Abstract
Research has shown early and sustained relief with a combination therapy of a corticosteroid (CS) and hyaluronic acid (HA) in knee osteoarthritis (OA) patients. This can be administered via a single injection containing both products or as separate injections. The former may be more expensive when considering only product cost, but the latter incurs the additional costs and time of a second procedure. The purpose of this study was to compare the cost-utility of the single injection with the 2-injection regimen. The results of this analysis revealed that the single-injection formulation of a CS and HA may be cost-effective, assuming a willingness-to-pay of $50 000 per quality-adjusted life year gained, for symptomatic relief of OA symptoms. This treatment may also be more desirable to patients who find injections to be inconvenient or unpleasant.
Introduction
Osteoarthritis (OA) of the knee is one of the most prevalent conditions in the world.1–6 The number of people diagnosed with knee OA is increasing due to the ageing population and other associated risk factors, such as obesity, reduced physical activity, and a greater rate of knee injuries among young, active individuals.1,4,7 By the year 2020, OA is expected to be one of the top 10 leading causes of disability-adjusted life years (DALYs) in developed countries. 4 Knee OA also has a substantial economic impact on both the health care system and societal costs.1,4–7 Not only is therapeutic intervention expensive, especially when surgery is required, but the disease also leads to productivity loss due to a patient’s inability to work.4–6 As the prevalence of OA rises, the financial burden associated with this condition will also continue to increase,2,5,7 highlighting the importance of effective, yet affordable, treatments.
Intra-articular injections are typically used to treat knee OA after more conservative methods fail, and available options include corticosteroid (CS), hyaluronic acid (HA), platelet-rich plasma, and mesenchymal stem cell therapy.8–18 The latter 2 methods are more recent developments, and the evidence on their efficacy in this patient population is limited18–22; however, there are numerous published research articles on the efficacy of CS and HA injections for treating knee OA. The evidence suggest that CS injections are beneficial in the short term (ie, up to 4 weeks post injection) but show little to no effect beyond this time point.17,23–27 Additional studies have demonstrated the therapeutic value of HA injections from 3 to 6 months post injection.17,23,27–34
The earlier symptomatic relief attributed to CS and more long-term improvements following HA injections were the rationale for a number of randomized trials that have evaluated CS plus HA versus HA alone. These studies have demonstrated more rapid pain reductions when a CS is added to viscosupplementation with HA, with no difference between treatment groups beyond 4 weeks.3,35–38 This form of treatment may be administered via a single injection containing a combined CS-HA formulation35,38 or sequential injections of each product3,36,37 – the former potentially being more expensive when looking strictly at the product cost, but the latter incurring the additional costs and inconvenience associated with an additional visit and injection procedure. Therefore, the purpose of this study was to compare the cost-utility of the single-injection combined CS-HA formulation with a 2-injection regimen consisting of sequential CS and HA injections.
Methods
Treatment utility scores
The Western Ontario and McMaster Universities Arthritis Index (WOMAC) scores were obtained from the intention-to-treat analysis population of the Cingal 13-01 trial (NCT01891396), a 3-arm, multi-centred, double-blind clinical trial that randomized knee OA patients to Cingal (n = 149), Monovisc (n = 150), or saline (n = 69) (see Supplementary Data for additional details regarding the trial). Cingal is a single-injection cross-linked sodium hyaluronate (Monovisc, 88 mg) combined with a corticosteroid (triamcinolone hexacetonide, 18 mg), supplied as a 4-mL unit dose in a 5-mL syringe. 35 Monovisc is a single-injection cross-linked hyaluronic acid injection (molecular weight: 1000-2900 kDa) indicated for the treatment of pain in knee OA. 39 Table 1 presents the baseline characteristics of the patients included in this trial. In short, the inclusion criteria of this study were patients diagnosed with Kellgren-Lawrence grade I to III knee OA, aged 40 to 75 years, with a body mass index ≤40 kg/m2 and a baseline WOMAC pain score between ≥40 mm and ≤90 mm on a 100-mm scale. 35 As WOMAC scores in this trial were measured on a 100-mm visual analogue scale, these values were converted to the WOMAC Likert scale to estimate Health Utilities Index Mark 3 scores using the methods proposed by Grootendorst et al. 40 Utility scores range from 0.0 to 1.0, which represent the health states ranging from death to perfect health. 41 For the purpose of this analysis, the utility scores of the Cingal arm represented the single-injection (combined CS-HA formulation) regimen group and those of the Monovisc arm represented the 2-injection (sequential CS and HA injections) regimen group. This decision was made based on the notion that CS provides little to no benefit beyond 3 to 4 weeks post injection and treatment effects in this group at 6 months would mainly be due to viscosupplementation with HA.3,17,23–27,35–38 There were also no statistically significant differences in WOMAC subscales between the 2 treatments arms (Cingal vs Monovisc) at baseline and at the final follow-up visit at 6 months, suggesting a limited or no synergistic effect when combining CS and HA and that any difference in utility scores between the 2 groups at 6 months would be minimal. 35 It was also assumed that any one of 3 single-injection HA products (Durolane, Monovisc, Synvisc-One) could be used in the 2-injection regimen and that treatment efficacy was similar across all 3 brands as they are all cross-linked HA preparations. 32 The utility scores were calculated at baseline and at 6 months post injection to determine the utilities gained over this time frame.
Baseline characteristics of patients in the Cingal 13-01 trial.

Abbreviation: WOMAC, Western Ontario and McMaster Universities Arthritis Index.
Treatment cost data
A single payer perspective was used with respect to costs. Cost information was obtained from the Ontario Schedule of Benefits and Québec List of Medications; wholesale product costs of the individual injections were received; and data were also retrieved from 2 previously published economic studies conducted by Losina et al 1 and Bellamy et al.7,42–44 Values in US dollars were converted to Canadian dollar (Can$) and priced in 2016 Can$ using the methods provided by the Bank of Canada and US Bureau of Labor Statistics.45–47 The total treatment cost per patient included the product (at wholesale value), initial consult visit, injection procedure, and additional conventional care (ie, non-steroidal anti-inflammatory drugs, acetaminophen, physical therapy, and assistive devices) for 6 months. The cost of Cingal was used for the combined CS-HA formulation. 44 Three different single-injection HA products (Durolane, Monovisc, Synvisc-One) were considered, 44 and an average cost across the 3 brands was calculated for the primary analysis. A range of different costs was found for CS injections,7,43 and the midpoint value of this range was used in the primary analysis. The costs of a single CS injection and a single HA injection were added together to calculate the total product cost for the 2-injection group. Losina et al 1 determined the cost of conventional care over a 1-year period, which was adjusted accordingly to account for only 6 months of treatment. Table 2 provides a summary of the cost estimates. The total cost of each treatment over 6 months was calculated by the following formula:
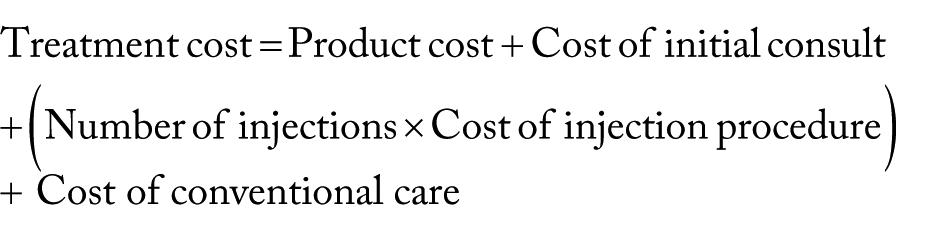
Cost data.

Abbreviations: Can$, Canadian dollar; CS, corticosteroid; HA, hyaluronic acid.
Cost-utility analysis
Cost-utility was evaluated using the cost per quality-adjusted life year (QALY) gained metric. The QALY is a health outcome that simultaneously captures, in a single measure, both changes in the quantity of life (mortality) and changes in the quality of life (morbidity). 41 To determine the number of QALYs gained by a treatment, the average baseline utility score was subtracted from the average 6-month post-treatment utility score.
Incremental cost-utility ratios (ICURs) were used to compare the 2 treatment regimens. The ICUR was calculated using the formula (CB − CA)/(Q B − Q A), where CB and CA represented the cost of treatment B and treatment A, respectively, and Q B and Q A represented the QALYs gained for treatment B and treatment A, respectively. A treatment was considered cost-effective if it fell below the willingness-to-pay (WTP) threshold, which ranged from $50 000/QALY to $150 000/QALY in the published literature, and is dominant if it is both more efficacious and less costly compared with the alternative.48,49 A WTP threshold of $50 000/QALY was chosen as this is the most conservative value. Data extraction and analysis were completed using Microsoft Excel (Version 2010; Microsoft, Redmond, WA, USA).
Sensitivity analyses
Sensitivity analyses were performed to determine the robustness of the results. The ICUR was re-calculated under different cost scenarios for the CS and HA injections, after increasing the overall cost of the single-injection (combined CS-HA formulation) regimen by 20%, decreasing the utilities gained by the single-injection regimen by 20%, and increasing both the overall cost and utilities gained in the single-injection group by 20%.
Results
Utility scores
The utility score of patients who received the single-injection (combined CS-HA formulation) regimen improved by 0.261 QALYs, increasing from 0.501 QALYs at baseline to 0.762 QALYs at 6 months (Table 3). In the 2-injection (sequential CS and HA injections) group, utility scores improved from 0.486 QALYs at baseline to 0.740 QALYs at 6 months post treatment, a difference of 0.254 QALYs (Table 3).
Utility scores.

Cost-utility
The ICUR from the primary analysis (base case) demonstrated that the single-injection regimen (combined CS-HA formulation) dominated the 2-injection (sequential CS and HA injections) regimen as the single-injection treatment was both less costly and more efficacious (Table 4; Figure 1). The difference in utilities gained in the single-injection group relative to the 2-injection group was an additional 0.007 QALYs, and the single injection was also $47.08 cheaper.
Incremental cost-utility ratio of the single-injection (combined CS-HA formulation) regimen vs the 2-injection regimen (sequential CS and HA injections) – base case.
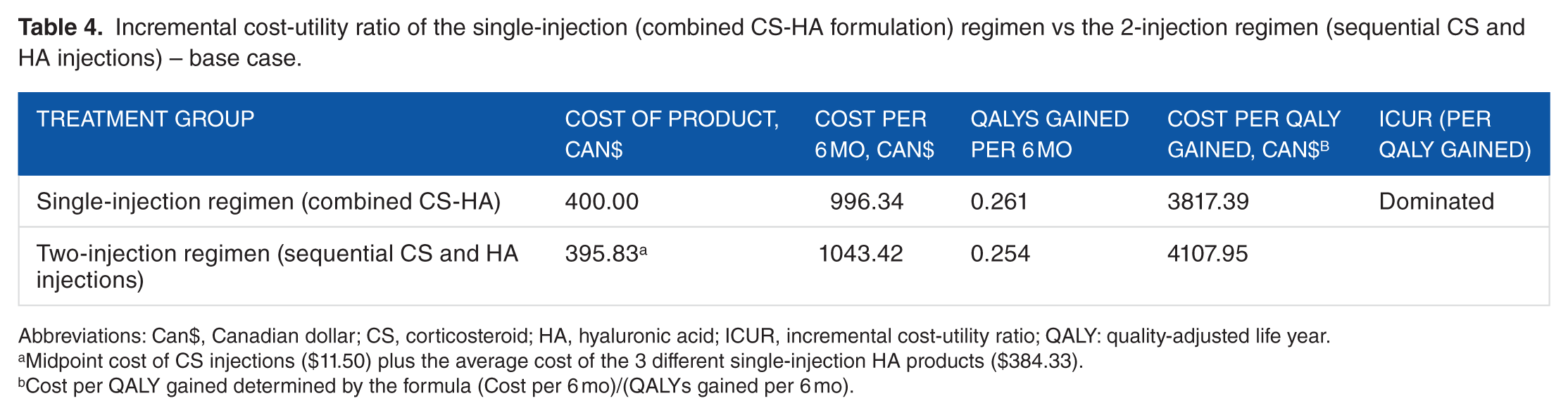
Abbreviations: Can$, Canadian dollar; CS, corticosteroid; HA, hyaluronic acid; ICUR, incremental cost-utility ratio; QALY: quality-adjusted life year.
Midpoint cost of CS injections ($11.50) plus the average cost of the 3 different single-injection HA products ($384.33).
Cost per QALY gained determined by the formula (Cost per 6 mo)/(QALYs gained per 6 mo).

Incremental cost-utility ratios (ICURs) for all models at 6 months. Negative values indicate that the single-injection regimen was both less costly and more efficacious than the 2-injection regimen.
Sensitivity analyses
The results of the sensitivity analyses (Table 5; Figure 1) ranged from the single-injection regimen still dominating the 2-injection regimen (when the lowest reported cost of the individual CS injection was used – sensitivity analysis 1) to $21 741.14/QALY gained (when the total cost of the single-injection regimen was increased by 20% – sensitivity analysis 5). The single-injection regimen remained below the WTP threshold of $50 000/QALY gained in all scenarios when compared with the 2-injection method.
Results of the sensitivity analyses.

Abbreviations: Can$: Canadian dollar, CS: corticosteroid, HA: hyaluronic acid, ICUR: incremental cost-utility ratio, QALY: quality-adjusted life year.
Cost per QALY gained determined by the formula (Cost per 6 mo)/(QALYs gained per 6 mo).
Values adjusted for the given sensitivity analysis relative to the primary analysis.
Discussion
The purpose of this study was to determine how the cost-utility of a single-injection combined CS-HA formulation compared with that of a 2-injection (sequential CS and HA injections) regimen for the treatment of knee OA over a 6-month period. An ICUR was calculated between the 2 treatment methods, under a number of assumptions, using the data from a previously conducted randomized clinical trial, and the robustness of the results was tested with a variety of sensitivity analyses regarding costs and efficacy.
The results of the primary analysis showed that the single-injection regimen dominated the 2-injection treatment method, as the former was less costly yet also resulted in more utilities gained after 6 months; however, clinical trials evaluating comparisons between the 2 treatments directly are limited, and this outcome was based on particular assumptions of the included data. If it was assumed that both interventions would result in identical utility scores at 6 months post injection, a simple cost analysis would reveal that the single-injection formulation is approximately $50 cheaper than the 2-injection method. Several different sensitivity analyses also demonstrated the cost-utility of the single-injection regimen over the 2-injection method, using a WTP threshold of $50 000/QALY gained; the largest ICUR was $21 741.14/QALY gained, which was the result when the total cost of the single-injection regimen was increased by 20% (sensitivity analysis 5 in Table 5). In particular, under 2 alternate scenarios where greater differences between utility scores were proposed (sensitivity analyses 6 and 7 in Table 5), the single-injection regimen was still considered cost-effective even if its utility score was 20% lower (ICUR = $1046.22/QALY) or if it was both 20% more expensive and 20% more efficacious (ICUR = $2579.46/QALY).
Finding cost-effective methods of treating knee OA should be a primary concern among health professionals due to its increasing prevalence and burden on the health care system.1,50,51 It has been established through previous research that a combination treatment of both CS and HA injections should be considered a more favourable option than either treatment alone to obtain early and sustained symptomatic relief.3,35–38 The current analysis suggested that a single-injection combined CS-HA formulation, such as Cingal, is cost-effective compared with a 2-injection regimen consisting of sequential CS and HA injections, as the difference in product costs between the 2 methods may be mitigated by the latter’s requirement for a second injection procedure. A 2-injection treatment regimen may also be less desirable to patients simply because of the treatment delay or time and discomfort associated with the injection procedure itself. Although the results of this analysis are not definitive and should be interpreted with caution, they provide rationale to investigate this topic further in future studies.
Through sensitivity analyses, the biggest driver of cost differences between the 2 treatments was attributed to which HA product was used in the 2-injection regimen. There is considerable variability in the costs of the 3 HA brands, with Monovisc priced at $320 per injection, Durolane at $409 per injection, and Synvisc-One at $424 per injection, the last 2 being even more expensive than Cingal ($400 per injection). 44 It is also important to note that the price of one of these products would vary from region to region.
This study has a number of limitations. The data for the analysis were acquired from just 1 clinical trial. Additional evidence of comparable experiments is needed, which would then be combined in a meta-analysis to ensure more accurate and valid estimates of treatment efficacy of the 2 treatment groups. The patient population of this trial included patients diagnosed with knee OA severity ranging from Kellgren-Lawrence grades I to III. Treatment efficacy and costs may vary depending on baseline OA severity and it would be worthwhile to examine these potential differences in future studies. Also, it was assumed that the utilities gained in the Monovisc arm of the Cingal 13-01 trial was representative of a treatment group that received both a CS and HA intra-articularly as sequential injections. The rationale for this assumption was that CS injections have no benefit beyond 3 to 4 weeks post injection and effects observed at 6 months may only be attributed to viscosupplementation with HA.3,17,23–27,35–38 There were also no statistically significant differences at baseline and at final follow-up in WOMAC subscales between the 2 treatments arms (Cingal vs Monovisc), suggesting no synergistic effect when combining CS and HA and that any difference in utility scores between the 2 groups at 6 months would be minimal. 35 This was done to ensure comparable patient populations between groups; however, additional research is required that directly compares a single-injection combined CS-HA formulation with a 2-injection CS and HA treatment regimen, within a homogeneous sample, to obtain more accurate utility scores. It was also assumed that patients in both groups received the same amount of conventional care. Patients receiving such treatments may not require all aspects of conventional care or it may differ between groups. The wholesale prices of the injections were used in our analyses, but retail costs of these products can differ for various reasons, such as location and method of acquiring the product. There may also be differences in the additional costs related to adverse effects or use of other treatments between groups. The economic impact of a patient’s time off work, travel costs, and productivity loss during the 6 months was also not assessed, which would likely have a considerable effect on the total cost of the 2-injection regimen as it requires an additional visit. All of these considerations would have an impact on the ICURs. Future randomized clinical trials on the topic should also collect patient-level economic data, including indirect costs, during the study to acquire more accurate estimates of the overall monetary value of care. Another assumption was that all single-injection HA products had the same treatment effect as Monovisc, which is why the average cost of the 3 different brands was used in the primary analysis. Although Durolane, Monovisc, and Synvisc-One are all chemically cross-linked products, Monovisc is categorized as a moderate-molecular-weight HA (>1500 to <3000 kDa), whereas the other 2 are classified as high-molecular-weight (≥3000 kDa) products, and there is evidence that suggests that molecular weight influences clinical outcomes.28,39,52 Therefore, even though both Durolane and Synvisc-One are more costly than the single-injection combined CS-HA formulation of Cingal, their efficacy at 6 months may very well be different than that of Monovisc. This would result in different values for utilities gained after treatment and different ICURs if either Durolane or Synvisc-One was used instead of Monovisc. This was explored with a sensitivity analysis where the cost of Monovisc was used in the calculation (sensitivity analysis 2 in Table 5). The result provided evidence that Cingal is cost-effective compared with a treatment group receiving a 2-injection regimen of a CS and Monovisc; however, it cannot also be generalized that Cingal is definitely cost-effective relative to a comparison group that receives a 2-injection treatment with a CS and viscosupplementation with either Durolane or Synvisc-One. This study is strengthened by the sensitivity analyses that were conducted, which confirmed the robustness of the results. These results showed no variability in cost-utility of the single-injection combined CS-HA regimen, assuming a WTP threshold of $50 000/QALY gained. Also, the data used in the analysis were obtained from a randomized clinical trial, which ensured comparable patient populations and similar distributions of prognostic factors between groups at baseline. 53
Conclusions
In conclusion, a single injection containing a combined CS-HA formulation can be a cost-effective method for early and sustained symptomatic relief in the treatment of knee OA compared with a 2-injection regimen of sequential CS and HA injections (Monovisc). A single-injection regimen may also be a more desirable option for patients who find the injection procedure itself to be inconvenient or displeasing. Additional sensitivity analyses surrounding the cost and efficacy data confirmed the robustness of the results, as ICURs did not exceed the WTP threshold of $50 000/QALY gained. Additional clinical trials providing direct comparisons between the 2 treatment options, with collection of patient-level cost data, are required to validate these findings.
Footnotes
Author Contributions
ELB, RTD, MB, ML, and RM conceived and designed the experiments. CV analysed the data. CV wrote the first draft of the manuscript. ELB, RTD, CV, MB, ML, and RM contributed to the writing of the manuscript, agree with manuscript results and conclusions, jointly developed the structure and arguments for the paper, and made critical revisions and approved the final version. All authors reviewed and approved of the final manuscript.
Peer review:
Four peer reviewers contributed to the peer review report. Reviewers’ reports totalled 1856 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a research grant from Pendopharm.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Etienne L. Belzile discloses speaking for Pendopharm, consulting for Zimmer Biomet and Bodycad, and teaching for Smith & Nephew and Stryker. Dr Robert Deakon discloses speaking for Pendopharm, consulting for Anika Therapeutics, and teaching for Arthrex. Dr Mohit Bhandari is a consultant for Pendopharm, Sanofi, Amgen, DJO, and Ferring. Dr Martin Lamontagne is a consultant for Pendopharm. Dr Robert McCormack has received an honorarium as an educational consultant for Pendopharm, Sanofi, and Bioventus.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
