Abstract
Background
Nicotine pouches (NPs) are smokeless, tobacco-free products gaining global popularity, particularly among youth, for perceived harm reduction. However, many concerns remain regarding their side effects, addictive potential and cardiovascular risks as they avoid the inhalation of smoke but still pose toxicity. Research on NP use patterns in regions such as Saudi Arabia is lacking.
Methods
A cross-sectional survey was conducted online from March to August 2025 targeting NP users aged 18 years or older in Saudi Arabia. The employed questionnaire evaluated demographics, usage habits, health impacts, dependency levels and risk perception. Descriptive statistics and Chi-square tests (P < .05) were used to analyze the collected data.
Results
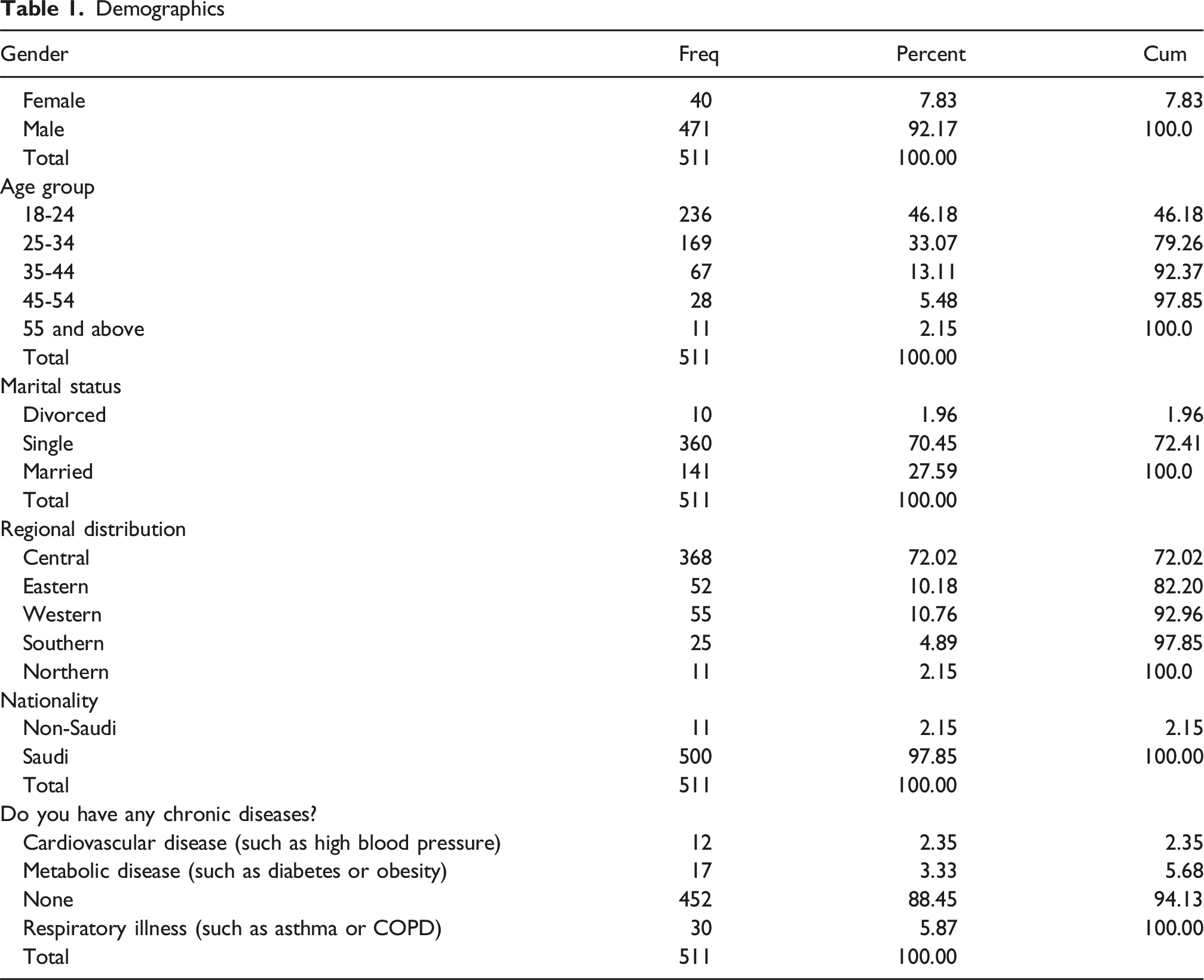
Most participants were male (92.17%), aged 18-24 (46.18%), and Saudi (97.85%), with 88.45% reporting no chronic disease. Nearly half were former smokers (49.51%), 29.35% current, and 21.14% never smokers. The most used NP strength was 10 mg (59.16%), with 61.45% using 1-5 pouches/day; 72.41% reported adequate craving control. Main perceived benefits included being smoke-free (61.45%), odor-free (61.25%), and aiding cessation (58.12%). Common symptoms were nausea (47.55%) and dizziness (32.68%). Craving control was associated with dosage (P = .018), dependence with perceived advantages (P < .001), and quitting intentions with smoking status (P < .001).
Conclusion
Nicotine pouch consumption in Saudi Arabia is prevalent among young current and former smokers, valued for perceived safety, affordability, and cessation support. Stronger strengths and frequent use correlated with greater dependency and motivation to quit. While effective for craving control, reported side effects highlight the need to raise awareness of potential health risks from long-term use.
Implications
An increasing number of young adults in Saudi Arabia, particularly current and former smokers, are adopting NPs as an alternative to traditional tobacco products. Users report that these products assist in managing nicotine cravings and facilitate cessation attempts. Nevertheless, adverse effects such as nausea, dizziness, and alterations in gum health suggests potential health risks. The observed associations among usage patterns, dependence levels, and cessation intentions underscore the necessity for targeted educational initiatives and regulatory measures. Public health strategies should address misconceptions about safety, monitor emerging usage patterns, and ensure policies balance harm reduction potential with prevention of nicotine addiction.
Introduction
Nicotine pouches (NPs) are a relatively new category of smokeless, tobacco-free nicotine delivery products that have attracted attention in public health discussions; however, switching to NPs does not constitute quitting but rather substituting one nicotine product for another. 1 Unlike combustible tobacco products, NPs do not involve inhalation of smoke or exposure to tobacco-specific toxicants. As a result, they significantly reduce exposure to harmful and potentially harmful compounds (HPHCs) when compared to cigarettes, and even Swedish snus, which is already regarded as a less harmful alternative to smoking.2,3
Nicotine pouches have recently gained in popularity across the world, especially among young adults and teenagers alike.4-6 This trend is driven by several factors, including perceptions of less harm than traditional tobacco products and the appeal of flavored options.5,7 In fact, the increasing popularity of NPs, particularly among adolescents and young adults, pose public health concerns due to the lack of regulatory oversight in many countries. According to the 2021 National Tobacco Youth Survey, 1.9% of middle and high school students reported ever using NPs, with 0.8% reporting current use. 8 NPs use is more common among adults, current or former tobacco users, although overall prevalence remains lower than that of e-cigarettes. 9 Stronger policies are needed to prevent never users from initiating use and to discourge current tobacco users from dual or concurrent use with other nicotine products.10,11 This is worrisome as nicotine exposure during adolescence can disrupt brain development and increase long-term addiction risk. 12
Emerging evidence suggests that NPs may support smoking cessation by reducing cravings and decreasing smoking frequency. Nicotine pouches containing higher levels of nicotine appear to be more effective at influencing smoking behavior, while maintaining a lower potential for abuse relative to cigarettes. 13 Those who use NPs are exposed to significantly lower amounts of toxicants than smokers, with toxicant profiles more comparable to nicotine replacement therapies (NRTs). 14 The WHO Clinical Treatment Guideline for Tobacco Cessation in Adults (2024) emphasizes that evidence-based cessation treatment consists of behavioural support and approved pharmacotherapies (such as NRT, varenicline, bupropion, and cytisine). The guideline does not recommend the use of any tobacco product for cessation, nor does it consider tobacco products an evidence-based cessation method.
Despite these benefits, concerns remain regarding potential side effects. Contrary to claims that NPs may aid smoking cessation, current evidence indicates growing concern over their potential to promote youth initiation, dual or poly-use with other nicotine products, and possible long-term oral and cardiovascular health effects.15,16 Recent studies highlight concerns regarding the potential for abuse of nicotine pouches, with documented instances of nicotine poisoning linked to their repeated use. 17 High nicotine content pouches can result in rapid nicotine uptake, increasing heart rate and arterial stiffness, which may pose cardiovascular risks. 18 Additionally, users frequently report mouth irritation across all nicotine strengths, and high-dose NPs may carry addiction risks comparable to cigarettes.19,20
Nicotine pouches are often used in conjunction with other tobacco products, such as cigarettes and e-cigarettes. This dual use is prevalent among young adults who are already using non-combustible or combustible tobacco products.5,6,21 While NPs have gained popularity globally, there is a notable lack of region-specific research, particularly in Saudi Arabia. This study aimed to examine the patterns and perception, and side effects of nicotine pouch use among adults in Saudi Arabia.
Method
Study Design and Study Participants
This research utilized an online survey to assess nicotine pouch use patterns, perceptions, and associated side effects among adults in Saudi Arabia. Data collection took place between March 12, 2025, and August 01, 2025.
Ethical Consideration
Ethical approval was obtained from an Institutional Review Board at King Saud University (IRB Approval of Research Project No. E−25-9494).
Study Participants
Because there is no defined sampling frame for nicotine pouch users in Saudi Arabia, a probability-based sampling strategy was not feasible. Consequently, we used a non-probability convenience sampling approach, recruiting participants via social media platforms and online networks. To contextualize the precision of the estimated proportions, we conducted a post-hoc precision estimate using the standard formula for a single population proportion with 95% confidence and a 5% margin of error under conservative assumptions (P = .50). Under these assumptions, the minimum required sample size is approximately 384 participants. The achieved sample size in this study (n = 511) exceeds this threshold, suggesting acceptable precision for estimating proportions within these assumptions, while recognizing that the use of convenience sampling still limits statistical inference and external validity.
The target Participants for this study include adult users of nicotine pouches in Saudi Arabia. Inclusion is limited to individuals aged 18 years and above who actively use nicotine pouches, ensuring that participants are legally able to consent and are representative of the adult population engaging in this study.
Participants were recruited through an online approach to maximize reach and diversity. The online questionnaire was shared via popular social media platforms such as Telegram, WhatsApp, Twitter (X). These platforms were selected because they are widely used within the target population and allow rapid, high-volume dissemination and sharing of survey links, making them effective channels for reaching diverse participants. This method is intended to receive responses from various regions, age groups, and socioeconomic backgrounds, therefore improving the generalizability of the study results. Participation was anonymous, and respondents were informed that the questionnaire should only be completed once and the form was configured to accept only one response per Google account.
All participants were presented with an online informed consent form preceding the survey. The consent process clearly explained the study’s purpose, procedures, and voluntary nature of participation. Participants were required to provide written electronic consent by selecting the “I agree to participate” option before accessing the questionnaire. Consent was thus electronically recorded through the survey platform, and no personally identifiable information was collected.
The online survey was programmed so that all questions were mandatory, and participants could not submit the survey unless every item was answered. Consequently, only fully completed questionnaires were stored, and there were no missing data for the variables used in this study. We also checked the dataset for obvious out-of-range or impossible values and did not identify any implausible responses; therefore, all completed questionnaires were included in the analyses.
Questionnaire Tool
The questionnaire used in this study consisted of 22 structured questions, designed to assess patterns of use, perceptions, and potential health effects of nicotine pouch (NP) consumption among adults in Saudi Arabia. The tool was developed in Arabic and English and distributed digitally. A group of academicians with expertise in smoking behaviour developed this questionnaire. Subsequently, experts evaluated its face and content validity, followed by a pilot test. A pilot test was conducted with 10 participants, who were representative of the target population of adult nicotine pouch users, to assess the clarity and suitability of the questionnaire. Notably, participants involved in the pilot testing phase were excluded from the primary study. Based on the pilot test and expert feedback, adjustments were made, including refining the wording of certain items for clarity, reorganizing the order of questions to improve flow, and modifying response options to ensure they appropriately captured the intended information. The internal consistency reliability of the questionnaire was assessed using Cronbach’s alpha, yielding a coefficient of 0.84, which indicates good internal consistency among the items. The revised version was then distributed for data collection (See Supplementary Material). The final version comprised five sections:
Section 1: Demographics and Health Background
This section collected information on participant ID, region of residence, nationality, gender, age group, marital status, and chronic health conditions.
Section 2: Use Patterns and Exposure
Participants were asked about their current and past use of both nicotine pouches and traditional smoking, including usage frequency, dosage, and comparisons between NP and cigarette use.
Section 3: Health-Related Observations
This section explored self-reported oral and gum health changes due to prolonged NP use, as well as any physical symptoms experienced during use.
Section 4: Perceived Effects and Dependence
Questions assessed the degree of psychological or physiological dependence on nicotine pouches and their perceived effects on mood, alertness, and stress relief.
Dependence on nicotine pouches was assessed using a single self-report item asking participants to classify their current level of dependence (full dependence with increasing use, full dependence without increasing use, partial dependence, no dependence). For analyses, this item was treated as an ordinal indicator of perceived dependence. Daily frequency of nicotine pouch use was assessed with the question, ‘How many times a day do you use nicotine pouches?’ and analyzed as a behavioral indicator of dependence (higher values indicating greater use).
Section 5: Perceptions and Harm Reduction Potential
This section is designed to capture participants’ perceptions of how well nicotine pouches reduce cravings, their safety and affordability compared to smoking, and their perceptions of nicotine pouches as a tool for quitting cigarette smoking.
Craving control was measured using a single item asking whether nicotine pouches sufficiently controlled nicotine cravings compared to smoking (response options: sufficient vs insufficient, cravings increase over time), which was analyzed as a binary variable.
Statistical Analysis
Descriptive statistics were used to summarise the study data. Categorical variables were presented as frequencies and percentages, and comparisons between groups were performed using appropriate inferential statistical tests for the data type, including the Chi-square test for categorical variables. Statistical significance was defined as a P-value <.05.
Multivariable logistic regression analyses were conducted to examine factors associated with reporting nicotine pouches as “not sufficient” and with increased craving. Smoking status, gender, nationality, age group, and nicotine strength were included as independent variables, with appropriate reference categories specified for each. All variables were entered simultaneously into the models based on conceptual relevance and prior evidence rather than stepwise selection procedures. Standard diagnostic checks were performed to assess model assumptions, including evaluation of multicollinearity and overall model fit, and no violations were identified.
For analyses examining perceived advantages, each advantage was treated as a binary outcome and analysed using a separate logistic regression model with dependence status entered as the independent variable. Results from all logistic regression analyses were reported as odds ratios (ORs) with 95% confidence intervals (CIs). Estimates derived from categories with limited sample sizes were interpreted cautiously due to the potential for reduced precision and unstable effect estimates.
All statistical analyses were performed using STATA, and figures were prepared using Microsoft Excel.
Results
Demographic Characteristics
Demographics
Nicotine Use Behavior
Descriptive of Nicotine Use

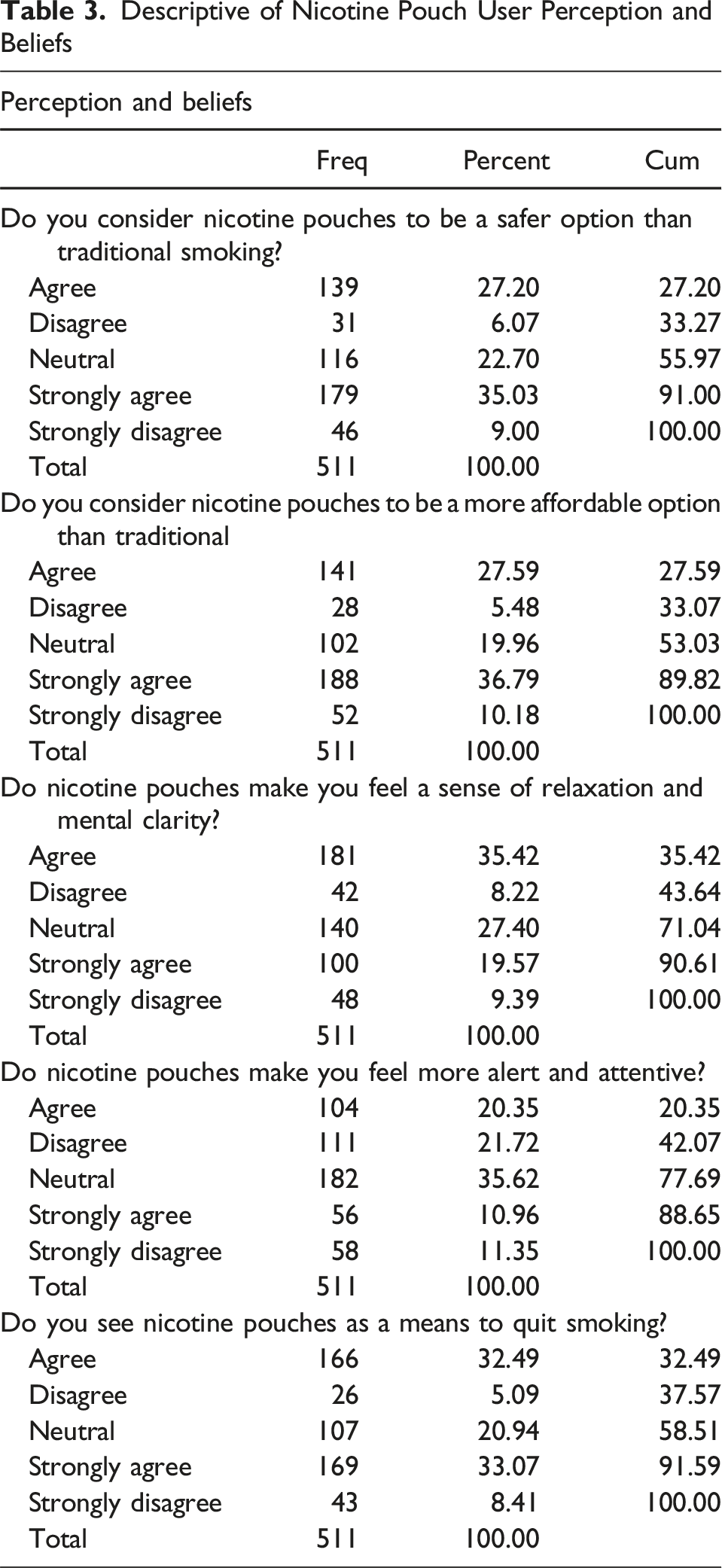
Perceptions and Beliefs
Descriptive of Nicotine Pouch User Perception and Beliefs
Perceived Advantages
Perceived advantages included being smoke-free (61.45%), having no smell (61.25%), and acting as a means to quit smoking (58.12%). Other advantages cited were affordability (36.20%), flavor variety (24.27%), and variety of nicotine strengths (22.11%).
Reported Symptoms
Reported symptoms among users included nausea (47.55%), dizziness (32.68%), shaking (16.05%), difficulty concentrating (15.46%), breathing difficulty (12.13%), seizures (3.33%), and fainting (1.96%). Notably, 29.16% reported no symptoms at all.
User Perceptions of Craving Control
User Perceptions of Craving Control in Relation to Smoking Status

Note. ***P < .001.
User Perceptions of Craving Control in Relation to Nicotine Pouch Dosage

Logistic Regression Analysis of Factors Associated With the Perceived Sufficiency of Nicotine Pouches
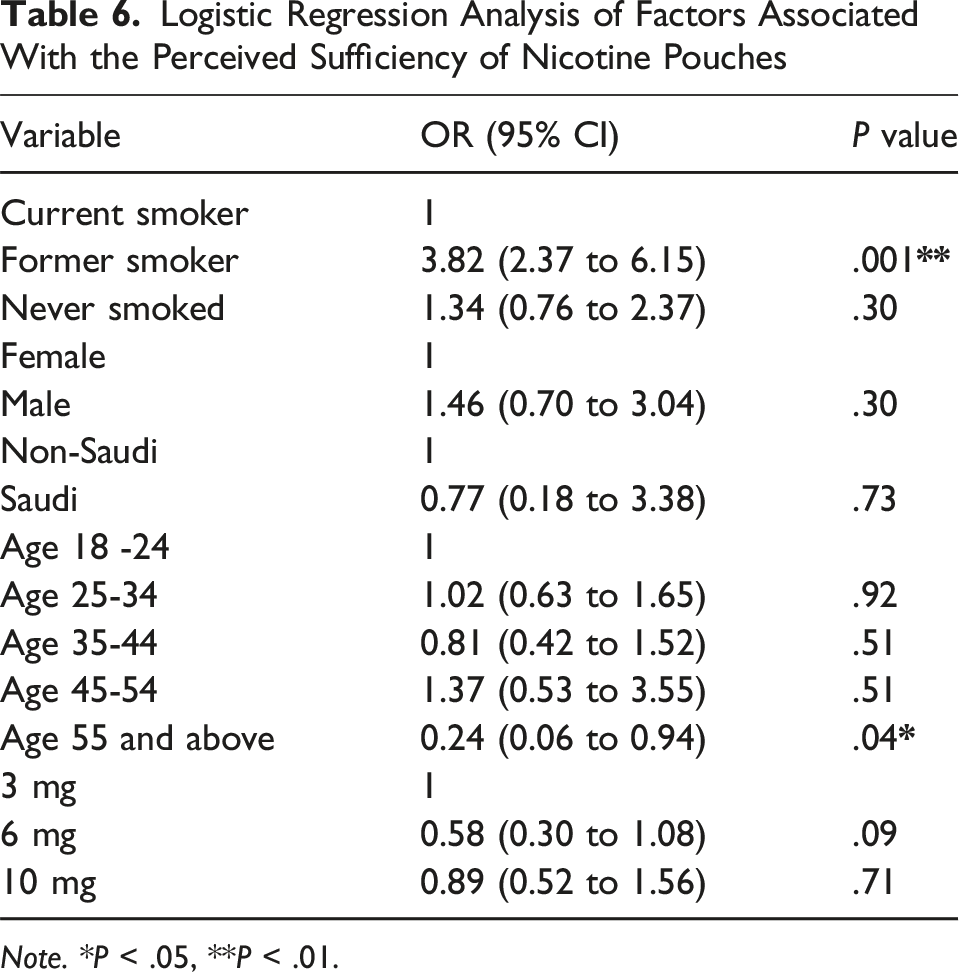
Note. *P < .05, **P < .01.
User Perceptions of Dependence vs Perceived Advantages
Dependence vs Advantages
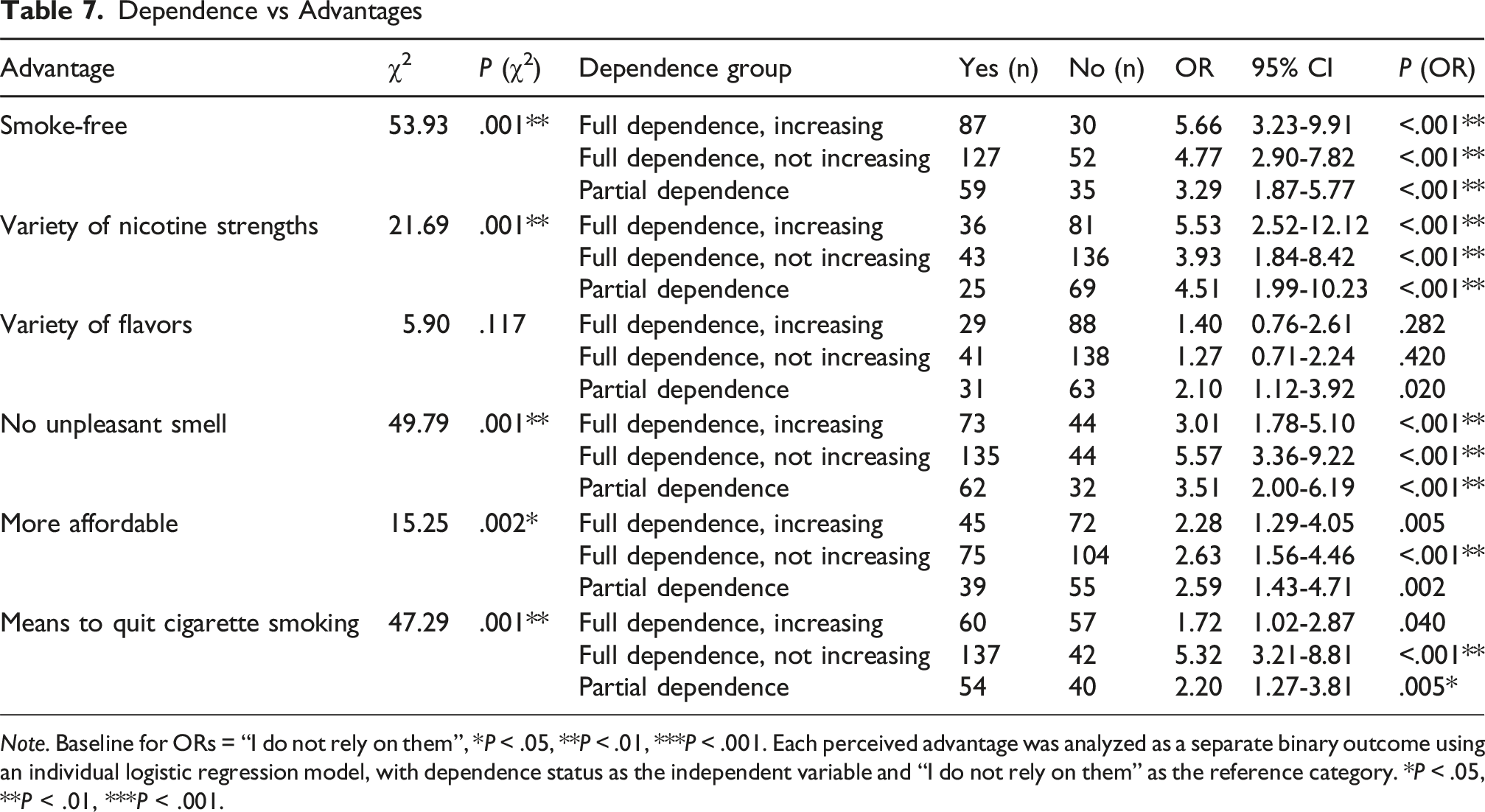
Note. Baseline for ORs = “I do not rely on them”, *P < .05, **P < .01, ***P < .001. Each perceived advantage was analyzed as a separate binary outcome using an individual logistic regression model, with dependence status as the independent variable and “I do not rely on them” as the reference category. *P < .05, **P < .01, ***P < .001.
User Perceptions of Means to Quit vs Other Variables
Comparison of Using Pouches as a Means to Quit

Note. ***P < .001.
Discussion
The findings indicate that the majority of NP users were former smokers who predominantly used the highest available strength (10 mg), often with the intention of supporting smoking cessation. Most participants reported believing that NPs could help them manage nicotine cravings during attempts to quit smoking. They also perceived NP use as adequate in satisfying their nicotine needs. The smoke-free nature and absence of odor were the most frequently cited advantages.
In our study, most respondents were former smokers who transitioned to NPs using high dosages. They reported that NP use was perceived as sufficient to manage or reduce cravings for nicotine. This subjective perception of sufficiency was reflected in our logistic regression analysis, which identified smoking status as a significant predictor of reporting NPs as sufficient. Compared with current smokers, former smokers had nearly four times the odds of reporting NPs as sufficient, indicating that former smokers were more likely to perceive NPs as an adequate substitute for combustible tobacco products. In contrast, never smokers and current smokers did not differ significantly in their perceived sufficiency of NPs, suggesting that perceptions of how effective NPs are in managing cravings may be related to prior experiences of nicotine dependence. These findings align with a survey conducted across six major U.S. metropolitan areas, which concluded that young adult former smokers viewed NPs as less harmful and less addictive than combustible tobacco products, despite recognizing that all tobacco products, including pouches, remain harmful. 11
Participants expressed a strong belief that NP use is safer than combustible tobacco products, more affordable, and associated, in their experience, with relaxation and satisfaction. 11 Although these perceptions may vary depending on dosage, most of our participants used 10 mg pouches. Interestingly, participants also reported an equal perception regarding NP use and its potential, from their perspective, to enhance alertness and mental clarity. These results are consistent with a meta-analysis pooling experimental studies on acute nicotine administration via patches, nasal spray, or combustible tobacco which concluded that nicotine generally enhances attention, alertness, and reaction speed, with no significant differences between deprived smokers and users of modern nicotine delivery systems such as pouches, as the active substance remains nicotine. 22 It is worth noting that, although these findings are consistent, important differences exist between nicotine pouches and pharmaceutical nicotine products, particularly regarding their chemical composition and nicotine absorption profiles.
The smoke-free and odorless characteristics of NPs were particularly appealing to users, as these features allow discreet use in enclosed spaces without attracting attention. This observation concurs with findings from the Swedish study “Power in a Pouch”, which reported that NPs offer a socially acceptable alternative to smoking by replicating the sensory experience without producing smoke or vapor. 23 Furthermore, over half of our participants considered the perceived role of NPs in aiding quitting to be a major benefit. Although a randomized 4-week trial showed reductions in cigarette consumption when nicotine pouches were available and used, it did not demonstrate a reduction in overall tobacco-related harm exposure, as biomarkers of exposure (NNAL) did not decrease. 13
Given that NPs contain nicotine, the primary component of combustible tobacco, similar post-use symptoms were expected. Reported symptoms varied depending on dosage, frequency, and individual tolerance. Nausea emerged as the most frequently reported symptom, followed by changes in gum health and dizziness. Shaking and difficulty concentrating were also noted, while seizures and fainting were rare. These findings align with a cross-sectional study that identified nausea as one of the most common adverse events associated with NP use.24,25 Other literature has reported adverse effects predominantly localized to the oral cavity. 16
In terms of subjective satisfaction, former smokers in our study reported that NPs provided, from their perspective, a level of satisfaction comparable to combustible tobacco. The preference for the 10 mg dose suggests that higher strengths may be perceived as providing better craving control. In agreement with this observation, a 4-week study on adult former smokers reported a tendency to prefer NPs over other nicotine delivery forms. 13 Notably, our results indicate no association between NP use frequency and craving control, possibly reflecting individual variability in nicotine tolerance.
Our data also highlight several product attributes, such as smoke-free use, multiple strength options, absence of odor, affordability, and perceived cessation support as major factors influencing NP uptake. Among these, the strongest association with dependence was linked to the smoke-free feature. The odorless quality was also valued, as it enables discreet use in diverse settings. These findings are consistent with a cross-sectional survey of NP users that assessed use patterns, dependence levels, and adverse events using the modified Fagerström Test for Nicotine Dependence Smokeless Tobacco (FTND-ST). That study reported a mean dependence score of 7, indicating substantial dependence, with users averaging 2 ± 1 pouches per session over approximately 11 ± 7 min; primary motives for use were flavor preference (53%), nicotine level (25%), and brand loyalty (22%). 25 In contrast, our findings did not demonstrate a relationship between flavor preference and dependence, diverging from the cited survey. 25 However, The FDA has not approved any nicotine pouches to help people quit smoking, 26 these perceived benefits may not translate into successful quitting, highlighting the need for further evaluation of their long-term impact on dependence and smoking behaviors.
Our analysis revealed that former smokers, users of higher-dose pouches (eg, 10 mg), frequent users, and those with greater dependence were significantly to perceive nicotine pouches as a quitting aid. This finding aligns with findings that interest in using nicotine pouches is higher among adult smokers planning to quit—especially those with prior quit attempts—indicating intentions to leverage pouches as cessation tools. 27 From a pharmacological standpoint, higher-dose pouches deliver nicotine in a dose-dependent manner that can approach levels from cigarettes and provide acute craving relief 28 ; such potent delivery may explain the linkage between dependence severity and the perception of pouches as quit aids. Tobacco cessation should be guided by evidence-based approaches, as outlined in the WHO Clinical Treatment Guideline for Tobacco Cessation in Adults (2024). Moreover, a recent comprehensive review reported that there is currently no published evidence supporting the effectiveness of nicotine pouches as a smoking cessation intervention, highlighting continued uncertainty regarding their utility in helping users quit. 29
Strengths and Limitations
This study has several strengths. It represents, to our knowledge, one of the first nationwide investigations in Saudi Arabia to compare nicotine pouch use with cigarette smoking, providing comprehensive data on prevalence, perceived benefits, and reported side effects across diverse demographic and regional groups. The large-scale, online format enabled broad geographic reach and anonymity, which may have encouraged more honest reporting on a culturally sensitive behavior. The questionnaire included items addressing different dimensions of product use to provide a more complete understanding. Moreover, by aligning the results with previously published studies, the study reinforces the relevance and consistency of its observations. However, certain limitations should be acknowledged. The cross-sectional design precludes causal inference, and reliance on self-reported data introduces the potential for recall inaccuracies and social desirability bias. As participation was limited to individuals with internet access and a willingness to respond, selection and nonresponse biases may have affected representativeness. This study used convenience sampling via online recruitment (no available sampling frame for nicotine pouch users in Saudi Arabia), which may introduce selection bias and limit representativeness and generalizability. Reported benefits and side effects were not clinically verified, and some misclassification of product type is possible. Multiple Saudi data sources show substantially higher tobacco use in men than women including nationally representative surveys (GATS 2019: 30.0% vs 4.2%; Saudi Health Interview Survey: 23.7% vs 1.5%) and other national survey analyses so our male-predominant sample may partly reflect underlying sex differences; however, women’s use may also be underreported due to stigma, and the very small number of female participants limits generalizability especially to women.30-34 Finally, unmeasured confounding factors may have influenced both product choice and perceptions, and changes in the tobacco market following data collection could affect the applicability of our prevalence estimates.
Conclusion
The findings indicate that nicotine pouch use is most prevalent among young, predominantly male Saudi nationals, with many being former or current smokers. Users commonly perceive nicotine pouches as safer, more affordable, and an effective aid for smoking cessation, particularly among those using higher nicotine strengths and those with prior smoking history. Significant associations were observed between dependence levels and key perceived advantages, as well as between smoking cessation perceptions and variables such as dosage, frequency of use, and dependence. Notable side effects, including nausea and dizziness, and changes in gum health, were relatively common. Overall, nicotine pouches appear to be widely accepted for craving control and perceived cessation support; however, it is important to note that nicotine pouches are not approved by the U.S. Food and Drug Administration (FDA) as smoking-cessation tools, and their use is not without reported health impacts.
Supplemental Material
Supplemental Material - Patterns and Perception of Nicotine Pouches Use Among Adults in Saudi Arabia
Supplemental Material for Patterns and Perception of Nicotine Pouches Use Among Adults in Saudi Arabia by Abdulrhman S. Alghamdi, Abdullaziz F. Albukhait, Faisal S. Aldosari, Adel S. Alkahtani, Khalid S. Alwadeai, Saleh S. Algarni, Abdulelah M. Aldhahir, Mushabbab A. Alahmari, Wayil H. Alanazi, and Rayan A. Siraj in Tobacco Use Insights
Footnotes
Acknowledgments
The authors would like to acknowledge the support provided by the Ongoing Research Funding Program (ORF-2025-1131), King Saud University, Riyadh, Saudi Arabia. The authors acknowledge the use of artificial intelligence (ChatGPT by OpenAI) for English language editing and refinement of the manuscript. No content generation or analysis was performed using AI.
Consent for Publication
All participants provided consent for publication.
Author Contributions
All authors were actively involved in the study’s conception, design, execution, or analysis, contributed to writing or reviewing the manuscript, approved the final version, agreed on the journal of submission, and take full responsibility for the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
