Abstract
Chronic lower back pain is one of the most common medical conditions leading to a significant decrease in quality of life. This study retrospectively analyzed whether the AxioBionics Wearable Therapy Pain Management (WTPM) System, a customized and wearable electrical stimulation device, alleviated chronic lower back pain, and improved muscular function. This study assessed self-reported pain levels using the visual analog scale before and during the use of the AxioBionics WTPM System when performing normal activities such as sitting, standing, and walking (n = 69). Results showed that both at-rest and activity-related pain were significantly reduced during treatment with the AxioBionics WTPM System (% reduction in pain: 64% and 60%, respectively;
Introduction
Pain not only affects individuals physically but can have detrimental emotional, mental, and financial effects as well. Approximately 11% of Americans aged 20 years and older experience chronic pain that lasts more than 1 year. 1 This figure increases to 17.7% when including adults who have experienced pain lasting for at least a month. 2 According to a survey, 13% of the US workforce has experienced a loss in productivity, either by decreased productive working hours or time off from a job, 3 and the annual estimated cost of this productivity loss was $61 billion. 3
Despite the numerous forms of pain management available, pain relief can be elusive for many individuals. In a survey of more than 2600 Americans with chronic, severe, non-cancer-related pain conducted in 1998 47% reported that they had changed their doctor, and 22% had done so 3 times or more. The most common reason for this change was the intense, remaining pain (42%).4,5
Low back pain (LBP) is defined as pain, muscle tension, or stiffness localized below the costal margin and above the inferior gluteal folds, with or without leg pain (sciatica). 6 Chronic LBP is a common neurological ailment, second only to headaches, and the most prevalent cause of disability that affects work productivity. 1 Approximately 28% of the patients surveyed with LBP reported a limitation of activity due to their condition. 1 They also had co-morbid manifestations such as loss of sleep, increased muscle stiffness, and reduced spine mobility, often leading to reduced daily energy and mental focus, depression, and job loss.3,7-9 Total annual health care expenditures attributed to back pain are estimated at $26.1 billion. 2
Standard therapies for chronic LBP include administration of pain medications (eg, opiates and non-steroidal anti-inflammatory drugs), physical therapy (PT), chiropractic care, transcutaneous electrical nerve stimulation (TENS), and epidural steroid injections. These treatments can effectively mitigate pain in patients with LBP; however, in those who do not fully respond to them, a combination of treatments may be needed to optimize results.
Pain medications have played a major role in managing chronic LBP for many years; however, according to Chou et al, 10 there is no medication superior to others because there are complex cost/benefit trade-offs to consider. This conclusion is consistent with another study, suggesting that opioids are not superior to non-opioid medication regarding chronic back pain alleviation. 11 In a recent systematic review and meta-analysis, Busse et al 12 concluded that opioids have a statistically significant impact, but a small improvement in pain and physical functioning compared to placebo. In another study, the authors concluded that there was no evidence supporting opioid therapy for moderate to severe chronic back pain. Additionally, opioids induce unwanted side effects such as sedation, dizziness, nausea, vomiting, constipation, physical dependence, tolerance, and respiratory depression. 13 In general, both short and long-term pain medication regimens can potentially cause undesirable side effects and health risks, thus contributing to a decrease in quality of life and poor response in pain reduction, both at rest and during physical activity. To further complicate the matter, the use of opioid therapy has contributed to a significant public health crisis in the United States, where more than 63% of the 52 404 drug-related overdose deaths in 2015 were opioid-related.14,15
PT also has limitations. Patients undergoing various PT-related treatments may not be relieved of pain either after each session or at the end of a multi-month program. Fritz et al 16 found no improvement in pain intensity at a 4-week, 3-month, or 1-year follow-up when comparing patients receiving conventional care and those undergoing PT. In a study comparing the McKenzie and Back School methods for treating LBP, Garcia et al 17 reported a 19% and 25% reduction in pain, respectively, while at a 6-month follow-up, there was an average pain intensity of 5.2 between the 2 methods. In a systematic review of PT techniques for managing LBP, the authors concluded that the treatment ineffectively managed pain. 18 In a retrospective study of 4597 patients with LBP, Eleswarapu et al 19 concluded that a substantial percentage of patients did not meet minimal clinically important difference (MCID) for pain and function, following LBP treatment with PT. Fritz also concluded that PT did not result in MCID among adults; however, this study cohort presented recent-onset LBP. 16
Considering the lack of sufficient pain relief and drawbacks associated with medication and PT, TENS has been considered among the choices for LBP management, especially considering the low risk imposed on patients. However, its efficacy has been questioned by the Department of Health and Human Services due to the lack of supporting scientific evidence. 20 In our experience, TENS can fail in pain alleviation for a variety of reasons, including electrode placement errors, electrode fixation issues, low power output, and lack of professional instruction. In a recent systematic review, the benefits of TENS in patients with LBP was found inconclusive because the quality of the studies was low, and adequate parameters and timing of assessment were not uniformly used or reported. 21 This study highlighted the importance of defining methods, electrical parameters, and quality of research protocols to better understand TENS.
TENS attempts to control pain by blocking the pain pathway proposed in the Gate Control Theory. 22 TENS is designed to stimulate only the cutaneous afferent nerves, without penetrating deeper into the body to stimulate and relax muscle spasms. This may be 1 reason some patients experience spasm relief while others do not. In a study of the direct and indirect benefits of TENS, Gladwell et al, 23 found that while 89% of subjects with musculoskeletal pain had pain relief from TENS, only 71% had relief of muscle spasms. An additional drawback of TENS is that large electrodes cannot be used to cover a large area of pain as it could significantly reduce the current output and distribution, thereby conferring limited pain relief. In addition to the deficiencies in its underlying principles, standard TENS technology, which uses adhesive skin electrodes, can fail to lower pain due to various issues. Patients with LBP commonly experience difficulty in placing the electrodes on the lower back due to the inability to twist their body or see the affected area. Even if patients succeed in placing the electrodes, there is no guarantee that the electrodes will be placed accurately or that they will stay affixed throughout the day. Sweat and body hair can interfere with electrode adherence, conductance, and performance. Furthermore, patients can experience contact dermatitis due to the chemicals present in the electrodes, which, for example, are added for adherence of electrodes to the skin. 24 A high prevalence of skin irritation has been noted in 40% after the use of TENS 25 and which can lead to rejection and failure of the therapy.
Although TENS can be useful in managing pain when being on the body, it loses its effect when removed, and there is no long-term, residual carry-over benefit. A meta-analysis of patients with chronic LBP showed that TENS was significantly different from placebo/control (
The output signal generated by TENS is much lower than that of neuromuscular electrical stimulation (NMES). There is growing evidence that NMES on trunk muscles in patients with LBP may increase core muscle strength, reduce pain, and improve function when compared to conventional care. The total charge generated is calculated by multiplying the amplitude and pulse width. TENS, with a maximum amplitude of 60 mA and 150 µs pulse width, produces a total charge of 9 µColoumbs. In contrast, NMES, with a maximum amplitude of 100 mA and a pulse width of 300 µs, produces a total charge of 30 µColoumbs and therefore is 3.3 times stronger than TENS. To stimulate muscle, an NMES signal must penetrate deeper than subcutaneous tissue to depolarize motor neurons and must be of sufficient current intensity to reach back muscles such as the multifidus muscle. 27 A deeply penetrating electrical signal is more effective at reducing pain than TENS in a comparative study of varying electrical stimulation devices. 28 Interestingly, there is scientific evidence for combining TENS and NMES into a treatment modality for pain. Moore and Shurman 29 assessed the use of TENS, NMES, and TENS-NMES and concluded that their combination was superior to TENS or NMES alone at reducing chronic back pain.
There is a growing body of evidence suggesting that NMES may be beneficial as a modality to strengthen muscles as well as to facilitate a reduction in chronic LBP.
30
In one study, electrodes were positioned over the abdominal and lower back paraspinal muscles using intensities sufficient to produce muscle contraction. The results revealed clinically and statistically significant improvements in both muscle groups’ performance, and the results were associated with significant improvements in self-reported pain levels, suggesting that NMES plays an important role in chronic LBP rehabilitation.
31
Another study showed that electrical stimulation significantly increased (
Volitional exercise may not provide enough tension through the atrophied type II fibers to induce hypertrophy of these fibers even when performed at near maximum intensities. 33 In a Cochrane review investigating physical activity and exercise, exercise did not consistently cause any change (positive or negative) in self-reported pain scores. 34 However, NMES has been shown to recruit type II muscle fibers and induce an increase in muscle strength,29,35-37 and this may have a beneficial effect on chronic LBP and function.33,38
Not all studies managed to show benefit from the use of NMES in chronic LBP. In a study by Guo et al, 39 there was no statistical difference between the NMES and control groups regarding pain reduction or disability score after a 4-week treatment period. However, the authors indicated that the study had limitations that should be overcome to produce a more confident conclusion in the future. NMES dose may need to be greater than that implemented in this study to effectuate positive changes in LBP.
An alternative to conventional care is needed to improve pain modulation and reduce the potential risks for patients. Thus, we investigated the effectiveness of the AxioBionics Wearable Therapy Pain Management (WTPM) System in the treatment of chronic LBP. This system employs both NMES and TENS technologies, which are integrated into a wearable system that automatically aligns electrodes on the body and keeps them affixed to it. It is hypothesized that the WTPM System would reduce at-rest and activity-based LBP.
The WTPM System employed both TENS and NMES to maximize the therapeutic result. The TENS modality (low-level, sensory stimuli) was employed to alleviate pain and the NMES (moderate-level, supra-sensory, supra-motor threshold) to help reduce muscle spasms and atrophy27,33 and enhance the pain alleviation effect over time. It has long been acknowledged that conditioning of the trunk muscles is effective in the management of acute and chronic LBP. 27 Rather than relying solely on TENS, the WTPM System sought to improve patient outcomes by combining these 2 modalities into a wearable system. The WTPM System assured proper placement and fixation of electrodes to ensure that electrodes were aligned over areas of pain and motor points. To our knowledge, this is the first study to delineate the effects of an electrical stimulation system on at-rest and activity-based pain.
Methods

The method used to stimulate the area of pain during the NMES/TENS trial period.
After the 4-week home trial, the patients returned to AxioBionics for follow-up, and the results of the NMES/TENS trial were documented. The test garments were fabricated from the measurements of the patients and individually tailored for a secure fit. The fitting process led to the customization of the WTPM Systems that captured the array of 4 electrodes that optimized pain relief for each patient. Each patient received a WTPM System configured as a belt, a brief, belt/sleeve, or shorts (Figures 2 and 3). For those who purchased the WTPM System following the 4-weeks, additional data were collected for another 4 weeks. No further data collection was performed on patients who did not purchase the system after the 4-week trial.

Various configurations of the WTPM system.

Actual depiction of the belt version of the WTPM system used to treat LBP.
Results
Table 1 shows the distribution of past treatments that patients underwent before treatment with the WTPM System.
Involvement in past treatments (N = 69).

Forty-eight of the 69 patients (70%) underwent the 4-week trial period and subsequently were fitted with the WTPM System, whereas 21 patients (30%) only underwent the 4-week trial.
Abbreviations: N, number of patients; TENS, transcutaneous electrical nerve stimulation.
Tables 2 and 3 show the change in pain levels before and during treatment with the WTPMS at-rest and with activity, respectively and whether the treatment met minimal important difference (MID).
Change in pain levels (at-rest).

Abbreviations: Tx, treatment; N, number of patients.
Change in pain levels (during activity).

Abbreviations: Tx, treatment; N, number of patients.
Table 4 shows the percentage of patients who achieved 100% reduction in pain during the trial period, when fitted with the WTPMS and the combination. Table 5 shows a comparison of the increase in pain related to activity with standard care and with the WTPMS.
Percent of patients who achieved 100% pain relief.

Abbreviations: N, number of patients; Tx, treatment.
Comparison of the increase in activity-related pain.
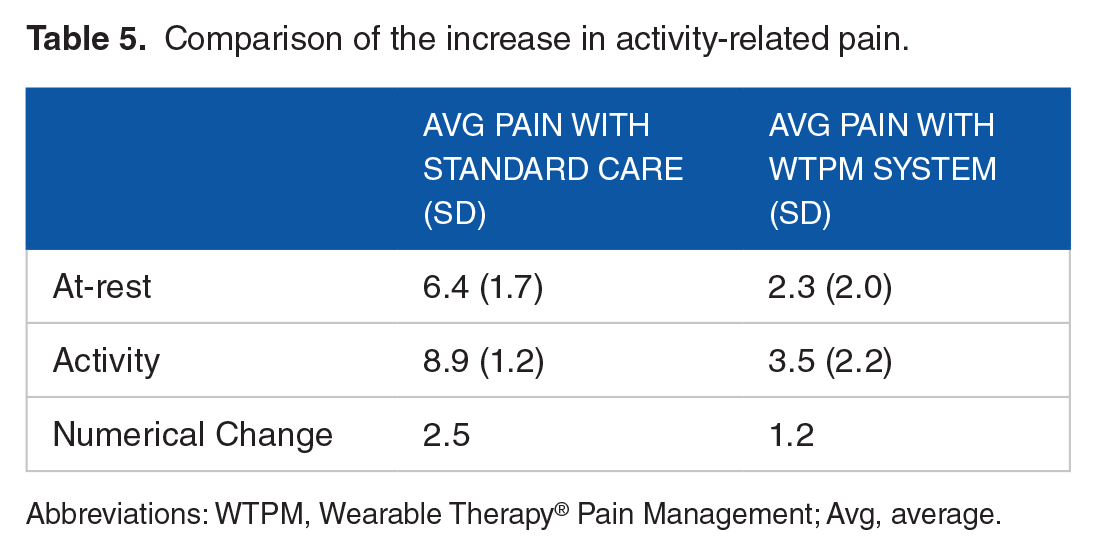
Abbreviations: WTPM, Wearable Therapy® Pain Management; Avg, average.
A 1-way univariate analysis of variance test was performed to assess the changes in pain between all combinations of groups (Table 6). Combinations not listed had insufficient sample size for statistical calculations. Significant differences are not present among the listed categories or the combinations of categories.
One-way univariate analysis of variance test results for the changes in pain between all combinations of groups.

Abbreviations: AgeCategory, the category of the age; PainMeds, pain medications; PhysTherapy, physical therapy; TENS, transcutaneous electrical nerve stimulation.
Figures 4a and b show the mean pain scores for all patients at baseline with the WTPM treatment and changes in pain scores. Both at-rest and activity-related pain significantly reduced during treatment with the AxioBionics WTPM System (

(a) AxioBionics Wearable Therapy Pain Management System significantly decreases both at-rest and activity-related pain, and (b) change in activity-related pain was significantly greater than that in at-rest pain with the use of the AxioBionics Wearable Therapy Pain Management System.
Figure 5 shows the number of patients who were stratified into each of the 3 categories of pain (low, medium, and high) based on their VAS scores. Although the bulk of the patients belonged to the high pain group before treatment, most of them were reassigned to the low or medium pain groups during the WTPM treatment.

Separation of the visual analog scale into 3 categories. At-rest and activity-related pain are shown by the number of patients before and during treatment with the Wearable Therapy Pain Management System.
Additional analyses were performed on the data of the study subgroups. Among the 56 patients whose data of at-rest pain (sitting) were recorded before the use of the WTPM System, all patients had difficulty in sitting for varying periods, ranging from 1 minute to 8 hours (Figure 6). Forty-nine of these patients reported improvement in their sitting ability using the WTPM System. The minimum improvement in sitting time was 50%. Figure 3 shows the number of patients in each range.
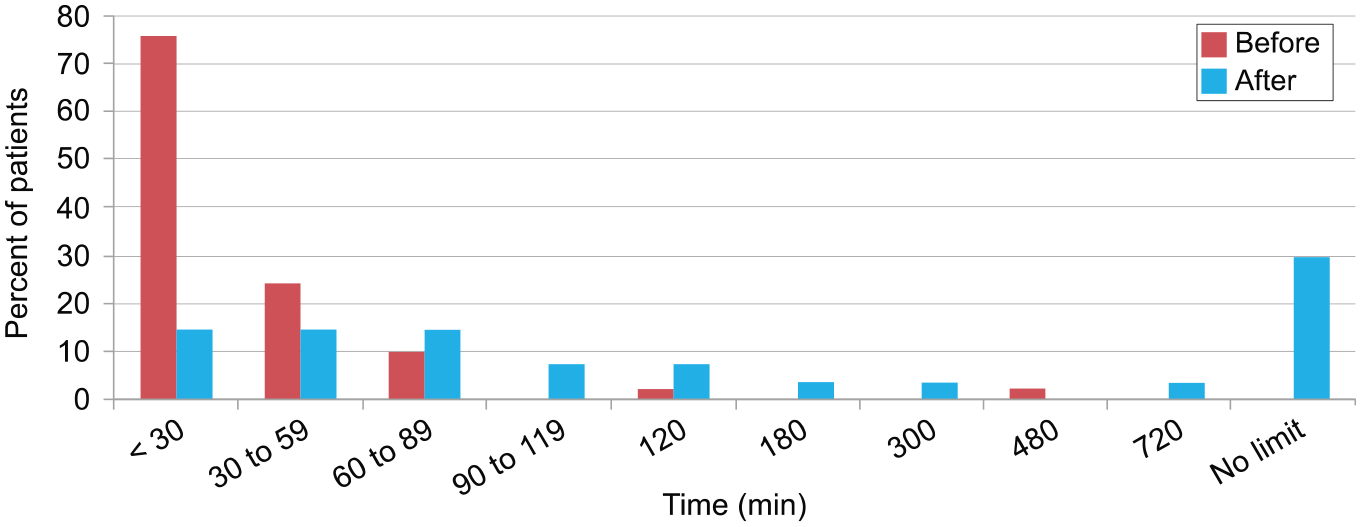
Percentages of patients categorized by the time they could sit before (N = 50) and during (N = 27) treatment with the Wearable Therapy Pain Management System.
Standing ability was also limited before the use of the WTPM System in all assessed patients (n = 49); times are shown in Figure 4. During the use of the WTPM System, 97.7% of the patients (n = 44) reported standing improvement. The minimum improvement in the amount of time the patients could stand was 40% (n = 20); the periods are shown in Figure 7.
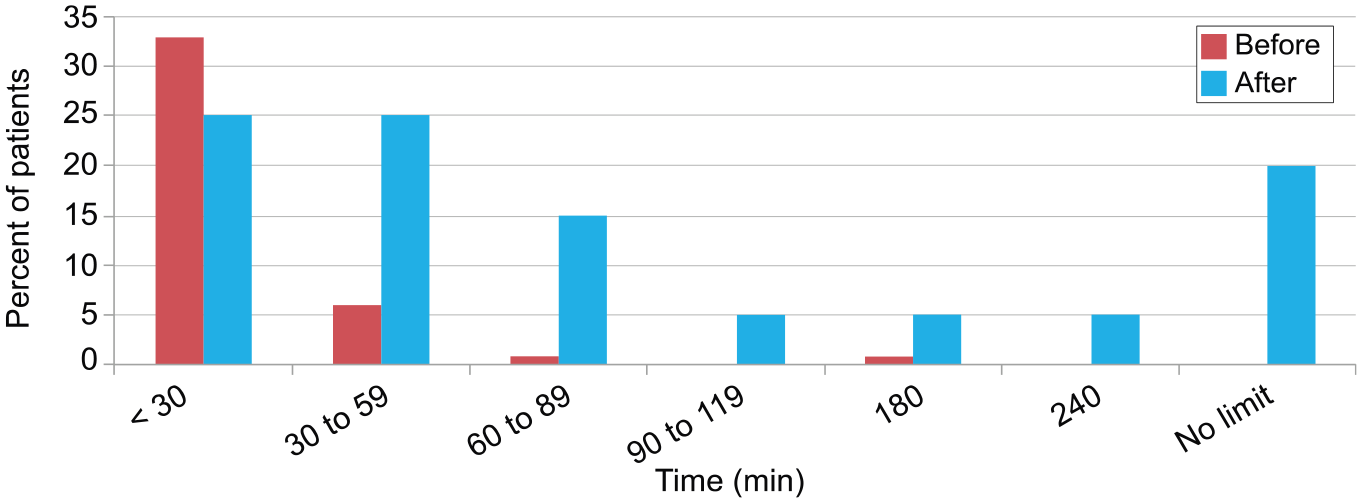
Percentages of patients categorized by the time they could stand before (N = 41) and during treatment (N = 20) with the Wearable Therapy Pain Management System. The minimum increase in standing time was 40%.
Overall, 96% of the patients who were investigated (n = 50) reported interruptions in their sleep. All 29 patients who were asked reported sleep improvements. All patients reported walking and concentration improvements, as well as decreased muscle spasm and stiffness; however, there were no consistent measures used that provided more quantitative results across the patient population. A graphical representation of the WTPM System pathway is provided in Figure 8.

Graphical representation of the WTPM system pathway.
Discussion
This study retrospectively investigated the data of all patients who were treated with the AxioBionics WTPM System for chronic LBP and elucidated the impact of the WTPM system on both at-rest and active-movement pain. Data collected from 69 patients, diagnosed only with LBP, were analyzed to determine the therapeutic effectiveness of the AxioBionics WTPM System to relieve LBP. The results showed that the WTPM System significantly decreased both at-rest and activity-related pain; given that these patients came to AxioBionics after undergoing conventional therapies and treatments, the outcome of this study suggested that the WTPM system was more effective at alleviating LBP in patients who did not respond well to conventional treatments. Our results suggest that high-level active pain can be adequately controlled with the WTPM System, perhaps better than common treatments for chronic LBP.
Our results showed a significant 2.5 level VAS increase in patient movement-related pain relative to the at-rest category in the standard treatment grouping, whereas, in the patients using the WTPM System, there was only a minor increase of 1.2. The fact that the WTPM System lowered both at-rest and activity-based pain is a substantial finding and may be the reason for the concomitant increase in the ability to sit and stand for a longer period while wearing the WTPM System during activities of daily living. It also suggests the importance of keeping electrical stimulation placed on the body with the stimulus on, to ensure that activity-related pain is held in check. We can postulate from these results that TENS/NMES is more effective at pain relief when operating on the body than when it is removed, and that it can play a significant role in helping patients remain active for a longer period. Resende et al 21 showed that pain relief from TENS abates when the stimulus is removed, whereas, our results indicate that it should remain on the body as much as possible to achieve the optimum relief either at-rest or when physically active. Although this study did not compare the efficacy of adhesive electrodes and the WTPM System, keeping electrodes firmly affixed to the body with a garment system may significantly help patients achieve longer wear time without losing electrode contact. Future studies should investigate the electrode interface to elucidate the effectiveness of these 2 systems.
Another significant finding of this study was the number of patients who reported no pain when wearing the WTPM System when at-rest (32%) and when active (8.7%), and while no one reported having no pain in the active group during the WTPM System trial, 13% of those who were fit with the WTPM System reported having no pain despite an activity increase.
Pain relief significance achieved with the WTPM System for LBP
Two researchers have derived standards for meaningful pain reduction that we can use as measures. Wickström and Edelstam studied women with endometriosis and found that, to achieve a minimally important difference, a 50% pain reduction had to be achieved. 26 Jensen, et. al. studied postoperative pain and concluded that to meet clinical significance, a 33% pain reduction had to be achieved. 40 In this study, both at-rest and activity-related LBP reductions averaged at 64% and 60%, respectively, which is well above these 2 standards.
As can be seen from these results, pain can fluctuate from a lower to a higher value when patients become more active; therefore, it is important to know if treatment for pain can impact both values and whether that reduction will lead to an improvement in function and work capability. Our results show a concomitant increase in function (sitting and standing) when pain is lowered and held in check as movement increases.
It is generally understood that not all treatments for chronic LBP will result in a favorable outcome. This study managed to show that, even when pain relief is inadequate from standard clinical options, such as pain medications, PT, and TENS, the WTPM System is a reasonable and efficacious alternative solution. The results also suggest that the WTPM System is equally effective at alleviating LBP in patients with and without a pain medication regimen.
Following, we present a brief discussion regarding possible mechanisms of action regarding pain improvement, following this study’s supporting evidence of its null hypothesis (reduced LBP). We present 7 mechanisms that could contribute to pain reduction; (1) gate control theory; (2) muscle spasm reduction; (3) endogenous opiates; (4) muscle strengthening from increasing physical activity; (5) muscle strengthening derived from stimulated muscle; (6) improved spinal posture; and (7) increase in intra-abdominal pressure from the WTPM garment. Furthermore, longer sitting time (>7 hours/day) further increases the risk of LBP. 41 Lessening sitting time, as well as sedentarism is, therefore, a reasonable pain reduction objective.
It is beyond the scope of this study to differentiate the contribution of each mechanism; however, there may be an additive effect. For example, conventional TENS may not be equally efficacious with this new system. Furthermore, all actions invoke pain relief by naturally-occurring means and not by pharmaceutical or invasive procedures; therefore, the potential for patient harm is significantly reduced.
Although the use of the AxioBionics WTPM System showed pain improvement during sitting, standing, walking, concentration, muscle stiffness, and sleep, the type of recorded data was not consistent within each activity group. This limited the number of quantitative analyses that could be performed in this retrospective study. Therefore, future studies should include consistent and quantitative measures to elucidate the effectiveness of the AxioBionics WTPM System in LBP treatment. Finally, the sample sizes of some subpopulations in this study were not large enough to be evaluated for differences in the effectiveness of the WTPM System. Therefore, additional studies should include larger sample populations to determine whether combinations of different treatments affect the outcome of the WTPM System regarding LBP. Although this was a retrospective analysis and did not include a sham or control device, future studies would benefit from inclusion of a sham TENS/NMES WTPMS to control for the placebo effect. It should also include patient-specific pain medications and dosages to elucidate the medications’ pain-relieving effects compared to the WTPM System. Future studies should also include prospective RCTs to establish unbiased evidence of the effectiveness of this unique approach.
Conclusion
The WTPM System has the potential to provide substantial pain alleviation in patients with chronic LBP when conventional treatments fail to deliver adequate relief. It eliminates the problems that have affected surface electrode use for TENS/NMES over the years by eliminating wrong electrode placement, electrode placement difficulty, electrode to skin surface integrity, and electrode failure. Unlike many conventional treatments for LBP, the WTPM System can be used independently, non-invasively, non-pharmacologically, and for any desired period.
Footnotes
Acknowledgements
We wish to thank David Durrant, D.C., Ph.D. for his science advisory and technical support.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research project was provided by Axiobionics, LLC.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Philip Muccio is an owner of Axiobionics, LLC
Josh Schueller is employed by Axiobionics, LLC
Norm Howe is a paid consultant to Axiobioncs, LLC
Miriam van Emde Boas did not receive remuneration for her contribution to the paper
Edward Dabrowski did not receive remuneration for his contribution to the paper
David Durrant did not receive remuneration for his contribution to the paper
