Abstract
Introduction:
Dermal fillers are used for multiple cosmetic indications including gluteal and thigh augmentation. Complications, although infrequent, are increasing due to the dramatic growth of dermal filler use. Our aim was to describe how the complication of infected silicone granulomas can present following lower limb augmentation.
Methods:
Two cases presented with pain, oedema, and erythema at the site of previous silicone filler injection, following a considerable delay after the last injection (range 4-7 years). We collected data on their biochemistry, haematology, histology, microbiology, and imaging at the time of presentation.
Results:
Complications included prolonged cellulitis with recurrent abscesses at sites of previous silicone dermal filler injection. Histology revealed infiltration of chronic inflammatory cells suggestive of silicone granuloma in both cases. Patients were reluctant to divulge use of cosmetic fillers or failed to recognise their significance given the time delay making diagnosis difficult. Delayed or recurring infections can suggest the presence of atypical organisms and we present the first reported case of silicone granuloma infection with Propionibacterium acnes.
Conclusions:
Microorganisms can induce immune-mediated hypersensitivity and are believed to be the trigger for delayed activation of a quiescent foreign body to a granulomatous reaction. The substantial time delay between injection and reaction must be recognised and may be attributable to atypical microorganisms or biofilm formation. Previous antibiotic use can affect expedient microbiological diagnosis and treatment requires close collaboration with microbiologists. It is important that clinicians are aware of these important complications which are becoming more common with increased use of filler augmentation.
Introduction
Over the past two decades, injections with filler agents for rhytid treatments and soft tissue or scar augmentation have undergone dynamic growth. According to statistics published by the American Society for Aesthetic Plastic Surgery, there were more than 1.2 million dermal filler injections in 2008, which represents a 200% increase since 1997. 1 They are increasingly attracting people who are eager to improve cosmesis without undergoing surgery. There are a huge variety of agents on the market, varying according to their biological half-lives and component materials. Agents that degrade within months include collagen (eg, Zyderm, Zyplast) and hyaluronic acid (eg, Restylane, Juvederm, Perlane). Agents that are resorbable within years include polylactic acid microspheres (eg, Sculptra, Newfill), calcium hydroxylapatite microspheres (eg, Radiance, Radiesse), and dextran microspheres (eg, Reviderm Intra). Permanent filler agents are not degraded by endogenous enzymes and include silicone, polymethyl methacrylate microspheres (eg, Artecoll), poly-hydroxy-ethyl-methacrylate (eg, Dermalive), and polyacrylamide (eg, Aquamid, Bio-Alcamid) and may have increased risk of chronic granulomatous reactions. 2
We report 2 cases of infective and granulomatous complications after injection of silicone dermal filler for lower limb scar correction, including the first report of Propionibacterium acnes infection.
Case Report 1
A 43-year-old woman was referred to our unit with left hip pain secondary to loosening of her left total hip arthroplasty (THR). She had a background of hepatitis C and sickle cell anaemia, which had resulted in bilateral hip avascular necrosis. She subsequently underwent primary right THR in 1994 with revision in 2001 and primary left THR in 2001. Due to loss of muscle bulk in both buttocks and indentations posterior to the scar, she had bilateral silicone filler injections in 2003 for buttock augmentation by a practitioner that visited her home. After 7 years, she developed spontaneous, gradual, bilateral buttock induration, oedema, and erythema, particularly evident on the right side (Figure 1), as well as left hip pain. Further history revealed silicone breast implants in 2010, and lip and cheek hyaluronic acid dermal filler augmentation in 2009.

Photograph of right thigh and buttock induration and oedema in case 1 from previous bilateral silicone filler augmentation.
Radiographs revealed loosening of the left THR components, with superior migration of the acetabulum (Figure 2). A bone scan demonstrated asymmetric, low-grade, increased tracer uptake around the left THR on both dynamic and early blood pool, with increased uptake around the acetabular component on the delayed study. There was subtle curvilinear soft tissue uptake on the early blood pool study of both hips, suggestive of a low-grade infection.
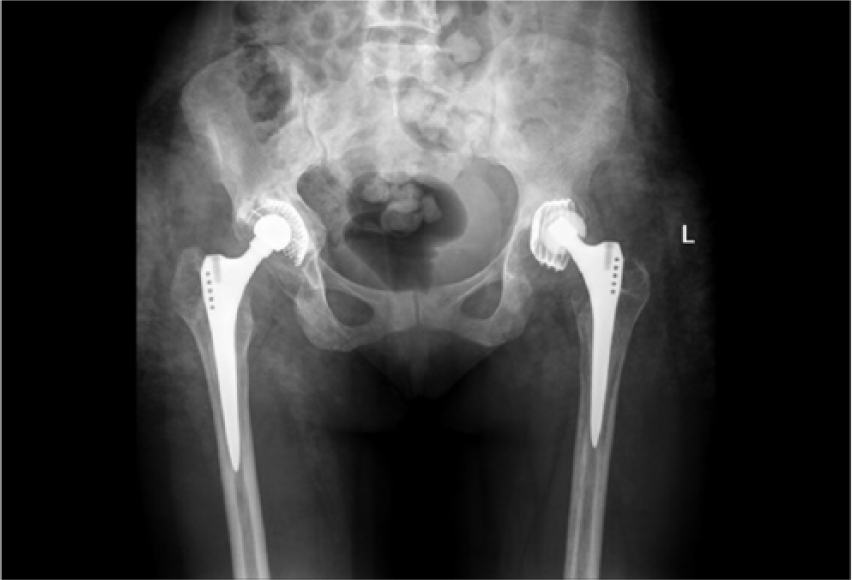
Anteroposterior radiographs demonstrating loosened left total hip arthroplasty components, with superior migration of the acetabulum in case 1.
Blood tests showed a normal C-reactive protein at 5 mg/dL (0-10 mg/dL), elevated white cell count at 13.7 (4.0-12.0 × 109/L), and a haemoglobin of 7.8 g/dL. Bilateral hip joint aspiration yielded negative cultures. Incisional skin biopsy of the right hip demonstrated deep dermal and subcutaneous infiltration of chronic inflammatory cells, mainly histiocytes, macrophages, and giant cells. Numerous empty vacuoles containing foreign material suggestive of silicone granuloma were found, but no eosinophils to indicate allergic reaction and no neutrophils to suggest infection.
The spacer was immersed in vancomycin and gentamicin prior to implantation and empirical Tazocin and Teicoplanin were started postoperatively pending cultures. The intraoperative specimens grew Propionibacterium acnes, sensitive to penicillin and ceftriaxone. The patient’s previous adverse reactions to ceftriaxone and clindamycin meant teicoplanin monotherapy continued for 6 weeks. At second-stage reimplantation, the cement contained vancomycin and gentamicin, and postoperative cover with amikacin, Tazocin, and teicoplanin for 3 days, with continuation with teicoplanin monotherapy. Seven specimens were sent at the second stage, and 2 grew 2 different strains of coagulase-negative Staphylococcus, not diagnostic of ongoing infection. However, teicoplanin was continued in view of the short duration of antimicrobial-free interval, wound dehiscence, and underlying haematoma that were washed out and reclosed (Figure 3). Samples from the washout grew Klebsiella, again of uncertain significance, and the patient was placed on oral co-amoxiclav treatment until the wound was dry. The patient then was discharged returning to lifelong penicillin prophylaxis in view of her sickle cell disease.
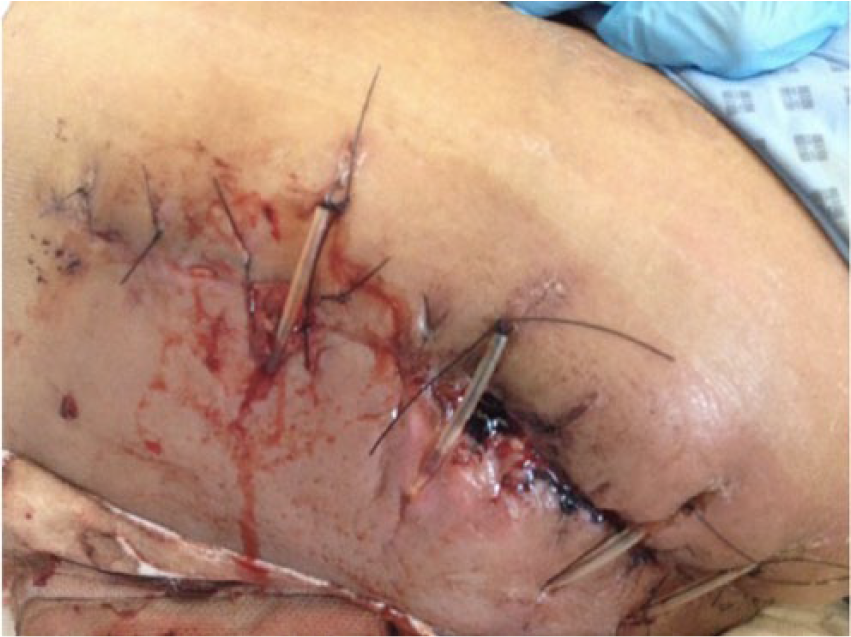
Photograph of left hip wound dehiscence in case 1 following 2-stage revision total hip arthroplasty.
Case Report 2
A 51-year-old woman presented with a 1-week history of worsening right lower leg pain, swelling, and erythema treated unsuccessfully with oral antibiotics in the community (Figure 4). She was involved in a road traffic accident at the age of 16 years, with severe soft tissue trauma of bilateral lower limbs and buttocks (Figures 4 and 5). She later received silicone soft tissue augmentation in buttocks and lower limbs to improve cosmetic appearance. After 4 years, she began experiencing bilateral lower limb swelling and pain secondary to subcutaneous silicone granuloma infection confirmed on biopsy, with recurrent cellulitis, abscesses, and ulcers. These required extensive antibiotic and surgical treatments. She had already had drainage of numerous abscesses, debridement, and split skin grafting of ulcers and maggot therapy when she presented to our unit.

Photograph of lower legs in case 2 demonstrating bilateral scarring and contour deformities from previous trauma, with right posterior leg swelling and erythema.

Photograph of right buttock in case 2 showing area of discolouration, scarring, and contour deformity from previous trauma.
At presentation, the right leg was indurated, erythematous, warm, but with no focal fluctuance (Figure 6). It was noted that the silicone was not encapsulated, but diffuse with widespread granuloma, precluding excision. On admission, she had a raised C-reactive protein at 155 mg/L (0-5 mg/L) but a normal white cell count. Magnetic resonance imaging showed a subcutaneous and intra-muscular foreign body material with no evidence of collection. She was managed for her infected silicone granuloma, with intravenous flucloxacillin and fusidic acid and improved clinically, but worsened in the community on oral flucloxacillin and subsequent doxycycline. She then developed an abscess requiring incision and drainage which grew Pseudomonas aeruginosa.

Photograph of right lower leg in case 2 to demonstrate extent of redness, swelling, and induration from previous silicone filler injections.
Discussion
Granuloma formation following injectable dermal filler has an incidence of between 0.02% and 1% and varies slightly between filler type, although all have been shown to have some propensity towards granuloma formation. 3 The time between injection and initial presentation with granuloma is usually between 6 and 24 months, although there have been reports of up to 10 years. 3 In both our cases, it appears that silicone dermal filler remained quiescent for 7 and 4 years, respectively, before becoming symptomatic and infected. The trigger for the activation of a quiescent foreign body to a granulomatous reaction is not known but has been suggested to be secondary to systemic infection 3 or immune hypersensitivity following local microbial growth.4,5 Microorganisms can induce immune-mediated diseases through protein-induced polyclonal activation of lymphocytes, bacterial-induced T–cell-stimulated cytokine upregulation, activation of costimulatory molecules, microbial encoding super antigens, and molecular mimicry. 5
Identifying the responsible microorganism in these cases is not always straightforward. We found that prolonged exposure to various antibiotics, often in the community, prior to operative samples as well as biofilm formation, yielded negative culture results and made targeting antibiotics difficult. Fluorescent in situ hybridisation analysis and Gram stain histologic diagnosis have sometimes been necessary in other reports. 6 Polymerase chain reaction analysis is less effective because structural genetic changes may hinder epitope targeting. 7 Histology plays the main role in establishing the presence of foreign body reactions, but macrophages and giant cells are usually present when there is no infection and so therefore cannot exclude an infective component.
Previous reported bacteria associated with silicone granulomas include Staphylococcus aureus, streptococci, and Pseudomonas aeruginosa, the latter of which was also isolated in our second case. 8 Local bacterial growth with bacteria that are not normally considered pathogenic has also been shown to trigger a granulomatous response. 6 The first case is the first documented infection of dermal filler by Propionibacterium acnes. This is a slow-growing, aerotolerant anaerobic Gram-positive rod, a known skin commensal usually present in deep pores and follicles. It is primarily known for its role in acne but is also a recognised opportunistic pathogen associated with device-related infections, 9 which may account for its presence in this case, especially given the intracutenous location of the filler material. Delayed infections of silicone granulomas can suggest the presence of atypical organisms, rather than the more common skin and soft tissue pathogens such as Staphylococcus aureus and streptococci, and previous reports include mycobacteria, Nocardia spp, and Sporothrix.10,11
Delayed infections and complications from dermal filler injection have also been attributed to biofilm formation which can respond to stimuli or become activated by bacteraemia and grow weeks, months, or even years after formation. 1 This may account for the chronic remitting and relapsing course of symptoms in both our patients whose infections were difficult to treat with antibiotics. Biofilms can interfere with phagocytosis and facilitate microbial resistance making infected granulomas very difficult to treat, particularly those formed on non-water-based fillers.1,5 This highlights the importance in preventing biofilm formation in the first instance through strict aseptic technique for injection, reducing the size of the needle and covering the puncture mark with a sterile dressing to lower risk for external bacterial entry.1,5
Silicone granulomas are difficult to treat. Previous reports describe treatment with pharmacologic agents, such as steroidal and non-steroidal anti-inflammatory drugs, localised steroid injections, tetracycline antibiotics, and immunomodulatory drugs (eg, etanercept and topical imiquimod).2,12–16 Surgical methods such as liposuction and laser therapy have also been reported. 16 Surgical excision should generally be avoided, as removal is usually incomplete due to the infiltration of finger-like projections into tissue. 17 In both our cases, the infected silicone granulomas in the soft tissue of the lower limb were managed using targeted antibiotic therapy, with surgical intervention restricted to complications such as abscesses or separate revision arthroplasty procedures. The first case, where the revision THR was complicated by wound infection and dehiscence, highlights the potential hazard of operating in a previously dermal-filled area which contains chronically inflamed soft tissue and biofilm with potential for bacteria re-activation.
Conclusions
The exponential increase in dermal filler injections for rhytides and soft tissue augmentation means that microbiologists, surgeons, physicians, and radiologists should be aware of the new diagnostic trap caused by granulomatous reactions and infections from inert material, injected intracutaneously. Identifying a specific microorganism is pivotal to be able to target antibiotics accordingly, which is the mainstay of treatment. However, we have yet to fully understand the pathogenesis of these complications, what triggers the delayed-onset granulomatous reactions, and what is the exact role of infection in the development of these complications. The substantial time delay between injection and reaction must also be recognised and may be attributable to atypical microorganisms or biofilm formation.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MW, NG and SH conceived the study and analyzed the data. ZMJ, MW and EZ analyzed the data and wrote the manuscript.
