Abstract
We report the case of a 46-years-old man with long-term asymptomatic hyperuricemia who started taking colchicine (0.5 mg/day) and allopurinol (100 mg/d) for normalization of biochemical values. After the third week of starting treatment, acute weakness was present; and by the fifth week, profound weakness in lower extremities and tenderness and cramps on thighs and calves with inability to climb stairs were also observed. Biochemical evaluation showed elevated muscle enzymes (creatinine kinase [CK] raised to five-folds its normal value) and electromyographic features were consistent with myopathy (at rest, fibrillations, positive sharp waves, high-frequency myotonic discharges; motor unit action potentials [MUAPs] of small amplitude, small duration, increased polyphasic Index and occasional satellite potentials; at maximal effort, interferential recruitment pattern with reduced amplitudes were observed). Normal motor and sensitive nerve conduction studies and normal late F-responses and H-reflex discarded neuropathy. Rapid improvement in muscle strength and prompt resolution of abnormal elevated muscle enzymes was observed after withdrawal of both medications. Colchicine is associated with some cases of myotoxicity but very small cases of colchicine-induced rhabdomyolysis are reported on the literature. Colchicine-induced rhabdomyolysis is related to the concomitant use of drugs (statins, steroids, erythromycin, and cyclosporine), renal, and/or hepatic impairment. To the best of our knowledge, this is an uncommon presentation of a case of colchicine-induced rhabdomyolysis reported in a patient without renal or hepatic dysfunction. Therefore, patients receiving colchicine even in the absence of renal insufficiency should be monitored for the development of myopathy and more rarely to rhabdomyolysis.
Introduction
Since the first report of myoglobin-induced renal failure after crush injury during War World II, the cause of acute rhabdomyolysis has diversified. Causes include polymyositis, heat stroke, prolonged convulsions, marathon running, hypocalcaemia, and viral illnesses (such as Epstein-Barr, Influenza, and Coxsackie viruses). Lately, drugs (alcohol included) are responsible in nearly 81% of published series of cases.1,2 Alcohol accounts for 20% of acute cases of rhabdomyolysis. 3 In the case of drugs, depending on the mechanism of action, primary or secondary rhabdomyolysis can be induced. 1 Whatever the case, drug-induced rhabdomyolysis is an infrequent and often unrecognized problem which occurs most commonly in drug over dosage and in toxicity. 4
Colchicine is a drug that has been used for inflammation for almost 250 years. 1 Colchicine is an alkaloid from Colchicum autumnale, and it has existed for centuries. Its therapeutic uses date back to the fifth century for the management of gout.5,6 Today, colchicine primary indication is for gout management; however, thanks to the capability to suppress mitosis, colchicine utility has extended to some other rheumatic and nonrheumatic disorders, such as pulmonary fibrosis, liver cirrhosis, pericarditis, psoriatic arthritis, vasculitis, scleroderma, familial Mediterranean fever, amyloidosis, Sweet’s syndrome, and Behcet’s disease. 6
Long-term colchicine therapy is rarely associated with myopathy. Myotoxicity secondary to colchicine therapy is dose dependent, and the onset of symptoms usually takes months or years to appear. 7 More recently, several reports have described the rapid onset of severe myopathy, including rhabdomyolysis after the concomitant use of simvastatin and colchicine therapy. 7 The reason for this adverse event is that colchicine used in combination with statins may induce cytoskeletal myotoxicity.8,9
For centuries, colchicine has been used to treat gouty arthritis. 9 In fact, clinical trial results have demonstrated that low-dose colchicine is effective for the management of acute flares as well as for long-term prophylactic maintenance.10,11 For that reason, the EULAR guidelines indicate that prophylaxis for acute gout attacks during the first 6 to 12 months of therapy with urate-lowering agents (such as allopurinol) can be achieved by colchicine. 12
Gastrointestinal side effects of colchicine are quite common and often minor in nature and duration; while neuromuscular toxicity is very uncommon, and rhabdomyolysis is extremely rare.6,13 Renal impairment is the primary risk factor for the development of colchicine-induced myotoxicity. 6 However, colchicine can be toxic even in patients with normal renal function.4,14 Therefore, cautious dosing of colchicine is important especially in the presence of renal impairment and any patient on colchicine should be monitored for neuromuscular adverse effects. 14
The objective of this case report is to describe a rare case of acute rhabdomyolysis in a patient with normal renal function following the use of colchicine and allopurinol for the management of asymptomatic hyperuricemia and to review the literature on this topic.
Case Report
We present the case of a 46-years-old man with no important illnesses other than asymptomatic hyperuricemia and the antecedent of small psoriatic plaques on lower limbs that disappeared without any special treatment 10 years ago. The patient started hypouricemic treatment because after three continuous annual medical evaluations ordered by preventive medicine, uric acid levels were abnormally high (8.4 mg%, 8.7 mg%, and 9.4 mg% respectively), and no gout attack was present at all. Patient had no known muscle, renal, or hepatic disease and used to consume fewer than 5 units of alcohol a week.
Patient has been complaining of a 3-week history of progressive muscle weakness and myalgia, mainly in lower limbs. These complaints were present 6 weeks after the beginning of hypouricemic treatment and prophylactic gout flare. Patient started with allopurinol 100 mg/day and the dose increased progressively 100 mg/day every 2 weeks to reach a maximal dose of 300 mg/day, together with 0.5 mg/day of colchicine to avoid gout flare.
After 3 days of starting the medication, mild diarrhea was observed, but it disappeared spontaneously. By the third week, progressive myalgia and by the fifth week, progressive weakness with difficulty to climb stairs and general malaise made the patient to stop medication and to search for medical evaluation. The patient also complained of tenderness and contractures on thighs and calves (Figure 1). The patient referred dark urine by the third week, but urine was neither analyzed nor was myoglobinuria/pigmenturia documented.

After the fifth week of colchicine and allopurinol treatment, patient complained of tenderness and contractures on thighs and calves. On EMG analysis, complete absence of any activity is observed while the gastrocnemius muscle is in the contracted state.
Physical examination revealed proximal muscle weakness of limbs (4/5 on Research Medical Council grading System) and tenderness on thighs and calves. Sensory system, coordination, and reflexes were intact.
On biochemical evaluation, creatinine kinase (CK) increased five-folds from its normal value to 1101 U/L (normal: 30-200 U/L). Hepatic enzymes were on normal levels, alanine aminotransferase (ALT) was 33 U/L (normal: 0-55 U/L) and aspartate aminotransferase (AST) was slightly elevated, that is 37 U/L (normal: 5-34 U/L). On the contrary, uric acid decreased to 5.5 mg/dL (before treatment previous value was 9.4 mg/dL). Creatinine was under normal values (0.99 mg/dL) and no other biochemical or hematological values were abnormal (Table 1).
Evolution of biochemical variables after colchicine-allopurinol treatment seen in the case report.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
One week after withdrawal of medication, electromyographic (EMG) and electroneurographic (ENG) testing was performed. On ENG testing, sensitive and motor nerve conduction studies, late F-responses and H-reflex were normal (Table 2, Figure 2). However, after EMG evaluation of unilateral right biceps brachii and bilateral quadriceps, bilateral medial gastrocnemius and bilateral biceps femoris, increased insertional activity and spontaneous activity such as fibrillations, positive sharp waves, high-frequency myotonic discharges (Figure 3), and myopathic motor unit action potentials (MUAPs) of small amplitude and duration and increased polyphasic index were observed. On biceps brachii and quadriceps muscles, at maximal effort, an interferential pattern with reduced amplitudes was observed (Figure 4). Motor unit action potentials showed a decrease in mean duration (36% and 39% decreased for both quadriceps and 17% decreased for biceps brachii), and their polyphasic index was also increased. Some MUAPs showed satellite potentials (Figure 5). Those abnormalities are typical features of myopathic disorders. Normal motor and sensitive nerve conduction studies and late responses discarded neuropathy.
Values of motor and sensory nerve conduction studies and late F-responses and H-reflex are normal.

Abbreviations: LAT, latency; CMAP d, conduction motor action potential duration; CV, conduction velocity; F, F waves; LAT F, latency of F waves; SAP, sensory action potential; R, right; L, left; p, proximal; d, distal; p, negative peak; p-p, peak-peak.
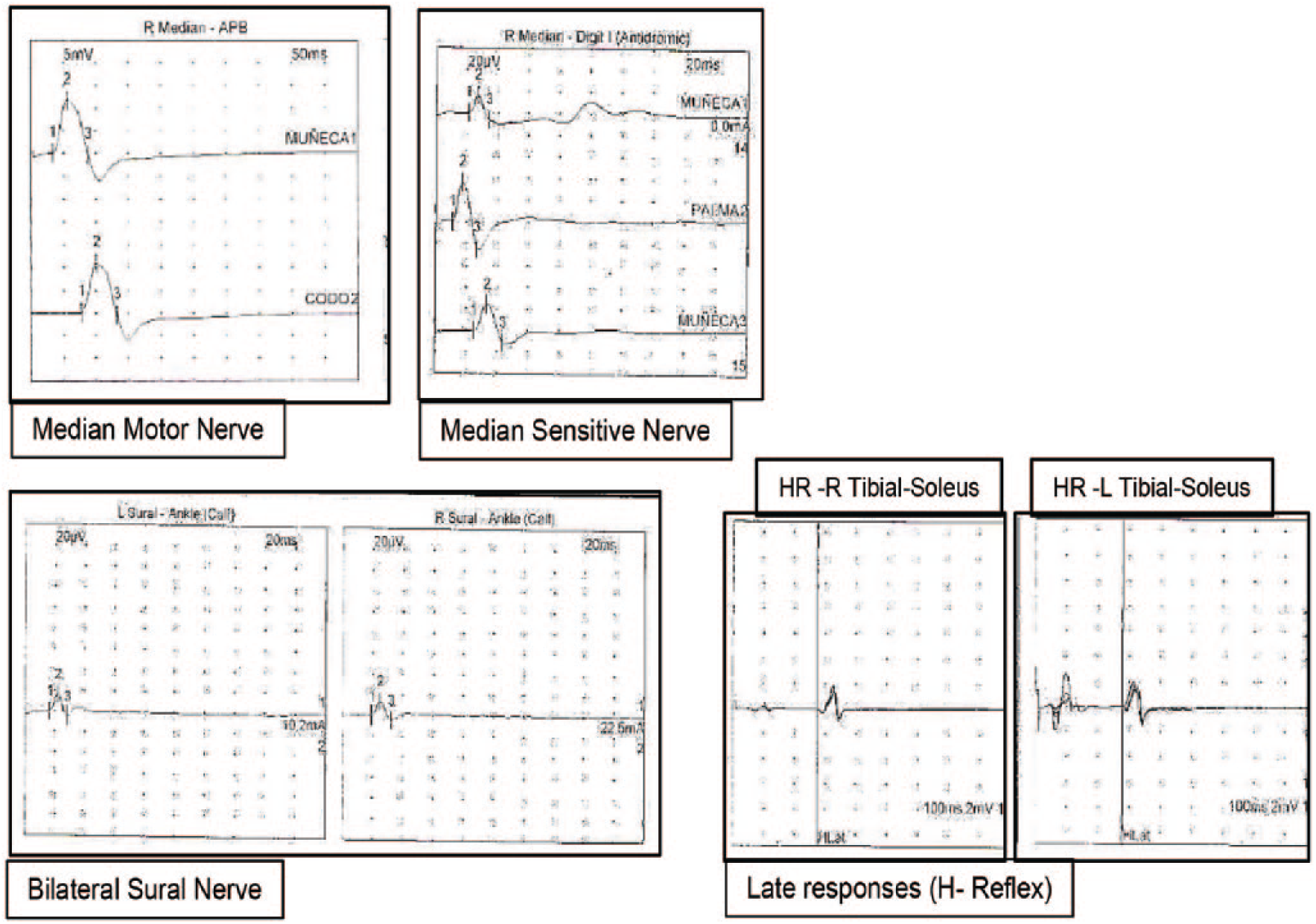
In our case report, on electroneurographic (ENG) study, motor and sensitive nerve conduction studies and late responses (H-reflex and f-waves) were all on normal values. Motor and sensitive median nerve, bilateral sural nerve and H-reflex are depicted as examples. These findings discard neuropathy.

Spontaneous activity such as (a) positive sharp waves, (b) fasciculations, and (c) myotonic discharges are observed on EMG testing.

Interferential recruitment pattern with reduced amplitudes was observed on biceps brachii and quadriceps muscles. Myopathic MUAPs are observed.

Myopathic Motor Unit Action Potentials (MUAPs) showed some satellite potentials, which are small MUAP separated from the main MUAP firing at a time locked and isoelectric interval (see blue arrows).
Based on previous clinical, biochemical, and neurophysiologic grounds, colchicine-induced rhabdomyolysis was diagnosed. Colchicine was thought to be the factor in the genesis of rhabdomyolysis because of the rapid onset of symptoms (6 weeks after allopurinol and colchicine treatment) and because of the lack of any alternative explanations for myotoxicity.
One week after colchicine was withdrawn, CK levels were reduced to 294 U/L and pain and general malaise ameliorated. Two weeks after colchicine withdrawal, CK levels reduced to 204 U/L (almost normal), and muscle power of lower limbs returned to normal, and patient was able to climb stairs normally. Discontinuation of colchicine resulted in resolution of clinical and biochemical features of rhabdomyolysis, confirming our hypothesis that colchicine induced this severe side effect.
Discussion
To the best of our knowledge, this case report presents the uncommon presentation of a case of colchicine-induced rhabdomyolysis in a patient without renal impairment. Myotoxicity is a well-recognized adverse effect of colchicine with 82 reported cases in the literature in the form of myoneuropathy and/or myotonia.6,14 On the contrary, rhabdomyolysis induced by colchicine is a very rare condition, with no more than six cases reported on the literature, and 58 cases registered on the Drug-Induced Rhabdomyolysis Atlas (DIRA). 15
Colchicine-induced neuromuscular toxicity and rhabdomyolysis have been reported with long-term treatment in therapeutic doses. Patients with renal dysfunction and elderly patients, even those with normal renal and hepatic function, are at increased risk. Concomitant use of statins, gemfibrozil, fenofibrate, fenofibric acid, or benzafibrate (all of them associated with myotoxicity) or cyclosporine may potentiate the development of myopathy. 15
Although patients with impaired renal function appear to be at higher risk for colchicine-induced neuromuscular adverse effects, 14 the drug can be myotoxic even in the presence of normal renal function 16 as in our present case report. This observation comes in line with the two previous case reports from Chattopadhyay et al 14 and Dawson. 17 Therefore, any patient on colchicine treatment, whatever the reason (rheumatic and nonrheumatic) should be monitored for neuromuscular adverse effects.
The most common adverse reactions reported from oral colchicine treatment are diarrhea (23%) and pharyngolaryngeal pain (3%), and these mild events usually resolve spontaneously,9–11 as it was in our case report. Diarrhea disappeared after 3 days of treatment without any dose reduction. The most severe adverse reaction of colchicine reported on literature was bone marrow suppression with agranulocytosis 18 and rhabdomyolysis after overuse. 9
The co-administration of additional drugs to colchicine constitute another risk factor that increases myotoxicity and rhabdomyolysis, either through inhibition of colchicine clearance (erythromycin or cyclosporine) or through their additional myotoxic effect (corticosteroids and statins).4,6,9 In the case of statins, they are metabolized by the cytochrome P450 (CYP)3A4 enzyme, in the same way as colchicine. These would explain the presence of rhabdomyolysis as a significant adverse drug interaction that has occurred in patients treated with colchicine and statins (simvastatin, 6 pravastatin, 19 fluvastatin, 20 and atorvastatin 21 ).
Colchicine myotoxicity is very variable according to age of patients and time of treatment. In a review of 75 cases with colchicine myotoxicity, Wilbur et al have stated that mean age of patients was 58 ± 16 years. Our case report was younger, 46 years of age. Duration of colchicine treatment ranged from 4 days to 11 years, with a mean of 40 ± 64 months. Cumulative daily dose of colchicine was 1.4 ± 0.9 mg/day. 6 In our case report, onset of symptoms appear after 3 to 6 weeks of treatment at a daily dose of 0.5 mg/day, between therapeutic margins. Although most patients in Wilbur’s study were receiving standard-dose therapy for long periods, colchicine toxicity becomes more apparent after recent changes in underlying disease state (solid organ transplantation, renal function impairment, or dialysis) or short-term use of increased doses. 6
The mechanisms of colchicine myoneuropathy are unknown. Colchicine affects the microtubullar cell function, and therefore, it may cause disruption of axonal transport and organelle trafficking in both nerve and muscle cells. Functional and structural changes in both axons and muscle fibers may be responsible of the clinical deficits. 4 Therefore, colchicine produces pathologic alterations on muscles and axonal neuropathy. 6 The neuropathic symptoms and signs reflect the presence of axonal atrophy and degeneration with loss of large myelinated axons and clusters of regenerating axons. 22 These changes account for the fibrillations, positive sharp waves and low-amplitude distal motor and sensory potentials found in electromyography examination. 23 If muscle biopsy is performed, vacuolar myopathy in the absence of necrosis is observed. 6
In clinical presentation, myopathic features predominate over neuropathic features. Patients typically complain of proximal muscle weakness but may have distal arreflexia and mild sensory change.6,22 Accompanying myotonia has also been reported on literature, 24 as it was observed in our case report. Contracture has been reported in metabolic myopathies. Contracture is the complete absence of any EMG activity in a muscle, while it is in the contracted state. On EMG, contracture is seen as completely electrically silent, 25 as it was in our case report. Active denervation usually is associated with neuropathic disorders. However, active denervation (fibrillation and positive sharp waves) also occur frequently in many myopathies, especially those associated with inflammation or necrosis, as it was in our case report. Denervation is believed to occur as a consequence of segmental inflammation or necrosis of muscle fibers, separating a distal, healthy portion of the muscle fiber from the part attached to the endplate. 25
Electromyographic findings in patients with colchicine myotoxicity show prominent fibrillations, positive sharp waves and excessively polyphasic motor action potentials in the proximal muscles that are brief and small in amplitude, but nerve conduction studies are normal.6,23 Electromyographic findings can also include low motor nerve response amplitudes if enough muscle fibers are lost.
As a resume, the neuromuscular toxicity of colchicine takes the form of subacute myopathy with features of mild distal polyneuropathy. 4 The weakness which may range from very mild to very severe, predominantly affects the proximal and axial muscles but also affect bulbar, facial, and even respiratory muscles.4,23 Our case report presented myopathic features of colchicine-induced neuromuscular myotoxicity. Normal motor and sensitive nerve conduction studies and normal late responses discarded neuropathy.
It is accepted that for colchicine toxicity, the prolonged duration of therapy is not as greater a risk factor as the total dose taken and the underlying disease state for developing myotoxicity. 6 Prophylactic dosages for colchicine range from 0.5 to 1 mg, one to four times a week, to 1 to 1.8 mg/day. 6 Patients with reported colchicine myotoxicity were receiving 0.6 mg/day to 0.8 mg eight times a day as needed. 6 Surprisingly, in our case report, prophylactic dose was 0.5 mg/day for 6 weeks; that is, the lowest dose recommended by rheumatologists. 12
In our case report, after medical history evaluation, clinical symptoms were related to recent usage of prophylactic colchicine for the management of hyperuricemia. Clinical, biochemical, and neurophysiologic signs are compatible with colchicine-induced rhabdomyolysis, in the absence of any other explanation for such acute abnormalities observed. Moreover, clinical and biochemical improvement was observed after cessation of colchicine medication. The importance of reporting adverse effects of colchicine usage is that there are only 82 cases of colchicine-induced myotoxicity, no more than six cases of colchicine-induced rhabdomyolysis (as reported by Del Favero, 26 Stefanidis et al, 27 Dawson and Starkebaum, 17 Chattopadhyay et al, 14 Montseny and Altmann 4 ) and only two cases of rhabdomyolysis and colchicine without renal impairment (as reported by Dawson and Starkebaum 17 and Chattopadhyay et al 14 ).
Conclusion
Colchicine-induced rhabdomyolysis is very rare with very few cases reported in the literature. Clinical picture reveals a history of proximal weakness and pain; laboratory findings indicate elevated CK due to muscle toxicity. Diagnosis is confirmed by clinical, biochemical, and EMG testing. Electromyographic testing reveals a myopathic disorder with spontaneous activity such as fibrillation potentials, positive sharp waves, high-frequency myotonic discharges, and MUAPs of small amplitude, small duration, and increased polyphasic index. Colchicine-induced rhabdomyolysis can occur even in the presence of normal renal function; therefore, any patient on colchicine treatment should be monitored for neuromuscular adverse effects to avoid such a rarely and severe complication.
Footnotes
Acknowledgements
Saturnino Diaz Trujillo is acknowledged for the article search.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors declare responsibility for the content of the manuscript sent having participated in the design and drafting of the Case Report and Review Article. M.E.-F.C. and G.-G.S.M. contributed to the drafting of the article. M.E.-F.C. wrote and translated the article. G.-G.S.M. performed the E.M.G. evaluation. B.-V.D.D. performed the clinical and biochemical evaluation. L.O.-C.B., P.M.-L.C., M.J-.A.F., R.A.L., and O.S.-P.M. reviewed and approved the article.
Ethical Approval
The case report was approved by the Ethical Committee of Hospital Universitario Santa Cristina.
Informed Consent
The patient has given written consent for the publication of photos and neurological and biochemical analysis.
