Abstract
Background:
Tumour heterogeneity is considered an important mechanism of treatment failure. Imaging-based assessment of tumour heterogeneity is showing promise but the relationship between these mathematically derived measures and accepted ‘gold standards’ of tumour biology such as immunohistochemical measures is not established.
Methods:
A total of 20 women with primary breast cancer underwent a research dynamic contrast-enhanced computed tomography prior to treatment with data being available for 15 of these. Texture analysis was performed of the primary tumours to extract 13 locoregional and global parameters. Immunohistochemical analysis associations were assessed by the Spearman rank correlation.
Results:
Hypoxia-inducible factor-1α was correlated with first-order kurtosis (r = −0.533, P = .041) and higher order neighbourhood grey-tone difference matrix coarseness (r = 0.54, P = .038). Vascular maturity-related smooth muscle actin was correlated with higher order grey-level run-length long-run emphasis (r = −0.52, P = .047), fractal dimension (r = 0.613, P = .015), and lacunarity (r = −0.634, P = .011). Micro-vessel density, reflecting angiogenesis, was also associated with lacunarity (r = 0.547, P = .035).
Conclusions:
The associations suggest a biological basis for these image-based heterogeneity features and support the use of imaging, already part of standard care, for assessing intratumoural heterogeneity.
Introduction
It has been an aspiration that increasing knowledge of the biological mechanisms that underpin cancer development and progression will lead to improvements in individualized therapy for cancers. The characterization of cancers is normally performed via a biopsy, either from a primary or metastatic lesion. There is increasing awareness that these small samples may not be representative of the whole tumour, or of other metastases, in the same patient. 1 Tumoural heterogeneity may be responsible for treatment failures where therapy is directed against a specific molecular characteristic. 2 Previous work examining multiple tumour samples from the same patient has identified significant intratumoural genomic heterogeneity between geographic regions in the same tumour (spatial heterogeneity) as well as between the primary tumour and the local or distant disease (intertumoural heterogeneity).3,4 Established receptor status such as oestrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER-2) have been shown to have considerable discordance in the same primary tumours, between primary and secondary tumours and also over time. 5 In addition, distinct populations of subclones can also vary throughout the course of the disease due to disease progression or metastasis or as a result of outside influences such as drug therapy (temporal heterogeneity).6,7
There is increasing awareness that non-invasive imaging may have a role in assessing tumour heterogeneity. 8 Imaging is already used widely in the management of a cancer patient including diagnosis, staging, assessment of treatment response, and surveillance. Computed tomography (CT) is well placed for tumour heterogeneity assessment as it is routinely used in clinical practice as the standard of care for cancer staging and response assessment and thus is cost effective. Computed tomography images are of high resolution (typically 0.7 mm × 0.7 mm in plane pixel size) allowing the spatial heterogeneity to be quantified by image analysis that provides both global (whole tumour) or locoregional (inter-pixel) measure of the distribution and spatial characteristics of pixel signal intensity within the tumour using statistical (first, second, or higher order) or model-based mathematical methods. 9 It is clear from visual inspection of medical images, for example, CT, magnetic resonance imaging (MRI), and positron emission tomography (PET), that there are phenotypical spatial (and temporal) differences in hounsfield units, signal intensity, or tracer uptake – so-called ‘imaging heterogeneity’. In the last few years, there has been increasing number of studies supporting the hypothesis that the phenotypic patterns on imaging reflect biological heterogeneity, for example, differences in angiogenesis, hypoxia, or gene expression. An increasing number of studies have also confirmed that imaging measures have an impact on outcome, for example, survival, relapse, or response to therapy. How these heterogeneity parameters relate to key biological tumour processes (angiogenesis, hypoxia, and proliferation) is an area of intense research interest.
A total of 4 key studies in renal cell cancer, non-small cell lung cancer, colorectal cancer, and oesophageal cancer have demonstrated the associations between angiogenic factors, hypoxic markers and proliferation and PET, PET/MRI, MRI and CT parameters.10–13 Radiogenomics, the use of quantitative data from imaging combined with clinical and patient-centred data, has been recognized as playing a potentially central role in the future for a personalized medical approach in breast cancer. 14 A number of studies have looked at a wide range of molecular data such as oestrogen status, HER-2, 15 gene expression analysis 16 and multigene assays such as Oncotype DX 17 and proliferation markers.18–20 These studies have looked at the relationships with a number of imaging techniques including dynamic contrast-enhanced magnetic resonance imaging (DCE-MR) 21 and 18fluorodeoxyglucose positron emission tomography (FDG-PET)22,23 and found a number of correlations that have the potential to be incorporated into future prospective studies for confirmation. Another specific tool under investigation has been the use of non-invasive imaging techniques such as PET using radiolabeled antibodies and in particular HER-2 PET/CT to help predict patient benefit from HER-2-directed therapies.24–30
Angiogenesis plays a driving role in cancer development and progression31,32 and has been defined as one of the hallmarks of cancer. 33 Data from breast cancer show that angiogenesis in the primary cancer can predict for disease-free and overall survival.34–40 Vascular endothelial growth factor (VEGF) is a vital member of a group of cytokines that play a critical role in angiogenesis. Of particular note, it increases microvascular permeability, induces endothelial cell migration, division, and survival as well as inducing angiogenesis through a variety of mechanisms.41,42
Hypoxia-inducible factor-1α (Hif-1α) is also a key transcription factor, induced by hypoxia, that can stimulate angiogenic factors including VEGF, proliferation, and cell-adhesion genes. 43 Hypoxia also activates carbonic anhydrase IX (CAIX) activity which is thought to reduce the pH of tumours thus increasing their growth and survival ability43,44; it is a marker of poor prognosis in breast cancer. 45 Glucose transporter-1 (GLUT-1), also regulated by Hif-1α, mediates cellular glucose uptake, which is an important part of cell proliferation and is required for tumour cell maintenance and growth. 46 It is overexpressed in many tumours and associated with poorer survival. Ki-67 provides a measure of cell proliferation and is a prognostic marker in breast cancer.47,48
Our hypothesis is that image-based heterogeneity is associated with hypoxia, angiogenesis, and proliferation. This prospective study aimed to examine the relationship between contrast-enhanced CT parameters and the accepted gold standard immunohistochemical (IHC) biological markers of angiogenesis, hypoxia, and proliferation in breast cancer.
Methods
Patients
A total of 20 women with histologically proven primary breast tumours were recruited from January 2009 to August 2010 and underwent a DCE-CT prior to treatment with neoadjuvant chemotherapy (NAC). Following NAC, patients then went on to receive surgery as standard of care, either mastectomy or breast-conserving surgery with an appropriate axillary procedure (usually axillary nodal dissection). Institutional review board approval was obtained for this study (08/H0311/147) and all patients gave written consent to participate in this substudy of a previously published study. 49
Computed tomography acquisition and analysis
Dynamic contrast-enhanced CT (SOMATOM Definition, Siemens Healthcare, Forchheim, Germany) was performed following intravenous iodinated contrast agent (50 mL ioversol 350 mg/mL iodine at 6 mL/s; Optiray; Covidien, Mansfield, MA, USA) using a 4-dimensional (4D) adaptive spiral technique (80 kV, 100-120 mAs, 0.33 seconds rotation time, matrix 512 × 512, scan field of view 300-450 mm, and reconstructed slice thickness 3 mm with 2 mm increment). Heterogeneity analysis was performed on the final enhancement phase axial images, 50-second post-intravenous contrast injection, using in-house software implemented with MATLAB (MathWorks, Natick, MA, USA). Following application of a smoothing filter, a volume of interest (VOI) was created for the entire primary tumour by outlining the whole tumour on all axial images where visible by 2 observers in consensus (with 1 and >15 years of CT experience). Global texture features were calculated for the whole tumour volume using (1) first-order statistical histogram methods (mean, skewness, kurtosis, energy, and entropy) and (2) model-based fractal analysis (fractal dimension and lacunarity). Locoregional texture features, examining the individual inter-pixel relationships, were calculated using (1) second-order statistical (grey-level co-occurrence matrix [GLCM]: energy) and (2) higher order statistical methods (neighbourhood grey tone-difference matrix [NGTDM]: coarseness, contrast, busyness; grey-level run length [GLRL]: long run emphasis; grey-level size zone matrix [GLSZM]: long zone emphasis) (see Supplementary Material for parameter definitions and equations). These were selected from their usage in previous publications and likely clinical relevance.9,50
Immunohistochemistry
All IHC procedures were performed on the core biopsies obtained for diagnosis; the formalin-fixed paraffin-embedded (FFPE) sections were de-waxed with xylene and brought to the aqueous phase with decreasing concentrations of industrialized methylated spirits. The 3,3′-diaminobenzidine (DAB) chromogen was used to provide the brown-coloured end-product staining visualized under a bright-field microscope. The negative controls comprised the known positive control tissue incubated with diluent under the same conditions at the same time as the test samples. The pretreatment methods are described as below.
Ki-67
Antigen retrieval was carried out by microwaving the sections in pH 6 citrate buffer. Using the Dako Autostainer (Dako Ltd., Cambs, UK) with REAL Kit, the sections were incubated with monoclonal mouse anti-human Ki-67 antigen clone MIB-1 (Dako) at 1/50 dilution for 20 minutes. Scoring of at least 500 invasive breast cancer cell nuclei in 5 high-power fields (HPFs) at ×400 magnification was carried out to obtain a percentage positive score. If 500 cells were not available in 5 HPFs, more fields were scored to achieve this. The control used was a previously validated Ki-67 positive tissue microarray.
Smooth muscle actin
No pretreatment antigen retrieval was needed. Avidin/biotin block was applied prior to application of anti-smooth muscle actin (SMA) monoclonal mouse antibody clone 1A4 at a dilution of 1/300 and using the Dako REAL Kit. A ductal carcinoma in situ (DCIS) breast cancer known to be positive for SMA was used as the control. The number of large vessels stained brown/HPF were recorded.
Carbonic anhydrase IX
No pretreatment was required. A serum-free protein block (Dako) was applied prior to incubating with anti-CAIX polyclonal antibody clone M75 (Abcam, Cambridge, UK) at 1/1000 dilution for 1 hour using the Dako EnVision FLEX+ Kit on a Dako Autostainer. Stomach tissue known to be positive for CAIX was used for the control. Scoring was by a modified H score which estimated by eye the percentage cytoplasmic positive invasive tumour and 0, 1+, 2+, and 3+ staining intensity as an average calculated from the 10 HPFs assessed.
CD34 (micro-vessel density)
No pretreatment was required. Monoclonal mouse anti-CD34 (Class II Clone QBend 10; Dako) ready-to-use undiluted was used with the Dako EnVision FLEX+ Kit. Placenta tissue known to be positive for this biomarker was used as the control. The number of vessels staining positive per HPF was estimated by eye and the average taken from 10 HPFs was the resultant score.
Glucose transporter-1
Pretreatment of the sections was carried out for 30 minutes in a water bath at 97°C in low pH Flex antigen retrieval solution. Sections were incubated for 20 minutes with 1/600 anti-human GLUT-1 mouse monoclonal antibody (Abcam SPM498). Dako EnVision FLEX+ Kit was used and liver was the control tissue. Percentage membrane positive staining per HPF was estimated from an average of 10 HPFs assessed.
Vascular endothelial growth factor
Antigen retrieval was carried out by microwaving the sections for 18 minutes in pH 6 citrate buffer. Serum-free protein block (Dako) was applied prior to incubation of the sections with 1/400 mouse monoclonal anti-VEGF (VG-1) antibody (Abcam 1316) for 1 hour. The Dako EnVision FLEX+ Kit was used thereon. Human breast angiosarcoma and pancreas were used as control tissue. The percentage positive cytoplasmic staining cells and the overall intensity of their staining were estimated, and this was used for the purposes of statistical analysis. The intensity was described as weak, moderate, or strong.
Hypoxia-inducible factor-1α
Pretreatment of the sections was carried out for 30 minutes in a water bath at 97°C in high pH Flex antigen retrieval solution. The sections were incubated in 1/200 mouse monoclonal Hif-1α subunit antibody (BD Biosciences, Wokingham, UK) for 20 minutes using the Dako Autostainer and EnVision FLEX+ Kit. A comedo breast cancer core-cut known to be positive was used as control tissue. Scoring was by a modified H score which estimated by eye the percentage nuclear positive invasive tumour which was used for the statistical analysis and 0, 1+, 2+, and 3+ staining intensity as an average calculated from the 10 HPFs assessed.
Statistical analysis
Correlation of the CT heterogeneity parameters with IHC scores was performed using the Spearman rank correlation coefficient, as the sample size was small and non-symmetrically distributed. Correlations between IHC scores were also assessed using the Spearman rank correlation coefficient. In this exploratory analysis, statistical significance was defined as a 2-tailed P-value of less than .05.
Results
Patients
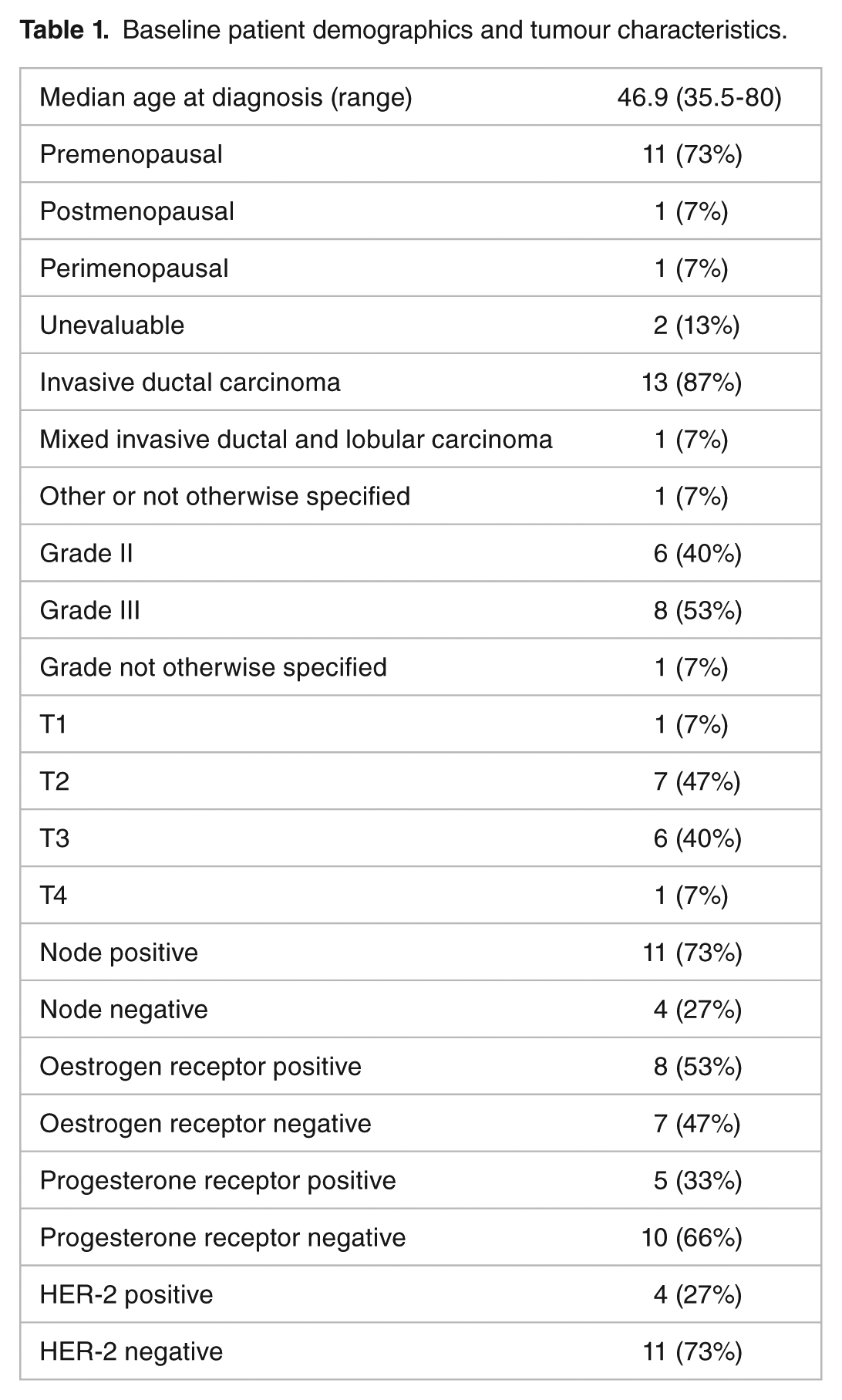
All 20 CT studies were analysed successfully but only 15 patients had an immunohistochemistry outcome; 3 patients had insufficient material to be able to perform further immunohistochemistry testing, and biopsies for 2 patients were unable to be retrieved for further IHC analysis by the laboratory. We therefore report the data for these 15 patients. Baseline patient demographics, tumour characteristics, and clinical outcomes are shown in Table 1.
Baseline patient demographics and tumour characteristics.
Association between CT heterogeneity analysis and IHC
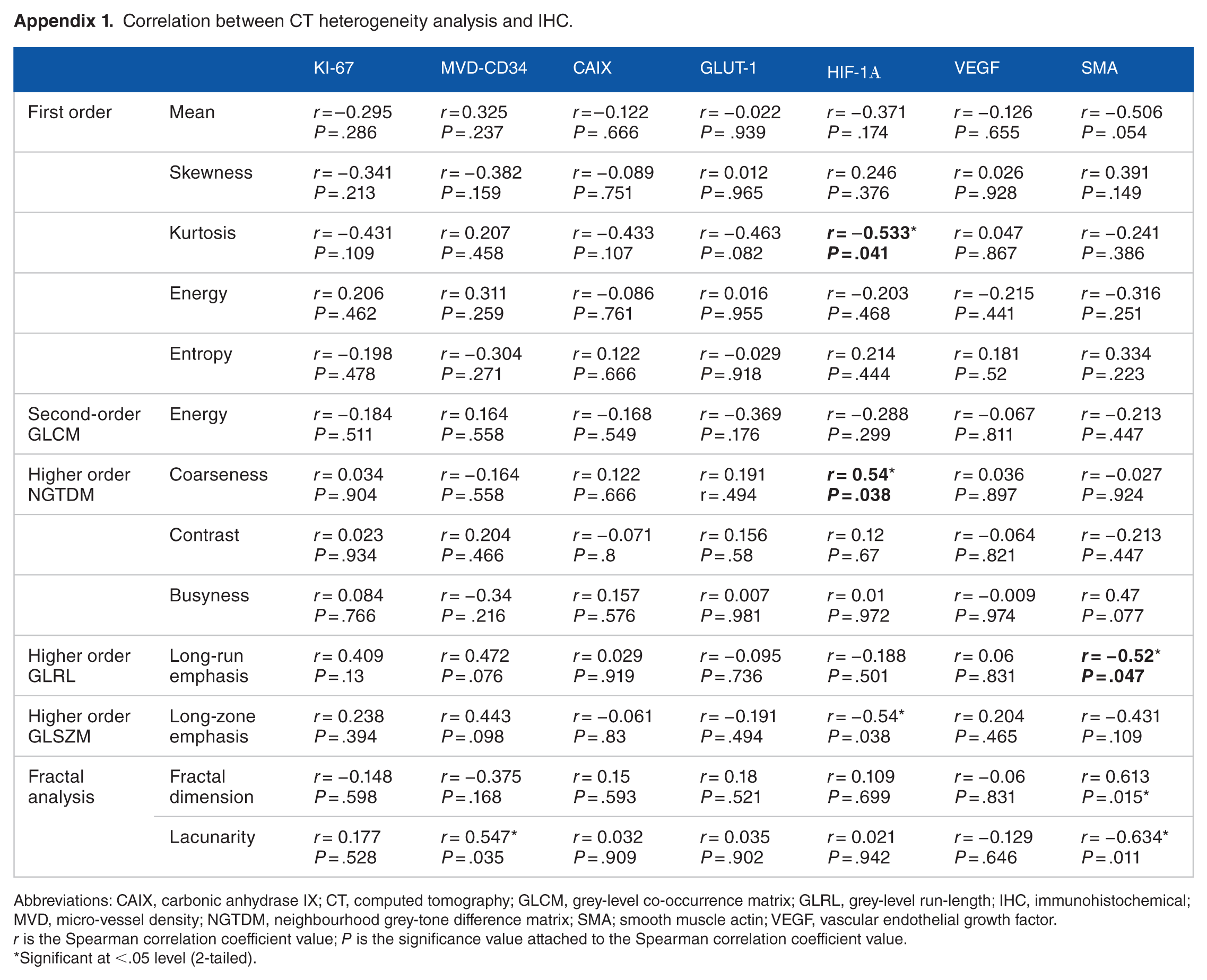
There were a number of significant associations (Table 2; Appendix 1). Hypoxia-related Hif-1α correlated negatively with global kurtosis (r = −0.533, P = .041) and positively with locoregional coarseness (r = 0.54, P = .038) and long zone emphasis (r = 0.54, P = .038). Smooth muscle actin correlated negatively with locoregional run-length long-run emphasis (r = −0.52, P = .047) and global lacunarity (r = −0.634, P = .011) and positively with global fractal dimension (r = 0.613, P = .015). CD34 micro-vessel density (MVD) was also associated positively with global lacunarity (r = 0.547, P = .035). No associations were found with Ki-67 (proliferation), VEGF (angiogenesis), GLUT-1, and CAIX (hypoxia).
Significant correlations between CT heterogeneity analysis and IHC.
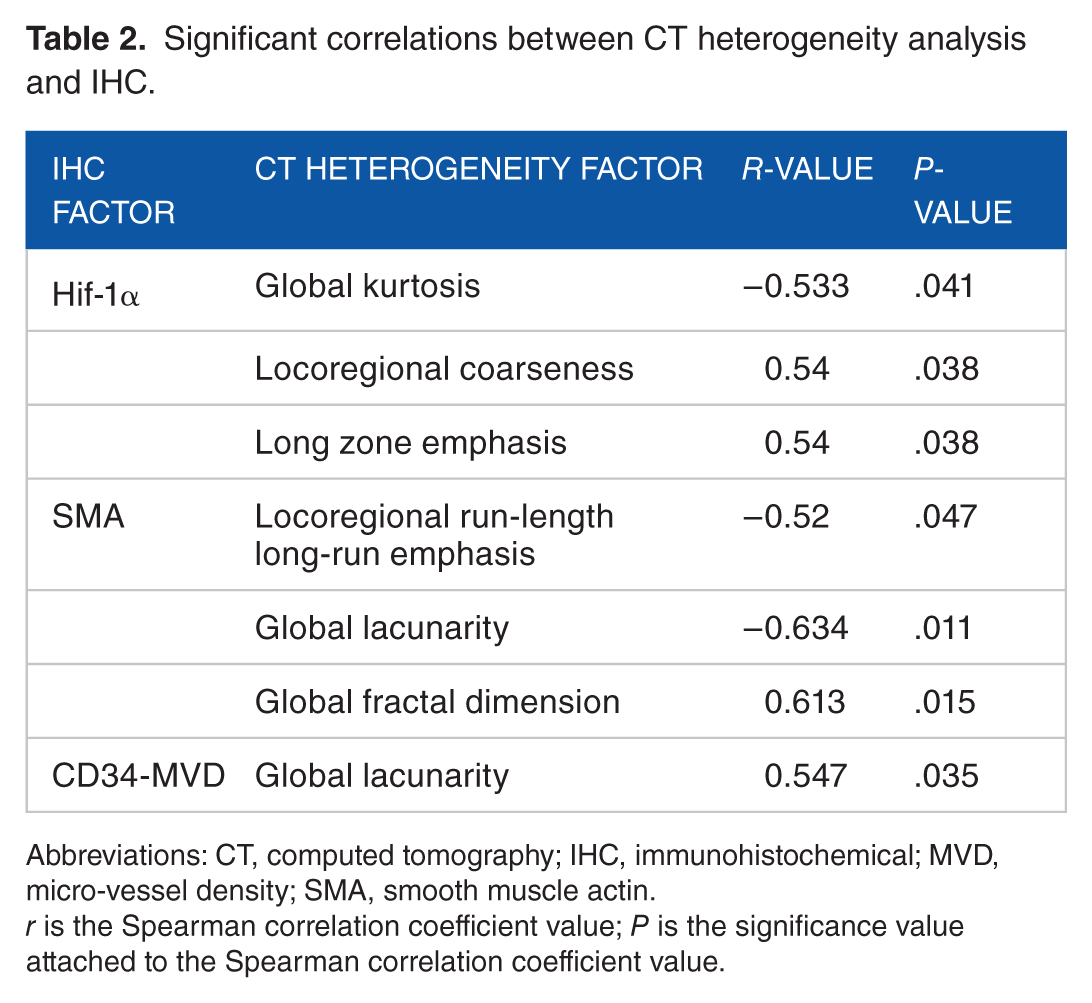
Abbreviations: CT, computed tomography; IHC, immunohistochemical; MVD, micro-vessel density; SMA, smooth muscle actin.
r is the Spearman correlation coefficient value; P is the significance value attached to the Spearman correlation coefficient value.
Associations between IHC parameters
Correlation of the IHC results with each other revealed a significant correlation between GLUT-1 and CAIX (r = 0.659, P = .007), GLUT-1 and MVD (CD34; r = −0.535, P = .04), and SMA and MVD (CD34; r = −0.546, P = .035) (see Supplementary Material S2).
Discussion
In recent years, there have been an increasing number of CT studies that suggested that imaging heterogeneity may be an important predictive and prognostic biomarker.51,52 In this exploratory study, we investigated the associations between primary breast CT image heterogeneity features with IHC markers of underlying tumour biology to inform on further directions of study. While there are a large number of features that can be extracted from an image, we selected a number of standard features, both global and locoregional, that have been reported on in previous publications and found to be clinically relevant.9,50,53
Our study findings suggest that there may indeed be a biological basis to image heterogeneity. Our study found that a number of primary breast CT heterogeneity features correlated with SMA and CD34-MVD as well as Hif-1α suggesting that these features may provide some information regarding underlying vascular heterogeneity. Our study complements data from a previous study correlating CT first-order features with hypoxia and angiogenesis in non-small cell lung cancer. 53 CD34-MVD provides an indication of the distribution of microvessels within a tumour. Smooth muscle actin is expressed by activated fibroblasts (myofibroblasts) and also vascular smooth muscle cells, pericytes, and myoepithelial cells 54 and reflects functional vessel maturity.
In particular, higher image fractal lacunarity, which is a measure of structural heterogeneity within an object, was associated with higher CD34-MVD but lower SMA indicating a relationship with angiogenesis and also areas of lower vessel maturity (lower SMA expression). There was also a positive association between fractal dimension and SMA expression, indicating an association between the spatial regularity within the tumour and increasing vessel maturity. The associations with Hif-1α were also logical in the light of associations with SMA and CD34-MVD. Hif-1α was negatively associated with kurtosis indicating hypoxia is associated with a tendency for platykurtosis, that is, a flatter peak with a negative kurtosis value. On a locoregional level, hypoxia was associated with higher coarseness and run emphasis, that is, greater locoregional heterogeneity.
Previous work has shown that the incorporation of spatial heterogeneity analysis with diffusion weighted (DW) and DCE-MRI may differentiate breast cancer by histological and molecular subtypes.55–57 The use of MRI spatial heterogeneity data has also shown an improvement in the ability to predict response to NAC in breast cancers both in mouse models 58 and humans.56,59 It has also been shown to correlate with Oncotype DX (Genomic Health, Redwood City, CA) recurrence scores, 60 which uses mRNA expression of a 21-gene panel, and is both predictive and prognostic of the outcomes for breast cancer and is widely used in clinical practice to quantify the benefit of adjuvant chemotherapy. 61 Our data add to the literature in supporting a biological basis for CT image heterogeneity and opens the potential for such analysis in the metastatic setting where CT is performed for diagnosis and response assessment.
We acknowledge that our study has a number of limitations. As previously discussed, the number of patients studied in our study was small, although comparable to other similar radiologic-pathologic correlative studies. The patient population also included variation in the traditional IHC markers such as grade, ER, and HER-2 status (Table 1). The immunohistochemistry samples were taken at baseline for correlation with the CT parameters rather than the post-operative specimen. This was necessary in a neoadjuvant setting as the post-operative specimen will be post-chemotherapy, but there are well-known sampling issues with core biopsies which may not represent the heterogeneity of the whole tumour. 62 We also had a relatively high ‘dropout’ rate as we were unable to analyse the core samples in 5 of the original 20 patients for the purposes of this study (25%). Acquisition parameters are known to affect CT texture analysis with kilovoltage and pixel size having a greater effect than milliampere (mA). In our study, we fixed these parameters so as to minimize the variability of the acquisition technique as our focus was the association of the parameters with IHC. Finally, multiple statistical testing was undertaken as part of this exploratory study which is acceptable practice but raises the possibility of type I errors.
In summary, the associations in our study between image heterogeneity features and CD34-MVD, SMA, and Hif-1α suggest a biological basis for these image features and support further investigation to confirm these preliminary findings.
Supplemental Material
Texture_appendix_1a_xyz1487273ad138b – Supplemental material for Assessment of the Spatial Heterogeneity of Breast Cancers: Associations Between Computed Tomography and Immunohistochemistry
Supplemental material, Texture_appendix_1a_xyz1487273ad138b for Assessment of the Spatial Heterogeneity of Breast Cancers: Associations Between Computed Tomography and Immunohistochemistry by David K Woolf, Sonia P Li, Simone Detre, Alison Liu, Andrew Gogbashian, Ian C Simcock, James Stirling, Michael Kosmin, Gary J Cook, Muhammad Siddique, Mitch Dowsett, Andreas Makris and Vicky Goh in Biomarkers in Cancer
Footnotes
Appendix
Correlation between CT heterogeneity analysis and IHC.
| Ki-67 | MVD-CD34 | CAIX | GLUT-1 | Hif-1α | VEGF | SMA | ||
|---|---|---|---|---|---|---|---|---|
| First order | Mean | r = −0.295 P = .286 |
r = 0.325 P = .237 |
r = −0.122 P = .666 |
r = −0.022 P = .939 |
r = −0.371 P = .174 |
r = −0.126 P = .655 |
r = −0.506 P = .054 |
| Skewness | r = −0.341 |
r = −0.382 |
r = −0.089 |
r = 0.012 |
r = 0.246 |
r = 0.026 |
r = 0.391 |
|
| Kurtosis | r = −0.431 |
r = 0.207 |
r = −0.433 |
r = −0.463 |
r = 0.047 |
r = −0.241 |
||
| Energy | r = 0.206 |
r = 0.311 |
r = −0.086 |
r = 0.016 |
r = −0.203 |
r = −0.215 |
r = −0.316 |
|
| Entropy | r = −0.198 |
r = −0.304 |
r = 0.122 |
r = −0.029 |
r = 0.214 |
r = 0.181 |
r = 0.334 |
|
| Second-order |
Energy | r = −0.184 |
r = 0.164 |
r = −0.168 |
r = −0.369 |
r = −0.288 |
r = −0.067 |
r = −0.213 |
| Higher order |
Coarseness | r = 0.034 |
r = −0.164 |
r = 0.122 |
r = 0.191 |
r = 0.036 |
r = −0.027 |
|
| Contrast | r = 0.023 |
r = 0.204 |
r = −0.071 |
r = 0.156 |
r = 0.12 |
r = −0.064 |
r = −0.213 |
|
| Busyness | r = 0.084 |
r = −0.34 |
r = 0.157 |
r = 0.007 |
r = 0.01 |
r = −0.009 |
r = 0.47 |
|
| Higher order |
Long-run emphasis | r = 0.409 |
r = 0.472 |
r = 0.029 |
r = −0.095 |
r = −0.188 |
r = 0.06 |
|
| Higher order GLSZM | Long-zone emphasis | r = 0.238 |
r = 0.443 |
r = −0.061 |
r = −0.191 |
r = −0.54*
|
r = 0.204 |
r = −0.431 |
| Fractal analysis | Fractal dimension | r = −0.148 |
r = −0.375 |
r = 0.15 |
r = 0.18 |
r = 0.109 |
r = −0.06 |
r = 0.613 |
| Lacunarity | r = 0.177 |
r = 0.547*
|
r = 0.032 |
r = 0.035 |
r = 0.021 |
r = −0.129 |
r = −0.634*
|
Abbreviations: CAIX, carbonic anhydrase IX; CT, computed tomography; GLCM, grey-level co-occurrence matrix; GLRL, grey-level run-length; IHC, immunohistochemical; MVD, micro-vessel density; NGTDM, neighbourhood grey-tone difference matrix; SMA; smooth muscle actin; VEGF, vascular endothelial growth factor.
r is the Spearman correlation coefficient value; P is the significance value attached to the Spearman correlation coefficient value.
Significant at <.05 level (2-tailed).
Acknowledgements
The authors thank Frances Daley for her help and advice with the immunohistochemical techniques. They are grateful to the Institute of Cancer Research Academic Partnership Challenge Fund, the Breast Cancer Campaign, and the Breast Cancer Research Trust for funding this study. They also acknowledge the support from the NIHR Biomedical Research Centre of Guy’s and St Thomas’ NHS Foundation Trust in partnership and King’s College London and UCL Comprehensive Cancer Imaging Centre funded by the CRUK and EPSRC in association with the MRC and DoH (England). The funders had no involvement in the study design, data collection and analysis, writing, or submission of the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Institute of Cancer Research Academic Partnership Challenge Fund, the Breast Cancer Campaign, and the Breast Cancer Research Trust for funding this study. They also acknowledge the support from the NIHR Biomedical Research Centre of Guy’s and St Thomas’ NHS Foundation Trust in partnership and King’s College London and UCL Comprehensive Cancer Imaging Centre funded by the CRUK and EPSRC in association with the MRC and DoH (England). The funders had no involvement in the study design, data collection and analysis, writing, or submission of the manuscript.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have made a substantial contribution to this study and approved the manuscript.
Ethical Approval
This study has been approved by the appropriate ethics committee and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed Consent
All patients gave their informed consent to the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
