Abstract
Objectives:
At normal doses of trimethoprim-sulfamethoxazole (TMP/SMX), trimethoprim inhibits tubular creatinine secretion, leading to a rapid but reversible increase in serum creatinine (SCr). Although patients with connective tissue diseases are often in the state of immunosuppression and TMP/SMX is an important prophylactic drug, clinicians often have to stop or reduce the dosage due to concerns regarding its effect on renal function. This study aimed to evaluate the effect of a prophylactic dose of TMP/SMX on SCr in Japanese patients with connective tissue diseases, the extent of SCr level elevation and the independent risk factors for creatinine elevation.
Methods:
A retrospective cohort study was undertaken. Participants included patients with connective tissue diseases who were treated with a prophylactic dose of TMP/SMX between 2004 and 2018. Using single and multiple regression analyses, the risk factors that affected SCr elevation were evaluated.
Results:
A total of 262 patients, females, n = 181; age, median (range) = 59 (19-89) years, were included. The median baseline SCr level before treatment was 0.62 (0.16-2.1) mg/dL. The median SCr elevation value was 0.07 (−0.54 to 0.84) mg/dL in 4 weeks after TMP/SMX initiation. Five (2%) participants had ⩾0.3 mg/dL SCr elevation. Multiple regression analyses, including age, baseline SCr, diuretic use, nonsteroidal anti-inflammatory drug use and diabetes mellitus, indicated that baseline SCr and advanced age were independent risk factors of SCr elevation.
Conclusions:
These results demonstrated that baseline SCr and advanced age were associated with SCr elevation by a prophylactic dose of TMP/SMX. However, a prophylactic dose of TMP/SMX rarely elevated the SCr level significantly. Therefore, other causes can be considered if patients show an SCr elevation ⩾0.3 mg/dL.
Keywords
Introduction
Prolonged treatment with high-dose steroids is a major risk factor for pneumocystis pneumonia (PCP) in patients with rheumatic diseases. 1 Patients with immune dysfunction induced by an inflammatory disease who receive more than 20 mg/d prednisolone for longer than 2 to 3 weeks should receive PCP prophylaxis.2-5 Prophylactic trimethoprim-sulfamethoxazole (TMP/SMX) substantially decreases the incidence of PCP in patients with rheumatic diseases who receive prolonged, high-dose steroid treatment.2,6,7 Low-dose oral TMP/SMX is the most common and effective prophylactic agent for PCP. 6
However, clinicians often have to stop the drug or reduce its dose owing to adverse events, such as gastrointestinal symptoms, rashes, increased serum creatinine (SCr) levels, elevated liver enzyme levels, and electrolyte disorder. 1 A previous study conducted in a Japanese patient population reported that the rate of TMP/SMX discontinuation within 30 days after starting PCP prophylaxis was 40%. 3
Pentamidine isethionate, dapsone, and atovaquone are sometimes used as second-line treatment drugs, but these drugs are less efficacious than TMP/SMX.8,9 An effective chemoprophylaxis regimen for PCP with a high drug retention rate is important for patients with rheumatic diseases, as these patients often require long-term or sometimes life-long immunosuppressive therapy.
With the usual dose of TMP/SMX, the TMP component inhibits tubular creatinine secretion, leading to a rapid but reversible increase in the SCr level10-16 without altering the glomerular filtration rate. 17 The increase in the SCr level causes an apparent decrease in the calculated creatinine clearance.
Studies on the effect of TMP on SCr in healthy individuals have shown an average increase in the SCr level of 15% to 35%.10-16 A study on TMP and its effect in patients with a normal renal function and those with chronic renal failure reported that the SCr level in patients with a normal renal function did not differ substantially from baseline levels, whereas that in patients with chronic renal failure elevated significantly from the baseline level. 12 In a previous study of outcomes in renal transplant recipients on TMP/SMX prophylaxis for 1 year, 38% of all patients suffered a complication necessitating cessation of TMP/SMX. Most commonly, it was acute kidney injury that resolved after the cessation of the drug. 18
Cystatin C, a low-molecular-weight (LMW) (13 kD) basic protein that is produced at a constant rate by all nucleated cells, is freely filtered by the kidney and is not secreted; proximal tubule cells reabsorb and catabolize the filtered cystatin C, so that little is normally excreted in the urine. Thus, serum cystatin C level depends on actual glomerular filtration rate and is not affected by TMP. Cystatin C levels are used in estimating glomerular filtration rate (eGFR). 19 Comparing SCr-based eGFR with serum cystatin C–based eGFR before and after administration of TMP/SMX may be useful for evaluating whether occurrence of SCr elevation was due to TMP or to other causes.
When patients with connective tissue diseases (CTDs) show elevated SCr levels, it becomes difficult to distinguish CTDs from worsening glomerulonephritis because of the rheumatic disease manifesting as chronic kidney disease (CKD) or renal-related adverse drug reactions (ADRs) of concurrent drugs. It is not clear whether prophylactic doses of TMP/SMX, especially single-strength doses available in Japan, increase SCr levels in patients with CTD. The aim of this study was to evaluate the effects of prophylactic doses of TMP/SMX on creatinine level in patients with CTD, the extent of creatinine level elevation exhibited in patients, and the independent risk factors for creatinine elevation.
Materials and Methods
Patients
The inclusion and exclusion criteria were as follows. Inclusion criteria included (1) patients with CTDs and treated with TMP/SMX at a prophylactic dose from 2004 to 2018, (2) patients older than 18 years at baseline, and (3) patients who underwent blood tests between weeks 1 and 4 from baseline and within a month before baseline, that was defined as the day that a prophylactic dose of TMP/SMX with prednisolone was prescribed for the first time during the observation period. Exclusion criteria included (1) patients with acute kidney injury owing to other causes (underlying disease and comorbidities, infection, dehydration, postoperation status, gastrointestinal bleeding, or other drug use).
This study complied with the tenets of the World Medical Association Declaration of Helsinki and the current ethical guidelines; it was approved by the institutional ethics board of St Luke’s International Hospital (18-R127). Written informed consent from the patients for participation in this study was not required because the data were obtained retrospectively and analyzed anonymously.
The selected patients were divided into 2 groups based on the elevation in the SCr level from baseline within 4 weeks of initiation of TMP/SMX at a prophylactic dose: Patients with SCr elevation of <0.3 mg/dL and those with SCr elevation of ⩾0.3 mg/dL, according to the Kidney Disease: Improving Global Outcomes (KDIGO) definition and staging system definition for acute kidney injury, which is the most recent and preferred definition. 20 The KDIGO guidelines define AKI as follows: (1) increase in SCr by ⩾0.3 mg/dL (⩾26.5 µmol/L) within 48 hours; (2) increase in SCr to ⩾1.5 times baseline, which is known or presumed to have occurred in the past 7 days; or (3) urine volume of < 0.5 mL/kg/h for 6 hours. As most patients did not undergo a urine test, we classified patients according to criteria (1) or (2) to the group of patients with SCr elevation of ⩾0.3 mg/dL.
Data collection
Blood test results before the initiation of prophylaxis (baseline) and between weeks 1 and 4 from baseline, and the SCr, serum cystatin C, urine qualitative, sediment test results, and serum electrolyte levels were obtained. The SCr elevation level was calculated accordingly.
The SCr-based eGFR was calculated using the following equation 21 :
Serum cystatin C-based eGFR was calculated using the following equation 22 :
Baseline patient characteristics, including the dose of prednisolone and TMP/SMX; concomitant use of drugs, such as diuretics, antihypertensive agents, and immunosuppressants; and pretreatment comorbidities, including CKDs, cardiovascular diseases, diabetes mellitus, and malignant disease were also obtained.
Chemoprophylaxis and patient follow-up
The TMP/SMX was the only drug used for PCP prophylaxis in this study and was administered as either 160 mg TMP 3 times a week or 80 mg TMP per day. The patients for PCP prophylaxis and duration of prophylaxis were mainly determined by the treating physicians. For patients with renal insufficiency, the TMP/SMX dose was adjusted accordingly (determined by SCr-based eGFR, n = 23). Second-line antibiotics against PCP, such as dapsone, atovaquone, and aerosolized pentamidine, were not used for the primary prophylaxis against PCP during the observation period.
Statistical analyses
First, we evaluated whether patients show an elevation in the creatinine level of ⩾0.3 mg/dL according to the KDIGO definition and staging system definition for acute kidney injury. Second, we assessed independent risk factors for creatinine elevation. Finally, we compared SCr-based eGFR and serum cystatin C–based eGFR before and after the initiation of TMP/SMX. Comparisons between the groups of patients (those with SCr elevation ⩾0.3 mg/dL and those with SCr elevation < 0.3 mg/dL) were performed using Mann-Whitney U test for continuous and ordinal variables and the χ 2 -test for categorical variables. Both univariate and multivariate analyses were performed using linear regression analysis to assess independent risk factors for creatinine elevation. Variables based on previous literature regarding risk factors of acute kidney injury and creatinine elevation due to TMP/SMX were analyzed using a multiple linear regression model.12,23-26 All statistical tests were 2-tailed, and the results were considered significant at P ⩽ .05.
All analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). EZR is a modified version of R commander designed to add statistical functions frequently used in biostatistics. 27
Results
Four hundred eighty-one patients with CTDs, older than 18 years at baseline and treated with TMP/SMX at a prophylactic dose, were enrolled in the study from 2004 to 2018. A retrospective review of all medical records of the patients was carried out. One hundred eighty-four patients who did not undergo blood tests within the observation period and 20 patients who did not undergo blood tests within a month before baseline were excluded. Fifteen patients with acute kidney injury owing to other causes (underlying disease and comorbidities, infection, dehydration, postoperation status, gastrointestinal bleeding, and other drug use) were also excluded. The data obtained from the remaining 262 patients were finally evaluated (Figure 1).

Process scheme for the application of the inclusion and exclusion criteria to arrive at the study population. eGFR indicates estimating glomerular filtration rate; TMP/SMX, trimethoprim-sulfamethoxazole.
Two hundred sixty-two patients (females, n = 181; age, median [range] = 59 [19-89] years; specific CTD subgroups: rheumatoid arthritis [RA], n = 74; systemic lupus erythematosus [SLE], n = 73; anti-neutrophil cytoplasmic autoantibody [ANCA]-associated vasculitis, n = 28; polymyalgia rheumatica/giant-cell arteritis [PMR/GCA], n = 29; systemic scleroderma [SSc], n = 30; polymyositis/dermatomyositis [PM/DM], n = 10; mixed connective tissue disease [MCTD], n = 5; and others, n = 55) fulfilled the criteria for the analysis. The median baseline creatinine level before treatment was 0.62 (0.16-2.1) mg/dL. The median dose of TMP/SMX was 1 (0.86-1) tablet per day. The median dose of prednisolone at baseline was 25 (1-1250) mg/d. The baseline characteristics of all patients and the comparison of variables between the groups of patients are shown in Table 1.
Baseline characteristics of all patients and a comparison of variables between the groups. a .

Abbreviations: ACE-i, angiotensin-converting enzyme inhibitor; ANCA, anti-neutrophil cytoplasmic autoantibody; ARB, angiotensin II receptor blocker; CKD, chronic kidney disease; DM, dermatomyositis; eGFR, estimated glomerular filtration rate; GCA, giant-cell arteritis; MCTD, mixed connective tissue disease; NSIADs, nonsteroidal anti-inflammatory drug; PM, polymyositis; PMR, polymyalgia rheumatic; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SSc, systemic scleroderma; TMP/SMX, trimethoprim-sulfamethoxazole.
Patients showing serum creatinine elevation by <0.3 mg/dL and those showing serum creatinine elevation by ⩾0.3 mg/dL.
n (%) or median (range).
The number of ANCA-associated vasculitis with renal manifestations was 12 out of 28, and 2 patients with renal manifestations showed serum creatinine elevation by >0.3 mg/dL.
The median SCr elevation level was 0.07 (−0.54 to 0.84) mg/dL. Among patients whose SCr-based eGFR was less than 60 mL/min, the median SCr elevation level was 0.07 (−0.54 to 0.31) mg/dL. In all, 75% of all patients showed SCr elevation after administration of TMP/SMX (matched paired t, P < .001). Distribution of SCr changes is shown in Figure 2.
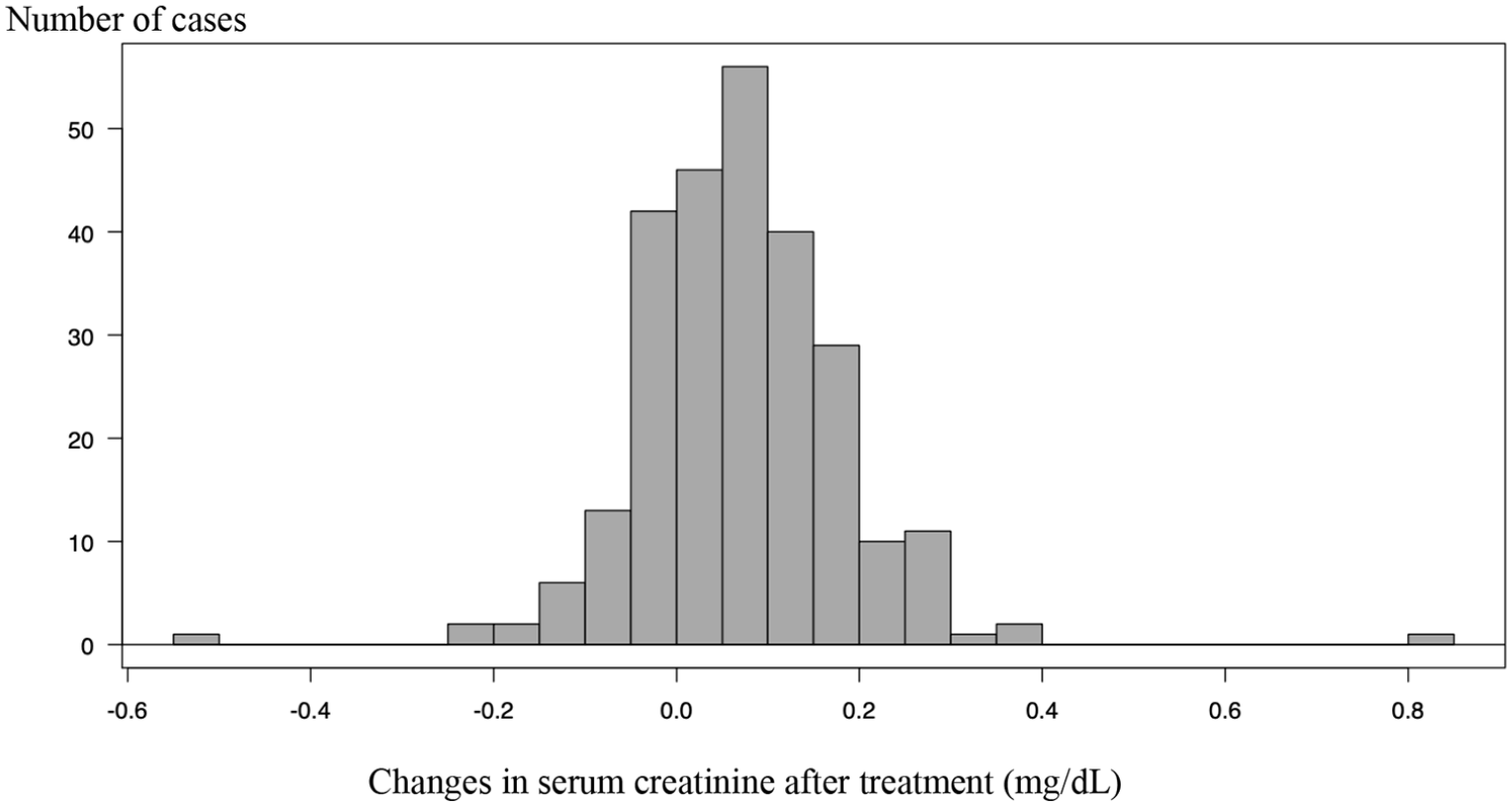
Changes in serum creatinine after treatment.
However, only 5 patients (2%) had an elevation in SCr by ⩾0.3 mg/dL. The details of the 5 patients are shown in Table 2. Two out of 3 patients who showed an elevation in SCr by ⩾0.3 mg/dL within a week from the baseline had ANCA-associated vasculitis with nephritis. The remaining 2 patients showed an elevation in SCr by ⩾0.3 mg/dL at 4 weeks from the baseline.
Details of 5 patients exhibiting elevation in the serum creatinine level by ⩾0.3 mg/dL.

Abbreviations: BUC, bucillamine; CKD, chronic kidney disease; CVD, cardiovascular disease; CyA, cyclosporine; eGFR, estimated glomerular filtration rate; GPA, granulomatosis with polyangiitis; GVHD, graft-versus-host disease; HTN, hypertension; MCTD, mixed connective tissue disease; MPA, microscopic polyangiitis; MTX, methotrexate; MZR, mizoribine; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis.
All patients continued to receive trimethoprim-sulfamethoxazole (TMP/SMX) during the observation period and showed return to the baseline level of serum creatinine shortly after the discontinuation of TMP/SMX.
Patient 1 showed serum creatinine elevation by >0.8 mg/dL.
Exacerbation of GPA was not detected in physical examination and blood and urine tests.
The patient started to take bucillamine 1 year before showing elevation in the serum creatinine level.
Serum trough of CyA was 111 ng/mL and within appropriate range.
Serum creatinine value returned to the baseline 1 week after the cessation of TMP/SMX.
Administration of TMP/SMX was reinitiated, and the serum creatinine level elevated again.
Among the 17 patients who underwent blood tests for cystatin C at the baseline and after the initiation of TMP/SMX, 1 patient whose SCr-based eGFR at baseline was <30 mL/min was excluded, and the remaining 16 patients were evaluated for changes in SCr-based eGFR and serum cystatin C–based eGFR. The median changes of SCr-based eGFR and serum cystatin C–based eGFR were −8 (−89 to −2) mL/min (matched paired t, P = .0235) and 4.7 (−11.7 to 16.6) mL/min (matched paired t, P = .135), respectively (Table 3). A graph of the upward slope was obtained by taking a ratio of SCr and serum cystatin C before and after administration of TMP/SMX, because SCr level was elevated and serum cystatin C level did not change after treatment (Figure 3).
Change in serum creatinine-based eGFR and serum cystatin C–based eGFR after the administration of trimethoprim-sulfamethoxazole in 16 patients whose cystatin C results were available.

Abbreviations: eGFR, estimated glomerular filtration rate; SCr, serum creatinine.

Change in the serum creatinine/serum cystatin C level after the administration of trimethoprim/sulfamethoxazole in 16 patients whose cystatin C results were available.
Multivariate analysis was performed using the following variables: age; baseline SCr level; use of loop diuretics, spironolactone, and nonsteroidal anti-inflammatory drugs (NSAIDs); and a history of diabetes mellitus. The results of the univariate and multivariate analyses are presented in Table 4. The multivariate analysis revealed that the baseline SCr level and older age were independent risk factors. The regression coefficient and P value for age and SCr were .0012 (95% confidence interval [CI]: 0.00037-0.002) and P = .0043, and −.0098 (95% CI: −0.155 to −0.042) and P = .00076, respectively.
Results of the univariate and multivariate analyses for risk factors of serum creatinine elevation.
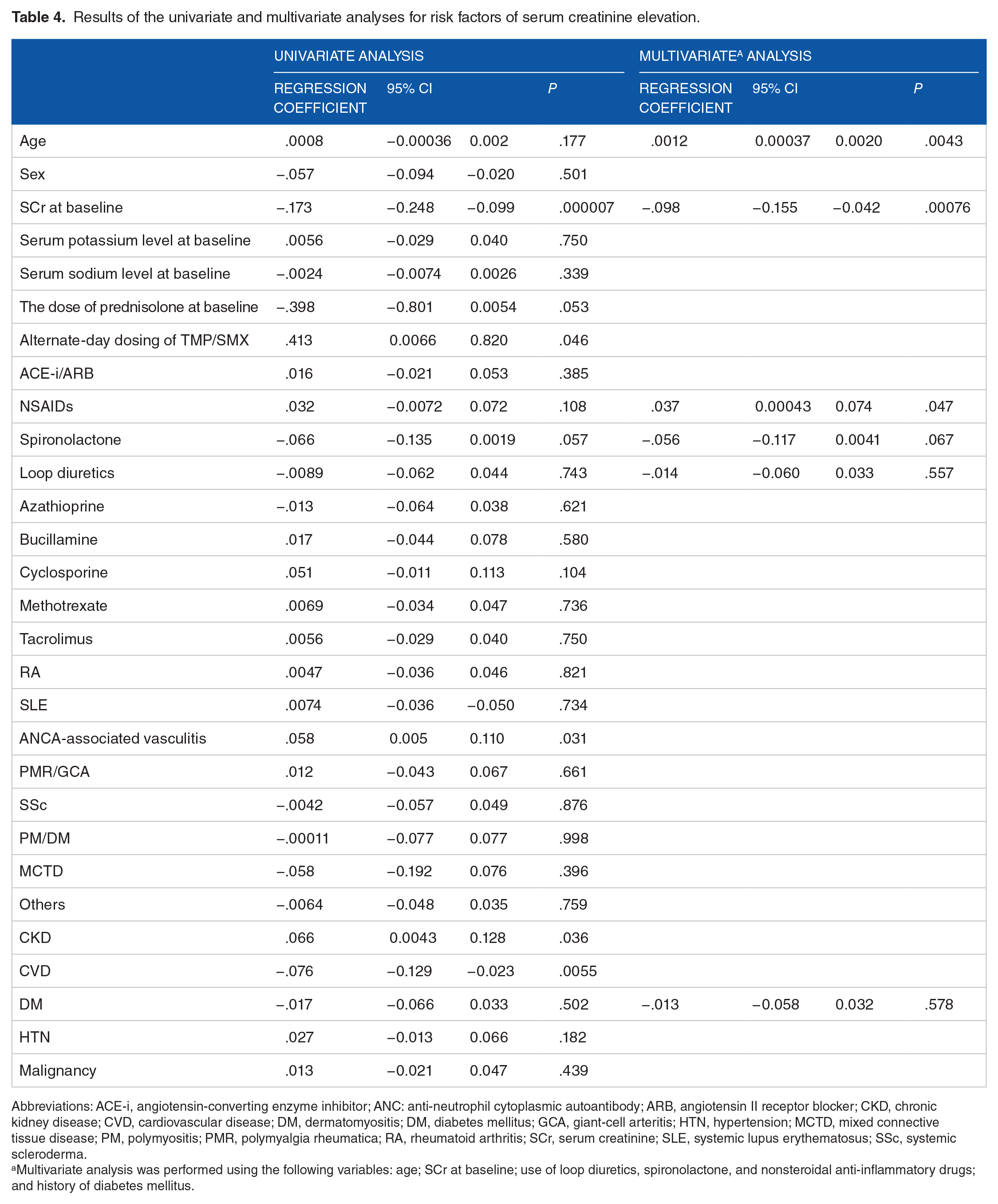
Abbreviations: ACE-i, angiotensin-converting enzyme inhibitor; ANC: anti-neutrophil cytoplasmic autoantibody; ARB, angiotensin II receptor blocker; CKD, chronic kidney disease; CVD, cardiovascular disease; DM, dermatomyositis; DM, diabetes mellitus; GCA, giant-cell arteritis; HTN, hypertension; MCTD, mixed connective tissue disease; PM, polymyositis; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis; SCr, serum creatinine; SLE, systemic lupus erythematosus; SSc, systemic scleroderma.
Multivariate analysis was performed using the following variables: age; SCr at baseline; use of loop diuretics, spironolactone, and nonsteroidal anti-inflammatory drugs; and history of diabetes mellitus.
The continuation rate of TMP/SMX for 12 weeks was 85%. The median dose of prednisolone at week 12 in patients who continued TMP/SMX for 12 weeks was 5 (1-90) mg/d. In the remaining 15% of the patients, 13 and 20 patients stopped TMP/SMX before 12 weeks because of adverse reactions and a decrease in the dose of prednisolone to <15 mg/d, respectively. One patient switched to atovaquone according to the request of the patient. One patient with systemic sclerosis and 1 patient with dermatomyositis died of deterioration of interstitial lung disease owing to an underlying disease. One patient developed PCP and the dose of TMP/SMX was increased. Three patients were not followed up because of transfer to another hospital.
During the observation period, ADRs occurred in 33 patients (13%), and 13 patients (5%) discontinued TMP/SMX (Table 5). The most common ADRs were hyperkalemia (serum potassium level >5 mEq/L) in 22 patients (8%), followed by a skin rash in 5 patients (2%). In 5 out of the 22 patients with hyperkalemia, TMP/SMX was discontinued; in 3 patients within 1 month and in 2 patients within 2 months. Seventeen patients continued TMP/SMX for 12 weeks (maximum serum potassium level = 5.4 mEq/L). Five out of the 17 patients had underlying CKD, and hence, hyperkalemia at baseline. Hyponatremia (serum sodium < 130 mEq/L) occurred in 4 patients, and 1 patient showed hyponatremia at baseline. However, all patients with hyponatremia were asymptomatic and continued TMP/SMX for 12 weeks. The ADR severity was mild to moderate. None of the patients had serious ADRs that led to prolonged hospitalization. All patients recovered shortly after the discontinuation of TMP/SMX. There was one case of PCP during prophylaxis.
Adverse drug reactions due to trimethoprim-sulfamethoxazole administration.

Conversely, 120 (55%) out of the 219 patients who were excluded could continue TMP/SMX for 12 weeks. In the remaining 45% of the patients, 27 and 29 patients stopped TMP/SMX before 12 weeks because of adverse reactions and a decrease in the dose of prednisolone to <15 mg/d, respectively. Five patients stopped TMP/SMX according to the request of the patient. Four patients died of deterioration of interstitial lung disease owing to an underlying disease. One patient died of a urinary tract infection. Eight patients died of malignant cancer. Twenty patients were not followed up because of transfer to another hospital.
In the group of patients who were excluded, ADRs occurred in 27 patients (12%), and all these patients discontinued TMP/SMX. The most common ADRs were rashes in 14 patients (6%), followed by liver function test abnormality in 6 patients (3%), allergy in 5 patients (2%), hyperkalemia in 1 patient (0.5%), and hyponatremia in 1 patient (0.5%). All patients recovered shortly after the discontinuation of TMP/SMX.
Discussion
In this study, we evaluated the effect of TMP/SMX at a prophylactic dose on creatinine level in patients with CTD and factors contributing to the elevation in the creatinine level. The medical history of 262 Japanese patients with CTD treated with TMP/SMX at a prophylactic dose was reviewed retrospectively. Our study showed that the SCr level was rarely elevated (⩾0.3 mg/dL) in patients with CTD treated with TMP/SMX at prophylactic doses.
Studies have shown that TMP/SMX at prophylactic doses is useful in the prevention of PCP. 28 However, the elevation in SCr is one of the effects caused by the drug that may limit its use. It is difficult to distinguish the elevation in SCr from renal damage owing to other causes, and the drug is often stopped because of the elevation in the SCr level. The risk factors for SCr elevation with TMP/SMX at prophylactic or therapeutic doses have been investigated previously; however, data on the level of increase in SCr by TMP/SMX at the prophylactic dosage in Japanese patients with CTD are scarce. Herein, we present the findings from 262 patients with CTD and conclude that the clinically significant increase in SCr by ⩾0.3 mg/dL after initiating TMP/SMX prophylaxis is rare (2%), although 75% of all patients showed SCr elevation after treatment. These results are consistent with those of previous studies showing that creatinine elevation after TMP/SMX therapy is dose-dependent. 29
After the administration of a single dose of TMP/SMX, approximately 60% is excreted in urine within 24 hours and 70% to 85% is excreted within 48 hours. The increase in the SCr level observed is not due to renal dysfunction but is caused by the inhibition of creatinine secretion in renal tubules by TMP. Hence, it is a functional attribute and is unrelated to drug-induced renal dysfunction. 30
Factors known to affect creatinine secretion from the tubules include drugs, such as trimethoprim, cimetidine, pyrimethamine, and dapsone, and impaired renal function. The renal tubules secrete increased creatinine with deteriorating renal function. This mechanism is saturated when the SCr level exceeds 1.5 to 2 mg/dL (132-176 µmol/L) resulting in SCr level elevation. 19
Although rare, 5 patients showed SCr elevation by ⩾0.3 mg/dL, presumably caused by TMP. One of them had ANCA-associated vasculitis with an SCr elevation of 0.8 mg/dL. In all 5 patients, TMP/SMX was discontinued, which subsequently restored the SCr level to the baseline levels. Other causes, including exacerbation of the primary disease, were ruled out based on physical examination, and biochemical and urine tests.
Increased age and high baseline creatinine levels were found to be risk factors for the elevation in the SCr level in this study, in line with the findings of previous studies. 12 As kidney function deteriorates with age, creatinine secretion from renal tubules inevitably decreases. Patients with CKD treated with 100 mg TMP every 12 hours for 10 days showed significant elevation of SCr level, which was more than 34.6% of baseline, compared with that of healthy subjects. 12
In the Japanese population, the eGFR tends to be overestimated when patients are older or have decreasing muscle mass. Although it is appropriate to evaluate creatinine clearance (CrCl) that is measured by collecting the patient’s urine for 24 hours
If patients show an SCr elevation by ⩾0.3 mg/dL while on TMP/SMX therapy, it is necessary to exclude causes other than TMP/SMX, such as exacerbation of the underlying nephritis or CKD, prerenal acute kidney injury due to dehydration or bleeding, postrenal acute kidney injury in hospitalized patients, and adverse effects of concomitant medications.
The SCr-based eGFR tended to decrease after TMP/SMX was taken (matched paired t, P = .0235). However, serum cystatin C–based eGFR did not change significantly in this study (matched paired t, P = .135). The clinical conditions and the results suggested that the elevation in SCr was because of the inhibition of tubular creatinine secretion by TMP/SMX. A comparison between SCr- and serum cystatin C-based eGFRs in 16 patients is important because it can clarify whether the creatinine elevation observed in this study is pseudo-elevation (simply caused by the inhibition of tubular creatinine secretion) or involves other causes.
Cystatin C levels were reported to be independent of sex, muscle mass, and age after 12 months of age, but there is a growing body of evidence suggesting that this may not be the case. Cystatin C levels may be affected by factors independent of renal function, such as corticosteroids, thyroid dysfunction, obesity, diabetes, smoking, and high C-reactive protein (CRP) value.31-36 A meta-analysis published in 2002 showed that serum cystatin C measured with an immunonephelometric assay is more accurate than SCr as a marker of GFR.37,38
Serum cystatin C level does react to slight deteriorations in renal function, which can cause an elevation in its serum concentration. While the SCr level is not elevated until the GFR declines to 30 to 40 mL/min, cystatin C can be detected in the early stages of renal failure, at around a GFR of 60 to 70 mL/min. 39 The concentration of serum cystatin C reaches its maximum when the renal function deteriorates to end-stage renal disease; thus, patients with an eGFR less than 30 mL/min were excluded from this evaluation. When it is difficult to distinguish whether SCr elevation has occurred due to TMP/SMX or to other causes, cystatin C might be used for evaluation.
There were some limitations to the present study. First, this was a single-center, retrospective study, and there may have been a selection bias at the time of patient exclusion. The continuation rate in the group of patients who were excluded is different from that of the study population because there was a large number of patients who discontinued TMP/SMX due to a decrease in the dose of prednisolone, transfer to another hospital, the patient’s own request, ADRs, such as rash and liver function test abnormality that warranted cessation of TMP/SMX, and death as a result of malignant cancer.
Second, the study did not include CrCl in urine tests, and only 16 cases were evaluated for SCr-based eGFR calculated with cystatin C, which was more reflective of actual renal function. Third, there are several confounding factors that can be implicated in the elevation in SCr. Comedications, including angiotensin-converting enzyme (ACE) inhibitors, cyclosporin, and NSAIDs and diseases, such as lupus nephritis and ANCA-associated vasculitis with renal involvement might have led to the elevation in SCr. The inclusion of a control group with similar manifestation and disease activity would be appropriate to attribute the renal dysfunction to TMP/SMX. Therefore, our findings need to be confirmed in a prospective study.
Conclusions
In conclusion, only 2% out of the 272 Japanese patients with CTD who received prophylactic TMP/SMX showed an increase in the SCr level by ⩾0.3 mg/dL. Increased age and high baseline creatinine levels have been shown to be risk factors for the elevation in SCr levels. Other causes of renal impairment should be considered if patients with low-dose TMP/SMX show creatinine elevation by ⩾0.3 mg/dL.
Footnotes
Author Contributions
RK and RR conceived the presented idea. RK developed the theory, performed the computations, and wrote the manuscript. MO, MM, KY, and NT reviewed the manuscript.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Human and Animal Rights
This study was conducted in accordance with the Declaration of Helsinki and was approved by the institutional ethics board of St Luke’s International Hospital (18-R127). Written informed consent for participation in this study was not required from the patients because the data were collected retrospectively and analyzed anonymously.
Data Availability Statement
The data underlying this article are available in the article.


