life study, neridronate parenteral treatment provided a full recovery of CRPS-1 in over 3 quarters of patients, provided they are treated early.
Introduction
Complex Regional Pain Syndrome (CRPS) is a severely disabling painful disease that, together with pain, an array of clinical features, including swelling, vasomotor instability, abnormal sensory findings and motor dysfunction represent the hallmark of the disease.1-3 As currently conceptualized, CRPS is distinguished into type 1 and type 2 according to the absence/presence, respectively, of clinical and/or electromyographic signs of peripheral nerve injury. Despite this clinical subdivision, CRPS-2 (previously called causalgia) shares with type 1 the same clinical features and the diagnostic criteria are identical across both subtypes. 4 Usually, inflammatory signs prevail soon after onset, whereas in the long-term, these features tend to subside and pain and motor functional limitations (stiffness, weakness and reduced range of motion) are the most prevalent symptoms, able to permanently affect patients quality of life.5,6 The actual prevalence of such outcomes is a debated issue with contradictory findings in the literature. As reviewed by Johnson et al, 6 22 long-term studies report a percentage as high as 51% to 89% of patients complaining of losses in motor function at 12 months and beyond from the onset of symptoms. Recent studies using more stringent diagnostic criteria (Budapest criteria) 7 seem to report long-term outcomes in higher proportions of patients than previously described, with motor dysfunction heavily affecting functional and occupational recovery.8,9 Still debated is the role of some prognostic factors reported to be associated with poor outcomes (more intense pain at onset, age, disease duration, fracture as inciting event, etc), with weak and inconsistent predictive features. 10 Accounting for the results of these studies, pain reduction and disability prevention must be the aims of the treatment.
The treatment of CRPS is another challenging issue. In contrast to the majority of proposed treatments, only bisphosphonates (BPs) have gained credibility in the last 3 decades through the results of 6 randomized controlled studies (RCT)11-16 and 3 meta-analyses.17-19 These results have been achieved by employing different BPs in many different regimens, mainly when the disease was treated in the early stage. In recent years, more convincing evidence has been obtained with parenteral neridronate.15,16,18
A long-term extension study recently published demonstrated that the benefit in pain is maintained and further improves over 12 months, confirming the results achieved a few weeks after parenteral neridronate administration. 20 As well as pain, all clinical features associated with the early stage of CRPS (allodynia, hyperpathia, oedema and pain at passive motion) improve along with pain. Nevertheless, the performance of neridronate in avoiding long-term disability remains an unexplored issue.
The aim of this real-life study (different from an RCT approach) was to evaluate whether neridronate is able to prevent long-term outcomes of disease. The persistence of motor dysfunction after a prolonged follow-up to consider this outcome as permanent was carefully assessed. At the same time, the possible occurrence of adverse events reported to be associated with the long-term assumption of BPs was investigated.
Materials and Methods
Study Design
We carried out an observational, retrospective clinical evaluation of patients with a diagnosis of CRPS-1 for more than 12 months referred to our unit for treatment with intravenous neridronate infusions to evaluate the residual motor dysfunction and other clinical outcomes and their predictors.
We followed the STROBE Guidelines when preparing this article (see Supplementary Material S1).
Patient Population
This case series study includes data collected from a population of patients with a new diagnosis of CRPS-1 who were consecutively attending a tertiary-care-level centre devoted to bone and joint diseases, between February 2017 and December 2021 in Italy. The general practitioners or community consultants referred the participants to the rheumatology and orthopaedic services and the emergency department of our hospital. Matching the diagnostic code with the hospital database that collects administrative information resulted in the retrieval of 262 patients. When a CRPS-1 diagnosis was made and patients were referred to our unit, participants were asked to complete a predefined questionnaire by checking boxes (Supplementary Material S2). The questionnaire was completed in a few minutes, and the results were discussed afterwards with a consultant, in order to avoid incompleteness or misinterpretation. Information was gathered on demographics (age, sex, region of origin); clinical variables (time of symptoms onset, inciting injury, disease localization, previous treatments), pain measured by visual analogue scale (VAS, 0-100 mm, ranging from no pain [0 mm] to maximal pain [100 mm]) and the Italian version of the McGill Pain Questionnaire. 21 No patients had previously taken CRPS-1 treatments, except opioid analgesics, non-opioid analgesics and non–steroidal anti-inflammatory drugs (NSAIDs). Complete data were available for all patients included in this study. All patients were treated with 100 mg neridronate every third day for 4 infusions each diluted in a 500-mL saline isotonic solution and infused in the morning over 2 hours according to the provision of the Italian National Healthcare System. 22 All patients were referred to our hospital’s physiatry service after BP infusions to receive physiotherapy treatment to improve the functional restoration of the upper limb. In cases of pain, patients were instructed to take pain reliever such as opioid, non-opioid analgesics and NSAIDs. None of patients were treated with anticonvulsants, antidepressants drugs, vitamin C and other non-drug therapies.
All medical records of patients were reviewed by 2 of the authors (FO and GC), who had not been involved in the clinical management of the patients, and data were extracted following a predefined data extraction form. Only patients with upper arm disease were selected (128 patients; 48.8% of the total population).
In this study patients were included if (1) diagnosed with CRPS-1 based on the 2013 International Association for the Study of Pain (IASP) criteria for research purpose (4 symptoms + 2 or more signs); 23 (2) older than 18 years; (3); able to read, write and understand Italian; (4) with negative history and no clinical and/or electromyography evidence of nerve damage possibly resulting in CRPS-2 or referring previous surgical procedures or other hand diseases (inflammatory arthritis, osteoarthritis, Dupuytren disease, etc) able to induce a permanent disability; (5) no other treatments for CRPS-1 performed after neridronate infusions, other than anti-inflammatory, or analgesic drugs. No patients involved in previous RCT studies were included in this sample. In November 2022, a telephone survey was conducted, calling all selected patients treated more than a year before and offering a further clinical evaluation free of charge. Patients who agreed to participate in this follow-up study were recruited. The use of clinical data in anonymous form was requested and obtained from each patient both at the time of the first assessment and at the clinical evaluation performed for this study. All patients provided written informed consent.
Data Collection and Outcome Measures
Medical records of patients who agreed to participate in the study were assessed after an appointment arranged in the hospital and included: (1) symptoms and signs of the 2013 IASP diagnostic criteria; 23 (2) pain measured by visual analogue scale (VAS, 0-100 mm, ranging from no pain [0 mm] to maximal pain [100 mm]); (3) the Italian version of the McGill Pain Questionnaire; 21 (4) current use of pain control drugs; (5) upper arm disability assessed by the Italian version DASH questionnaire (Disabilities of the Arm, Shoulder and Hand). 24 The DASH is a questionnaire (30 items, scored 1-5) designed to measure physical function, symptoms and level of disability in people with musculoskeletal disorders of the upper limb. 25 The assigned value for each patient is transformed to get a score out of 100, where 0 means no disability and 100 means the highest level of disability. According to the literature data, the DASH questionnaire describes the absence of functional limitations for a score ⩽15 (no problem with work), some functional disabilities for a score of 16 to 40 (problem, but working) and severe functional disabilities for a score >40 (unable to work); 26 (6) the compliance with the prescribed physiotherapy treatment.
Statistical Analysis
Descriptive results of demographic and clinical data were reported as frequency (percentage) for categorical variables and median values (interquartile range, IQR) or mean ± standard deviation for continuous variables, according to distribution. Normal distribution of data was checked with the Kolmogorov-Smirnov test. The bivariate analyses were carried out using the Student’s t-test for continuous variables normally distributed, and the Mann-Whitney test was performed when non-normally distributed variables were analysed. The Fisher’s exact test was applied to analyse categorical variables. The Spearman’s rank test was used to measure the correlation between 2 sets of data.
Multivariate statistical analysis was performed to evaluate predictors of disability (DASH score >15) using logistic regression model, adjusted for continuous (age, VAS score, time between symptoms onset and treatment, follow-up time) and categorical variables (sex, adherence to physiotherapy, fracture as inciting event). Results are presented as odd ratios (ORs) and 95% confidence intervals (CIs). A 2-tailed P-value < .05 was considered statistically significant. Statistical analysis was conducted using IBM SPSS software (v. 28.0.1.1, SPSS, Inc, Chicago, IL).
Results
Patient Disposition and Baseline Characteristics
The flow chart illustrating the disposition of patients is depicted in Figure 1, and the demographic and clinical variables at the onset of 49 patients representing the final study sample we recruited are reported in Table 1. The mean age at diagnosis was 61.1 ± 11.5 with a greater number of females (36; 73.5%). The dominant hand was affected in 32 patients (65.3%). The time elapsed between symptoms onset and the start of treatment was 9.9 ± 8.0 weeks. The most common inciting event was a fracture reported by 42 patients (85.7%): in 3 cases, a trauma without fracture was recognized as the initiating event; 3 patients developed a CRPS-1 following surgery (2 after carpal tunnel release and 1 patient after trapeziectomy for osteoarthritis) and 1 patient developed CRPS-1 after electrocution. When evaluated before treatment, patients exhibited sensory signs in 45 cases (91.8%); vasomotor signs in 39 (79.6%); sudomotor/oedema signs in 43 (87.7%) and motor/trophic signs in 49 cases (100%).

Patient disposition.
Demographic and clinical characteristics of 49 patients with upper limb CRPS-1 treated with neridronate at the onset of disease.

CRPS-1: complex regional pain syndrome type 1; SD: standard deviation; VAS: visual analogue scale.
When patients have undergone clinical evaluation for this study, the mean length of follow-up was 47.7 ± 22.0 months.
Change in Visual Analogue Scale Score and McGill Pain Questionnaire Items Following Neridronate Treatment
Thirty patients (61.2%) reported full adherence to the prescribed physiotherapy programme. Both instruments employed to assess residual pain (VAS and McGill Pain questionnaire) showed highly significant decreases when compared with values observed before treatment (Table 2).
Change in VAS score and McGill Pain Questionnaire (MPQ) items in 49 patients with upper limb CRPS-1 before neridronate treatment and at the end of the study (mean length of follow-up 47.7 ± 22.0 months).
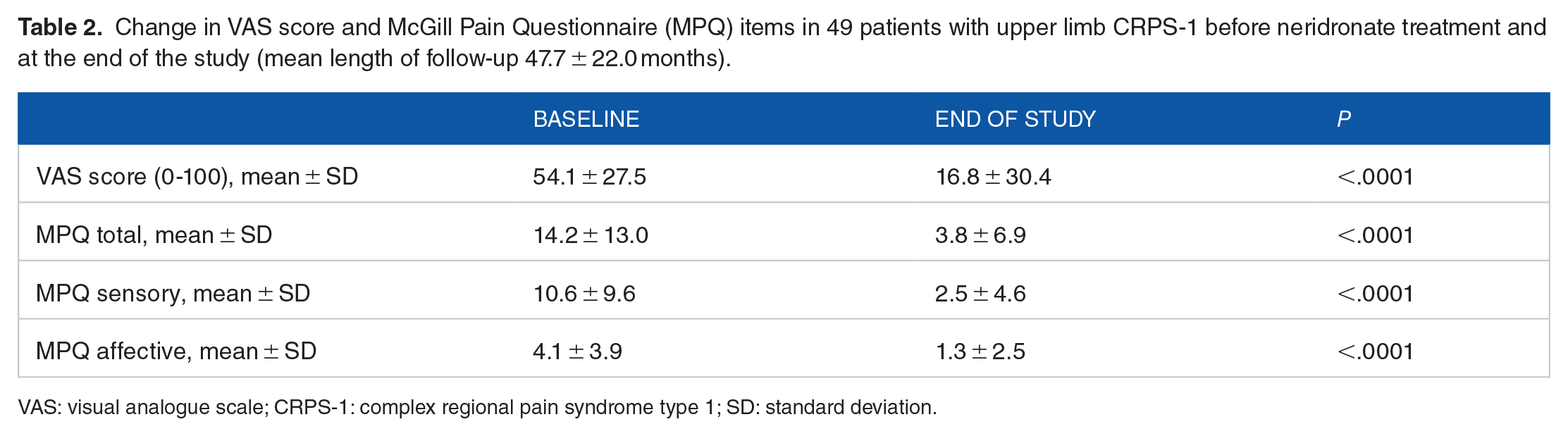
VAS: visual analogue scale; CRPS-1: complex regional pain syndrome type 1; SD: standard deviation.
Residual Symptoms and Signs of the Disease and Functional Outcome
Sensory symptoms were reported by 16 patients (32.6%; P < .001 vs prevalence at the onset), vasomotor symptoms by 7 patients (14.3%; P < .001), sudomotor/oedema by 4 patients (8.2%; P < .001) and motor/trophic symptoms by 14 patients (28.6%; P < .001). On clinical examination, all signs of disease were detectable with a significantly lower prevalence for all signs when compared with the clinical evaluation before treatment. Sensory signs were still recognized in 4 patients (P < .001); vasomotor signs in 2 patients (P < .001); sudomotor/oedema in 1 patient (P < .001) and motor/trophic signs in 11 patients (P < .001). Only 2 patients could still be diagnosed as suffering from CRPS according to the IASP Criteria (clinical purpose: 3 symptoms + 2 signs), nobody according to IASP criteria for research purpose. According to the DASH score, 38 patients (77.6%) resulted free of functional limitations (score ⩽ 15); 2 (4.1%) patients showed some functional disabilities (score 16-40) and 9 patients (18.4%) were still suffering from important functional limitations. Three patients sometimes took drugs to control pain (2 acetaminophen and 1 tramadol).
Stratification of Patients Based on Residual Functional Limitation According to Disabilities of the Arm, Shoulder and Hand Score
The comparisons between patients showing full functional restoration and patients still complaining of functional impairments are reported in Table 3. Age at onset (younger the disabled patients; P = .02); delay between symptoms onset and treatment (longer in disabled patients; P < .001) and greater residual pain in disabled patients (VAS score; P < .001) showed a significant difference between groups. Sex, VAS score and McGill pain questionnaire assessed before treatment, follow-up length, fracture as inciting event, and full adherence to physiotherapy showed no difference between groups. In the same way, prevalence at the onset of different signs of IASP diagnostic criteria showed no difference between groups (data not shown). By comparing the score values of some items of DASH assessing residual pain features possibly evocative for CRPS-related pain (spontaneous pain, numbness), patients with persistent functional limitations showed item values significantly higher (spontaneous pain: P < .001; numbness: P = .02). To further explore the clinical features of patients with residual functional limitations, we investigated the relationships between the DASH score and other variables. A significant correlation was found between the DASH score value and the residual VAS value (Spearman’s Rho = 0.61; P < .001). Conversely, no correlation was observed between the residual VAS values and follow-up length (Spearman’s Rho = −0.09; P = .53).
Comparison by DASH score result on residual functional limitation in a sample of 49 patients with upper limb CRPS-1 (⩽15: no functional limitations, >15 functional limitations).

Student’s test for continuous variables normally distributed and Fisher’s exact test for binary ones were used. The Mann-Whitney test was performed to analyse non-normally distributed variables.
DASH: Disabilities of the Arm, Shoulder, and Hand; CRPS-1: complex regional pain syndrome type 1; SD: standard deviation; IQR: interquartile range; VAS: visual analogue scale.
Predictors of Residual Disability by Multivariate Logistic Regression
Predictors of residual disability investigated by multivariate analysis showed consistent results with the data reported above (Table 4). Only age and the delay between disease onset and treatment acted as predictive variables for residual disability (DASH score > 15). By considering VAS score at onset as dichotomous instead of a continuous variable and employing a value greater than 75 mm (the highest quartile) to identify patients with higher pain intensity proposed as a negative prognostic factor, 27 the same results were observed (data not shown).
Predictors of residual functional limitations in a sample of 49 patients with upper limb CRPS-1 treated with neridronate.

CRPS-1: complex regional pain syndrome type 1; CI: confidence interval; VAS: visual analogue scale.
Multiple logistic regression model.
Safety
During the entire follow-up period, no patients complained of osteonecrosis of the jaw or other serious dental problems. Likewise, no atypical fractures or other adverse events possibly related to neridronate administration were reported.
Discussion
The results of this observational, real-life study provide evidence that ⩾75% of patients with CRPS-1 of the upper limb treated with neridronate administered intravenously experienced a satisfactory recovery of loss of function together with a significant improvement of all other symptoms of the disease. This result can be defined as an actual disease remission. These results confirm and strengthen what was observed in previous studies where a significant and clinically relevant benefit was achieved 40 days after starting treatment, 15 maintained and further improved over 1 year in patients with acute CRPS-1. 20 Not even the most optimistic reports28,29 showed long-term proportions of clinical remission as we observed after neridronate treatment.
Besides clinical features that characterize the disease in the early stage, pain and motor dysfunction are the most frequent long-term lasting symptoms of CRPS-1.5,6 The loss of limb function (reduction of range of motion, stiffness and weakness) with its effect on work status, activities of daily living, and health care costs, 30 is the most disabling marker of poor disease outcome.
The natural course of CRPS-1 with its ongoing clinical manifestations represents a controversial issue. Two comprehensive systematic reviews explored CRPS-1 symptoms recovery.5,6 As reported, several possible confounders do not allow definitive conclusions about an accurate outcome measure. The study design, the diagnostic criteria, the representativeness of the sample and the outcome measurements all affect the quality and relevance of specific studies. A further possible source of bias in studies on CRPS-1 outcome included different treatments that patients have followed that modified, hopefully, the course of the disease.
Overall, the clinical manifestations usually detectable at the onset of the disease (oedema, sensory disturbances, sudomotor and vasomotor changes) seem to improve in the first 6 to 12 months, 5 whereas the most relevant long-term symptoms (pain and motor dysfunction) can be prolonged to more than 1 year in 51% to 89% of patients. 6 An impaired limb function was observed to be much more frequent and disabling in recent studies than previously deemed, probably as a result of the use of stricter diagnostic criteria allowing a more accurate CRPS-1 diagnosis.6,31
In designing this study, we have chosen to employ restrictive selection criteria to obtain a sample as homogeneous as possible, even though this strategy resulted in a significant reduction in sample size. First, we included only patients diagnosed according to 2013 IASP criteria (Budapest criteria for research purpose), 23 recognized as the gold standard to ensure the highest specificity. 32 Second, only patients with upper limb disease were recruited because the ongoing impact of CRPS on manual dexterity and hand function activities of daily living and work status is more disabling than lower limb disease. To evaluate the persistence of disability, the DASH questionnaire has been employed as a widely used and validated tool to measure the physical function and symptoms in people with upper limb disorders. 25 Previous studies on CRPS-1 outcomes used this tool to quantify the residual dysfunction in these patients.9,30,33-35 Moreover, all patients have received the same treatment (4 intravenous infusions of 100 mg neridronate). Since 2014, this dosing schedule is registered and marketed in Italy for the treatment of CRPS. 22 The significant pain reduction evaluated with both VAS and McGill Pain questionnaire we observed in this study was consistent with results observed after 1 year. 20
In contrast to results observed in a recent study 36 in which male patients seem to have a better response after 12 months treatment of neridronate, our results do not support an influence of sex on residual disability. On the contrary, the influence of sex as a prognostic factor in CRPS is debated in the literature. 10
Looking at the predictors of a persistent loss of function, we found the time elapsed between the disease onset and the start of treatment significantly influences the odds of a full recovery. This result supports a widely shared expert opinion, ie, the shorter the disease duration, the better the treatment outcome, regardless of the therapeutic choice 37 and is consistent with the results of some studies that report an association between a poor outcome and the delay to receive treatment.38,39 The same variable also acts as a predictor of the short-term therapeutic response to BP treatment in patients with acute disease. 40 Speculatively, it might be assumed that a faster and more effective treatment may prevent permanent functional damage in the long term.
We found a strict relationship between the level of persistent disability and the intensity measure of residual pain. This association could be taken as a marker of chronic disease, being the 2 features with the more frequent long-term disease outcomes, thus identifying these patients as not responders. This hypothesis could be supported by some features of the painful symptoms investigated by specific items of DASH and is most likely related to CRPS-related pain (eg, spontaneous pain, numbness) and reported in a significantly greater percentage of patients showing a persistent loss of function. The residual pain is likely due to a disease that has become chronic.
As the disease progresses, there are morphologic changes that occur in the central nervous system. Thanks to recent imaging techniques (functional magnetic resonance imaging [MRI]), changes in the somatosensory cortex of long-lasting CRPS patients have been observed. 41 Therefore, in accordance with the disease duration, the therapeutic approach should be different. However, the residual pain could be at least partly the consequence of permanent anatomical damage due to a more intense and prolonged inflammatory process during the early stage of the disease. Muscle contracture, thickening and fibrosis of palmar fasciae, tendon sheaths, and joint capsules may be variables attributed to both loss of function and pain after functional activity.
Younger age at onset showed a significant predictive value on permanent functional impairment, thus confirming the results of studies reporting an unfavourable disease course in these patients and a negative prognostic value of the younger age at onset.38,42
A higher VAS value at onset was not predictive of a poorer prognosis and a permanent disability unlike reported elsewhere. 27 In the same way, full adherence to the prescribed physiotherapy programme does not modify the odds of suffering from a permanent disability. The role of physiotherapy in preventing long lasting disability is still debated. Some studies reported a significant improvement, particularly for graded motor imagery and mirror therapy, 43 whereas some others possibly do not. Unfortunately, none of patients included in our study had been treated with these specific physiotherapeutical approaches.
In our sample, fracture as an inciting event is largely represented (85.8% of patients) and likely due to the recruitment setting. Our Unit within a hospital setting is only devoted to bone and joint diseases and most of the recruited patients came from the emergency department and orthopaedic services. It has been reported that a persistent disease seems to be primed by a fracture as inciting event. 44 Nevertheless, patients with a CRPS triggered by a fracture did not show an increased risk to develop a permanent disability. This result could conceivably be consistent with the greater effectiveness of parenteral BP treatment in patients with CRPS-1 with a fracture as inciting event. 40
Regarding the safety issue, the absolute lacking of long-term side effects related to BP treatment confirms the excellent safety profile already reported after 1 year. 20 This further supports that a 10-day treatment cannot be considered a risk factor for adverse events observed in long-term BP users (osteonecrosis of the jaw, atypical fractures).
Study Limitations and Strengths
This study has some strengths and weaknesses. Patients were recruited according to 2013 IASP diagnostic criteria for research purpose, recognized as the diagnostic instrument with the highest level of specificity. 32 The final sample was selected starting from a series of consecutive patients who have joined the study with a low attrition rate. The same personnel using the same methodology assessed symptoms and signs of the disease along the study. However, some information possibly useful to explore the persisting disabling features (eg, work status and occupational recovery, psychosocial factors, legal claims) was not retrieved. Moreover, accounting for the number of patients who developed CRPS-1 after a fracture, the long-term disability measure would also be an outcome of the fracture itself, regardless of an unfavourable CRPS-1 course. 45 Finally, this study was performed in a specialist centre and the generalizability of the results needs to be confirmed in other clinical settings to include the CRPS-1 population as a whole. Owing to the retrospective observational design and the fact that patients consecutively arrived, no formal power calculation was performed.
Conclusions
This is the first real-life study exploring the long-term effectiveness of neridronate infusions to treat CRPS-1 and prevent a permanent loss of function in patients with hand disease. By considering these results together with the short-term efficacy in patients with an acute disease, it is an allowed expectation that an effective treatment administered soon after the onset could result in a full recovery.
